Impact of High Pressure Homogenization (HPH) Treatment on the Nutritional Quality of Egg/Yogurt, Vegetable and Fruit Based Creams ()
Keywords:High Pressure Homogenization; Nutritional Quality
1. Introduction
It is well known that thermal processing generally applied for extending shelf life of food products can affect food nutritional value due to nutrient and bioactive sensitivity to heat. In addition, thermal processing can cause the parallel formation of dangerous oxidized by-products. Lipid oxidation is one of the major degradative processes in food; it results in significant generation of cytotoxic compounds and free radicals which can oxidize vitamins, thus impairing the nutritional quality [1]. Degradation of lipids is accelerated by high temperature and high energy radiation.
Food composition tables are representative of foodstuffs consumed in their raw state, and they do not consider that concentration, availability, and biological activity of nutrients may be changed by processing. Due to the relative instability of many compounds, thermal processing can profoundly affect the nutritional value of vegetables [2] which contain many nutritional and healthful constituents, including minerals, vitamins, dietary fibers, and phytochemicals, and are considered to be the major contributors of antioxidant dietary intake [3]. As an example ascorbic chemical oxidation and/or thermal degradation is a consequence of pasteurization and sterilization [4].
In this light the development of mild non thermal processing is a key factor for preserving or improving the nutritional value of food. In this perspective, high pressure homogenization (HPH), which ensures product stability, shelf life, and taste [5] is considered one of the most promising non thermal food-processing technologies. Multipass homogenization treatment increases the safety features of food; Patrignani et al. [6] evidenced that the multi-pass high pressure treatment at 100 MPa of liquid whole egg inoculated with 7 and 4 log CFU/ml S. enterica serovar enteritidis results in a first order inactivation kinetic. Furthermore, the HPH ability to potentiate the antimicrobial activity of naturally occurring egg antimicrobials is reported [7,8].
Hyperbaric treatment can also have a positive impact on the technological characteristics of food; as an example, it increases the foaming capacity of liquid whole egg, while having a minor impact on product viscosity. This allows a more flexible utilization of whole liquid eggs in several based-egg products [6].
Although nowadays HPH treatment is a celebrated processing option experiencing worldwide commercial growth, little is known on its impact on the nutritional value of food. In this study we have evaluated the HPH induced nutritional modifications occurring in: 1) an egg-yogurt cream (EYC) formulated with different NaCl concentrations; 2) a mixed vegetable cream (MVC); and 3) a mixed fruit cream (MFC). EYC nutritional quality was evaluated measuring lipid peroxidation, triglyceride hydrolysis, fatty acid composition, and vitamin E content. MVC and MFC nutritional quality was assessed by measuring total antioxidant activity (TAA), and vitamin C content.
2. Materials and Method
2.1. Materials
Chloroform, methanol, isooctane, isopropanol, diethyl ether, ethanol and n-hexane were purchased from Carlo Erba (Milan, Italy); 2,2’-azino-bis-(3-ethylbenzothiazoline- 6-sulfonic acid) (ABTS), iodine, potassium iodide, phenolphthalein and anhydrous sodium sulfate were obtained from Sigma (Milan, Italy). All other chemicals and solvents were of the highest analytical grade.
2.2. Egg-Yogurt Cream (EYC) Formulation
The formulation used for the preparation of EYC was 60% (w/w) sunflower oil, 17% (w/w) water, 13% (w/w) yogurt, 10% (w/w) egg, and NaCl at different concentrations (0%, 1% and 2%). Briefly, whole egg and salt were mixed at 700 rpm attended by sunflower oil. Yogurt and water were added and mixed with other components at 50 rpm. Last, citric acid was included to adjust pH to 5.
2.3. Mixed Vegetable (MVC) and Fruit (MFC) Creams Formulation
The mixed vegetable cream had the following composition: 28.6% (w/w) zucchini, 28.6% (w/w) potatoes, 28.6% (w/w) beans and 14.2% (w/w) water. The mixed fruit cream was made of 32.2% (w/w) fresh orange pulp, 32.2% (w/w) fresh carrots, 32.2% (w/w) apricot juice and 3.4% (w/w) water.
After mixing the ingredients (vegetable or fruits) at 250 rpm under vacuum for 60 sec, a blenching (82˚C for 180 sec) was performed to inactivate endogenous enzymes. Finally water was added and the mixture cooled down to 2˚C under agitation at 2800 rpm.
2.4. HPH Treatment
A continuous industrial-scale high pressure homogenizer (Niro Soavi, Parma, Italy) was used for all the homogenizing treatments (maximum pressure = 400 MPa). The machine was supplied with R valve. In order to avoid the temperature increase, the products were cooled down to 4˚C by using a thermal exchanger (Niro Soavi, Parma, Italy). The EYC, MVC and MFC samples were subjected to HPH treatments at different pressure levels up to 400 MPa. In particular the pressure values applied for both MVC and MFC were 0.1, 100, 200, 300 and 400 MPa. For the EYC the following pressures were used: 0.1, 70, 100 and 150 MPa. The time of treatment was constant and istantaneus (1.6 ms) regardless the pressure levels and the food matrices.
2.5. Lipid Extraction and Fatty Acid Composition Analysis
Total lipids were extracted from EYC according to Di Nunzio et al. [9], and methyl esterified according to Di Nunzio et al. [10]. Fatty acid composition (as methyl esters) was determined by gas chromatography (GC 8000, Fisons, Milano, Italy) using a capillary column (SP 2340, 0.2 µm film thickness) at programmed temperature (160˚C - 210˚C, with a gradient of 8 ˚C/min) as previously reported [11]. The gas chromatographic peaks were identified on the basis of their retention time ratio relative to methyl stearate and predetermined on authentic samples. Gas chromatographic traces and peak area evaluations were obtained using a Chrom Card Softwer (Thermo Electron Scientific, Milan, Italy) computing integrator.
2.6. Triacylglycerol Hydrolysis.
Determination of free acidity, considered an indicator of triacylglycerol hydrolysis, was performed by titration using NaOH as the titrant for the free acids [12]. Free acids contained in 1 g of sample were dissolved in a neutralized diethyl ether/ethanol (2:1, v/v) solution, and titrated while stirring with 0.01 N NaOH using 1% (w/w) phenolphthalein solution for the detection of the endpoint. Titration was carried out till the formation of persistent (>10 s) slight violet colour. Free acidity was expressed as percentage of linoleic acid content, since linoleic acid was the most abundant fatty acid in the sample.
2.7. Conjugated Diene level
Oxidative damage was assayed in EYC by quantifying the content of conjugated dienes. Conjugated dienes were extracted from 0.1 g sample according to Chouinard et al. [13] using 3.6 mL of n-hexane/isopropanol (3:2, v/v) solution, followed by through mix and the addition of 2.4 mL of 7% sodium sulfate anhydrous solution (w/v). After phase separation, the upper layer was collected and transferred to a test tube, dried under nitrogen infusion, and the lipid residue was redissolved in 25 mL isooctane. Absorbance was measured at 232 nm against blank, and conjugated diene concentration expressed as specific extinctions (Kλ) calculated by the following formula (1):
 (1)
(1)
where g is gram of sample weighted and V the dilution.
2.8. Vitamin E content
Ten grams of EYC underwent cold saponification using 5 ml KOH 50%, 100 mL ethanol, and 200 mg of antioxidant mixture (butylated hydroxytoluene, EDTA, vitamin C). Samples were shaken for 15 min, incubated in the dark overnight, extracted twice with n-hexane, filtered, evaporated under vacuum, and resuspended in 10 mL of n-hexane. Vitamin E content was determined by direct phase normal-phase high performance liquid chromatography (NP-HPLC) using a Helwett Pakard-serie1100 Agilent apparatus equipped with a 5-μm Luna Si-60—Phenomenex column (250 × 4.6 mm i.d.). The mobile phase was n-hexane/isopropanol (99.2/0.8), in isocratic at a flow rate of 1.5 mL/min. The UV detector was set at 290 nm. Vitamin E content was calculated by comparison of the respective peak areas to standards, and expressed as mg/100g sample. Chromatograms were recorded and processed using HP Chemstation software.
2.9. Total Antioxidant Capacity (TAC)
MVC and MFC were homogenized in a food processor, and 10 g of the homogenized material were dissolved in 30 ml of cold 5 mM phosphate buffered saline, pH 7.4. Samples were filtered, and the resulting filtrate was used to determine the TAC. All phases of homogenization were done in ice, and each filtrate immediately analysed.
TAC was measured as previously reported [14], on the basis of the ability of the antioxidant molecules in the sample to reduce the radical cation of ABTS, determined by the decolorization of ABTS+•, and measured as the quenching of the absorbance at 734 nm. Values obtained for each sample were compared to the concentrationresponse curve of the standard trolox solution and expressed as micromoles of trolox equivalent (TE)/100g sample.
2.10. Vitamin C Concentration
Vitamin C content in MVC and MFC was assessed according to Ciancaglini et al. [15]. Samples were homogenized in a food processor without addition of any solvent, and 10 g of the homogenized material was dissolved in 30 ml of cold water, getting a dilution 1:3 w/v. Samples were then filtered and the resulting filtrate was titrated while stirring with 0.005 M I2 using starch indicator for the detection of the end point. Titration was carried out till the formation of persistent (>10 s) blueblack starch-iodine complex, and vitamin C concentration was expressed as mol/100g sample.
2.11. Statistical Analysis
Data are reported as mean ± SD. Differences were tested for statistical significance by the one way ANOVA using Tukey’s as post test. P values less than 0.05 were considered statistically significant.
3. Results
The EYC fatty acid composition is reported in Figure 1(a). The most concentrated fatty acids were linoleic > oleic > palmitic > stearic acid; myristic, palmitoleic, α-linolenic and docosanoic acid were also detected in traces. Neither NaCl concentration (data not shown) nor the pressure used for treatment caused significant modifications in the fatty acid composition. A representative chromatogram is shown in Figure 1(b).
Free acidity was influenced by NaCl concentration, while no differences related to HPH treatment were detected comparing samples with the same NaCl content (Figure 2).
As well, HPH did not cause any increase in lipid peroxidation, measured as concentration of conjugated diene containing lipids, independent of NaCl concentration (Figure 3).
In samples treated at atmospheric pressure (0.1 MPa)
 (a)
(a) (b)
(b)
Figure 1. Fatty acid composition of egg-yogurt cream after different HPH treatments (a) and representative chromatogram (b). Data are means +/− SD. Statistical analysis was by the one way ANOVA (n.s.), using Tukey’s as post test: no significant differences were detected among samples treated with HPH at different pressure.

Figure 2. Free acidity of egg-yogurt cream with different NaCl concentration after different HPH treatments. Data are means +/− SD. Statistical analysis was by the one way ANOVA (p < 0.01), using Tukey’s as post test. Different letters indicate statistical significance (at least p < 0.05).
and at 70 MPa HPH, vitamin E content appeared dependent on NaCl concentration, being about 40% lower in samples containing 1% or 2% NaCl (Figure 4). No detrimental effect due to NaCl concentration was evident in samples treated with the two highest HPH. The highest vitamin E content was observed in the 1% NaCl sample treated at 150 MPa.
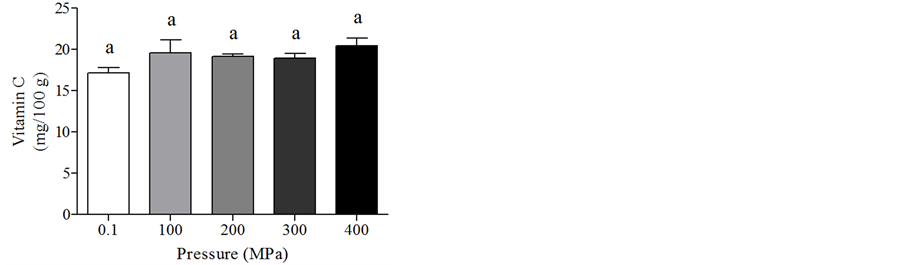
In the MVC, 300 MPa HPH treatment significantly increased the TAC (Figure 5(a)), while vitamin C content was similar in all tested conditions (Figure 5(b)).
In the MFC, HPH processing did not modify the TAC (Figure 6(a)), while vitamin C increased using the 200

Figure 3. Concentration of conjugated diene containing lipids in egg-yogurt cream with different NaCl concentration after different HPH treatment. Data are means +/− SD. Statistical analysis was by the one way ANOVA (n.s.), using Tukey’s as post test: no significant differences were detected among samples.

Figure 4. Vitamin E content in egg-yogurt cream with different NaCl concentration after different HPH treatments. Data are means +/− SD. Statistical analysis was by the one way ANOVA (p < 0.001), using Tukey’s as post test. Different letters indicate statistical significance (at least p < 0.05).
 (a)
(a) (b)
(b)
Figure 5. Total antioxidant capacity (a) and vitamin C content (b) in the mixed vegetable cream after HPH treatments. Data are means +/− SD. Statistical analysis was by the one way ANOVA (TAC p < 0.05; vitamin C content n.s.), using Tukey’s as post test. Different letters indicate statistical significance.
MPa treatment (Figure 6(b)).
4. Discussion
New processing technologies are necessary to deliver at the point of food consumption near-absolute chemical and microbial safety and to retain the sensory and nutritional quality of fresh ingredients. Particularly, important disadvantages of conventional thermal processing technologies are chemical reactions leading to off flavours, destruction of nutrients, and other losses of product quality. Among novel processing alternatives, high-pressure treatment has been adopted at the fastest rate, so a better
 (a)
(a)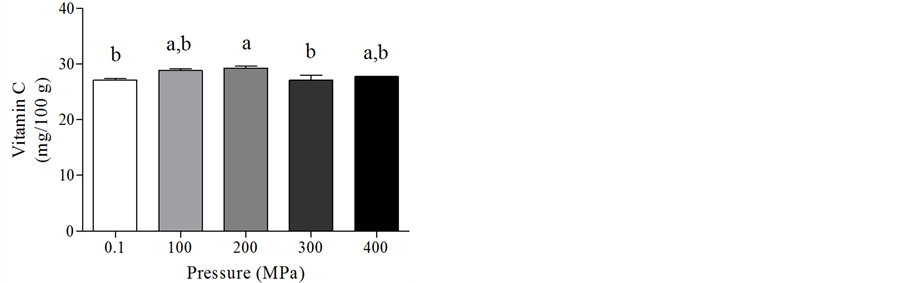 (b)
(b)
Figure 6. Total antioxidant capacity (a) and vitamin C content (b) in the mixed fruit cream after HPH treatments. Data are means +/− SD. Statistical analysis was by the one way ANOVA (TAC n.s.; vitamin C content p < 0.05) using Tukey’s as post tes. Different letters indicate statistical significance.
understanding of the impact of this technology on the nutritional value of food from both animal and plant origin is crucial.
Despite the presence of some studies evaluating the effect of HPH processing on foods of animal origin such as meat, milk and dairy products [16], little is known about eggs and egg creams.
In this study, an EYC was prepared using 60% (w/w) sunflower oil; EYC lipids, reflecting the fatty acid composition of the ingredients, had a high content of linoleic acid that could be easily peroxidized. HPH processing is reported to increase lipid peroxidation almost proportionally to the content of polyunsaturated fatty acids in the sample [17]; notwithstanding, we did not observe any increase in lipid oxidation in the EYC after HPH processing. It is conceivable that lipid oxidation has been avoided through the inactivation of lipoxygenase; according to the literature lipoxygenase, which catalyses the oxygenation of fatty acids into the corresponding lipid hydroperoxides, may be inactivated by high pressure in different food products [18-20]. In addition, antioxidants in the food matrix could have had a role in the prevention of peroxidation. In our study vitamin E was not reduced by HPH, in agreement with the findings of Hayashi et al. in eggs [21], and the preservation of tocopherol content could have contributed to the stability of food lipids.
HPH did not influence either the fatty acid composition or the free acidity of EYC, in agreement with Pereda et al. [22] and Rodriguez et al. [23]. The decreased free acidity observed in EYC containing the highest NaCl concentration could be accounted to changes in lipase activity, already reported in flours having 1% NaCl concentration [24].
Regarding vegetable and fruit, a correlation between food microstructure and in vitro bioaccessibility of antioxidant components as carotenes as been already reported [25].
Accordingly, the TAC increase observed in the MVC could be explained by the breakdown of intact vegetable cell walls after HPH, with consequent release of intracellular antioxidants, particularly polyphenols and vitamins. Similar results have been reported also by Patras et al. [26] and Suarez-Jacob et al. [27], who observed an increased anti-radical activity in strawberry and blackberry purées, and in apple juices after high pressure processing. On the contrary, no modifications in TAC were observed by Fernandez Garcia et al. [28] in an orange-lemon-carrot juice after high pressure processing, and in MFC in the present study.
This discrepancy could be accounted to the food matrix, and to the molecular relationships between nutrient and non-nutrient components of foods. In fact, there is ample evidence that the different physical state of the type of matrices plays a key role in the release, mass transfer, accessibility, and biochemical stability of many food components [29].
A food matrix influence could also explain data on vitamin C content, which was not modified in the MVC as reported by other Authors in different vegetable products [27,30,31], while was increased in the MFC.
In conclusion, the current work evidences that HPH does not cause lipolysis, lipid peroxidation and vitamin E loss, at least on the considered food matrix and in the condition studied. Peroxidation is the major cause of quality deterioration of many food products, impairing both flavor and nutritional value, and an increase in peroxidized lipids may pose health risks since they are implicated in the development of different diseases. Thus, the lack of any effect of HPH on the lipid fraction of foods is particularly important.
In plant food, HPH did not affect the nutriational quality but improved it, suggesting the use of this treatment as valid alternative to thermal pasteurization for obtaining high quality products with preserved nutritional characteristics. The ameliorating effect of HPH on antioxidant and vitamin availability was dependent on the food matrix; a better understanding of the correlation between the processing, the food matrix microstructure, and nutrient bioaccessibility will be fundamental in the production of food products with an increased nutritional value. Although further investigations are needed to better understand the effect of HPH, especially at extreme pressure and temperature combinations, our results open new perspectives for the production of ambient stable products, and contribute to process optimization and food regulation of pressure-sterilized food products.
Acknowledgements
This study was supported by the European Project “Innovative non Thermal Processing Technologies to Improve the Quality and Safety of Ready-to-Eat Meals (HighQ RTE)” (Project No. 023140) by the EU’s 6th Framework Programmés “Food Quality and Safety” activity.
NOTES