1. Introduction
Panthenol (Figure 1) is one of the components of some local cosmetic preparations. The USP [1] stated a non aqueous titration method for its determination. Despite the low molecular absorptivity of panthenol due to the lack of UV chromophores, yet direct spectrophotometric assay has been reported [2]. Several colorimetric analysis have been reported for the determination of panthenol after its hydrolysis [3,4]. As an alcohol, panthenol has been determined by gas liquid chromatographic method [5-7]. Liquid chromatographic methods either directly [8,9] or after derivatization and condensation with fluorescamine [10] was also used for assay of panthenol. Thin layer chromatographic determination of panthenol with spectrodensitometric detection was also mentioned in the literature [11]. The use of H1 NMR for the analysis of panthenol in mixtures has also been published [12]. Fluorimetric methods for panthenol after its hydrolysis to ß alanol and reaction with either ninhydrin or NBD-Cl were also reported [13].
Fluoride, like other halides, is a monovalent ion, which is a major ingredient in many toothpaste preparations. Literature review reveals that fluoride was determined by spectrophotometry [14-18], spectrofluorimetry [19], and voltammetry [20].
In this work, fluoride was determined in many of its commercial toothpastes applying spectro-fluorimetry [19]. Moreover, the present work is trying to present a stability indicating assay of some lotions and creams containing panthenol using a reported method [13] as a guide; which depends on the fact that NBD-Cl is a fluorigenic agent specific for primary amino group, which is a result of the alkaline hydrolysis of panthenol to ß alanol. Since panthenol by itself, does not contain a primary amino group, it is supposed that it will not react with NBD-Cl without hydrolysis, which was used in this work as a basic concept to estimate the stability of panthenol in its local cosmetic preparations.
This research focused on the application of stability studies to determine some cosmetic preparations depending on the high consumption of such products in the local market of our community, and counting on the fact that such products may undergo physical or chemical degradation as a result of inappropriate storage. So we tried in our laboratory to predict the best conditions for highest stability of such products to achieve better storage and hence consumer compliance. We have chosen spectrofluorimetry and spectrophotometry to access this purpose, due to their simplicity and high sensitivity, in addition to their availability in most laboratories which facilitates the use of the proposed method in quality control laboratories.
2. Experimental Procedures
2.1. Apparatus
1) Spectrophotometric analyses were carried out on a Dynamica Halo DB-20 spectrophotometer; UV-Visible double-beam spectrophotometer with matched 1 cm path-length quartz cells. Absorption spectra of the studied drug were recorded on a fast scan speed, setting slit

Figure 1. Structural formula of panthenol.
width to be 1 nm and sampling interval to be auto.
2) Spectrofluorometer: Perkin Elmer LS 45 Luminescence Spectrometer, equipped with a 150 W Xenon arc lamp, grating excitation and emission monochromators, and a recorder. Slit widths for both monochromators were set at 10 nm. A 1 cm quartz cell was used. Derivative spectra were evaluated using Fluorescence Data Manager (FLDM) software, Perkin Elmer Buck i.e. FL WINLAB, version 400.02.
2.2. Materials and Reagents
1) NBD-Cl (Aldrich Chemical Co., Ltd., Dorset, England) was freshly prepared by dissolving 5 mg in 50 ml volumetric flask using ethanol. The solution is made to volume with the same solvent to get a solution of a final concentration of 100 µg/ml.
2) Borate buffer of pH 8 was prepared by mixing 0.2 M boric acid and 0.2 M sodium hydroxide.
3) Ammonium fluoride (Riedel dehaen, Sleeze, Germany), a stock solution of concentration 0.5 mg/ml was prepared by dissolving 0.025 g NH4F in 50 ml volumetric flask using distilled water, and completing to the mark with the same solvent. This solution was further diluted to contain 12.5 - 500 µg/ml of fluoride in a series of 25 ml volumetric flasks.
4) Terbium chloride (Aldrich Chemical Co., Ltd., Dorset, England), working solution of 4 × 10–3 M was prepared by diluting the stock 0.08 M solution.
5) Hexamine (El Nasr Pharmaceutical Company, Egypt), was prepared as 10% w/v and its pH was adjusted to 8.5 with 0.2 M sodium hydroxide.
6) Sodium salicylate (El Nasr Pharmaceutical Company, Egypt), was prepared as 7 × 10–3 M in distilled water.
7) The cosmetic preparations include:
Toothpastes containing fluoride:
a) Signal 2
b) Close up
c) B-fresh
d) Colgate
e) Miswak Cosmetic preparations containing panthenol:
a) Panthenol cream
b) Panthenol lotion
c) Pantho EVA gel
d) Acopantin cream
2.3. Construction of the Calibration Curve for Method Ι
To each 25 ml volumetric flask, add 1 ml of TbCl3 solution (4 × 10–3 M), 1 ml of sodium salicylate solution (7 × 10–3 M), 1 ml of hexamine 10% solution previously adjusted to pH 8.5. To each flask, transfer 1 ml aliquots of fluoride solutions so that the solutions contain 0.5 to 20 µg/ml fluoride. A blank was made by adding 1 ml of sodium salicylate solution (7 × 10–3 M) into 25 ml volumetric flask. The flasks are then filled up with distilled water. The fluorescence intensities of the solutions are measured at 547 nm after excitation at 322 nm. A plot of the difference in fluorescence intensity against the concentration of the fluoride added is thus made to obtain the standard calibration graph (Figure 2). Alternatively, the linear regression equation was derived.
2.4. Construction of the Calibration Curve for Method ΙΙ
One gram of the cream or gel is grind with 1 gm of anhydrous sodium sulphate and dissolved in few milliliters of distilled water, filtered if necessary. The solution is then brought to 50 ml with distilled water. Aliquot volumes of the prepared preparations covering the concentration range of 2 - 20 µg/ml were transferred into a series of 10 ml volumetric flasks, 3 ml of borate buffer pH 8, followed by 2 ml of NBD-Cl were added, then the solutions are placed in a boiling water bath for 15 minutes. The solutions are cold, and made to volume with distilled water. The absorbance readings of the resultant solutions are measured at 480 nm. Base line correction was carried out to delete any absorbance reading of the blank. The measured absorbance values were plotted against the final concentration of the drug (µg/ml) to get the calibration curve (Figure 3). Alternatively, the corresponding regression equation was derived.

Figure 2. Calibration curve for fluoride applying the reference method.

Figure 3. Calibration curve for panthenol applying the reference method.
3. Results and Discussion
Fluoride was quantitatively assayed through its quenching of the fluorescence of a ternary complex of terbiumsalicylate-hexamine. The different experimental parameters affecting the intensity of the ternary complex were carefully studied and incorporated into the procedure. The method was successfully applied for the determination of fluoride ion in toothpastes.
Panthenol stability was estimated through is reaction with NBD-Cl without hydrolysis. Different experimental parameters affecting the formation of the reaction product were carefully studied and optimized. Such factors were changed individually while the others were kept constant.
3.1. Effect of Different Temperature Settings
The reaction was carried out at different temperature settings (room temperature, 60˚C, 70˚C, 80˚C, 90˚C, 100˚C) using a thermostatically controlled water bath. It was found that the absorbance readings increase gradually by increasing the temperature to be maximum by boiling (Figure 4). This could be explained that the higher the temperatures are, the more the drug undergoes degradation and hence starts to react with NBD-Cl giving higher absorbance reading.
3.2. Effect of Reaction Time
The effect of reaction time between panthenol and NBDCl was investigated over the period of (5 - 60 minutes), the absorbance values increase gradually by the increase in the reaction time (Figure 5). These results are expected since longer boiling times expose panthenol to be degraded more than short times, which in turn increases its ability to react with NBD-Cl.
Semilogarithmic plots of panthenol at different temperature settings after the addition with NBD-Cl was studied (Figure 6), The first order degradation rate constant and the half life time at each temperature were calculated according to the following equations: ln a/a-x = Kt

Figure 4. Effect of different temperature settings on the absorbance readings of panthenol.

Figure 5. Effect of time on the absorbance readings of panthenol.

Figure 6. Semi logarithmic plot of panthenol at different temperatures. Where 1) 100˚C; 2) 80˚C; 3) 70˚C; 4) 60˚C; 5) 40˚C.
where a is the initial concentration of the drug, x is the concentration of the resulting degraded solution after time t, and K is the reaction rate constant, while the half life time (t1/2 = 0.693/K).
The results obtained illustrated that panthenol is of high stability since it doesn’t react with the reagent except after is alkaline degradation to produce the primary amino group responsible for the reaction with NBD-Cl. Maximum stability was achieved after 5 minutes at 40˚C.
This method was also extended to study the stability of panthenol at different percentages of humidity at different time intervals (Figure 7). Maximum stability of panthenol was attained at 15% of humidity for 6 months which should be considered in the storage parameters.
3.3. Pharmaceutical Applications
The proposed methods were successfully applied for the determination of fluoride (Table 1) and panthenol (Figure 8) in their different cosmetic preparations.

Figure 7. Semi logarithmic plot of panthenol at different % humidity. Where 1) 95%; 2) 40%; 3) 15% relative humidity.

Figure 8. Calibration curve for panthenol cream.
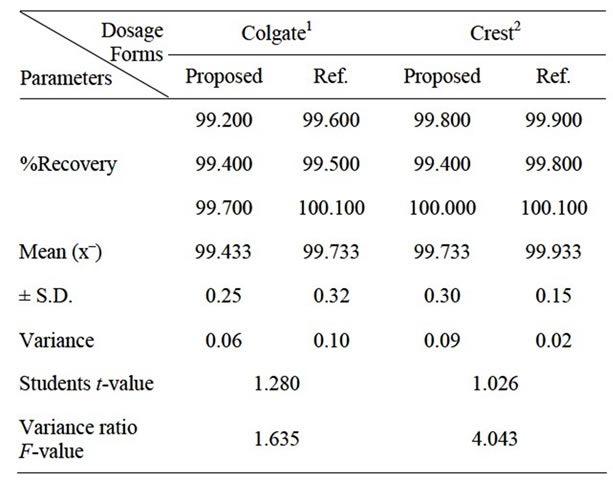
Table 1. Application of the reference method for determination of fluoride in toothpastes.
4. Conclusion
Two simple and sensitive spectrofluorimetric and spectrophotometric methods have been developed for the determination of some cosmetic preparations. Stability studies were conducted to predict the best conditions for storage of the studied compounds. The simplicity of the proposed methods allows their applications in quality control laboratories.
NOTES