1. Introduction
Growing demands for power sources for transient high power density have stimulated great interest in electrochemical capacitors. Among them a chemical battery (supercapacitor) with large specific capacitance is rising. It has much advantage such us high power density, Faraday large electric capacity, high energy, wide range of operating temperatures, extremely long service life and environmental protection [1]. Supercapacitor usually divides into electric double layer capacitors (EDLC) and Faraday quasi-capacitors according to the type of reactions of electrodes [2]. EDLC is a small part of whole capacitance, on the other hand pseudocapacitance is a large part of it. The EDLC occurs in porous active carbon/fiber with very high specific surface area [3], whereas the pseudocapacitance is observed in transition metal oxides [4,5] (RuO2 [4-10], IrO2, MnO2 [10-14], Co3O4 [15-17], NiO [18,19], SnO2 [9] etc.) and conducting polymers [5]. So the electrode material is one of the key factors that decided the electrochemical capacitance [3]. A most successful electrode material is composed with RuO2 due to the minimal energy requirements and oxide stability during proton diffusion into the crystal structure [4]. The highest capacitance of RuO2 was reported of 380 F/g in the case of crystalline RuO2 and 760 F/g in the case of hydrous RuO2 with the bulk form [6]. Although pseudocapacitor has higher capacity than EDLCthere are many disadvantages such as high raw material cost and process difficulty which limits its practical application. In order to overcome these disadvantages, some transition metal oxides which also characterize multivalent metal oxides are attracted to replace part of RuO2. Highly stable RuO2-Ta2O5/Ti electrodes with a specific capacitance of 170  and a lower capacitance (130 F
and a lower capacitance (130 F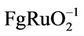 ) also are composed of crystalline RuO2 and amorphous tantalum oxide [6]. So a fact can be concluded that a high capacitance must have an amorphous structure. SnO2 is widely used as an additive in many catalysis fields because it is an inexpensive, environmentally friendly semiconductor material. For example adding SnO2 to titanium anodes coatings can efficiently improve stability and the selectivity of chlorine reaction [9]. The author in literature [19] used two-step hydrothermal synthesis method to prepare Ru-Sn binary oxide electrodes, whose electric capacity reached to 1150 F/g. But only Ru-Sn oxide powder can be prepared first using this method. Carbon black and adhesive must be added to prepare coatings, which can produce side-effect of pseudocapacitance. Therefore, it is necessary to look for one-step method to prepare Ru-Sn oxide electrodes directly. This paper presented a method to prepare RuO2- SnO2 coatings directly on titanium substrate by thermal decomposition at low temperature. The structural, morphological, compositional and supercapacitance properties had been studied.
) also are composed of crystalline RuO2 and amorphous tantalum oxide [6]. So a fact can be concluded that a high capacitance must have an amorphous structure. SnO2 is widely used as an additive in many catalysis fields because it is an inexpensive, environmentally friendly semiconductor material. For example adding SnO2 to titanium anodes coatings can efficiently improve stability and the selectivity of chlorine reaction [9]. The author in literature [19] used two-step hydrothermal synthesis method to prepare Ru-Sn binary oxide electrodes, whose electric capacity reached to 1150 F/g. But only Ru-Sn oxide powder can be prepared first using this method. Carbon black and adhesive must be added to prepare coatings, which can produce side-effect of pseudocapacitance. Therefore, it is necessary to look for one-step method to prepare Ru-Sn oxide electrodes directly. This paper presented a method to prepare RuO2- SnO2 coatings directly on titanium substrate by thermal decomposition at low temperature. The structural, morphological, compositional and supercapacitance properties had been studied.
2. Experimental
Commercial RuCl3 (36.9% Ru3+) and SnCl4 (98% Sn4+) were used as the starting materials and the mole ratio of Ru3+ and Sn4+ was 4 to 6. The metal chlorides in the required proportions were each dissolved in a required amount of anhydrous ethanol (C2H5OH). They were mixed uniformly. The resultant solution was brushed onto the pretreated Ti substrate. The coatings were dried at 260˚C in the oven for one hour.
X-ray diffraction (XRD: Rigaku, D/max Ultima III diffractometer with CuKa radiation) measurements were employed to investigate the crystallinity of the coatings. The scanning electron microscopy (SEM: HITACHI, S-4100) was performed to observe the surface. The transmission electron microscopy (TEM: JEOL-2000EX at 120 kV) was operated to analyze the microstructure. Electrochemical behaviors of the derived electrodes were examined using Cyclic voltammetry (CV) and galvanostatic charge-discharge in a 0.5 M H2SO4 electrolyte solution carried out in an electrochemical workstation (Auto Lab PGST-302A) at room temperature. Large area pure titanium plate was used as the counter electrode and saturated calomel electrode (SCE) as the reference electrode. CV test was performed between –100 and 1000 mV at a scan rate of 25 mV·s–1.
3. Results and Discussion
3.1. XRD Analysis
The XRD measurement was carried out to confirm the poor crystallinity of the as derived Ru0.4Sn0.6O2 coatings. It’s showed that the mixed oxides exhibit very broad and diffuse diffraction peaks. They can be due to poorly crystallized rutile phases. They are attributed not only to the RuO2 and SnO2 rutile-type oxide phase, but also to some new rutile-type oxide phases. These oxides show the strongest peaks with intermediate 2θ values between those of RuO2 and SnO2, suggesting that these oxides must form a solid solution. The reflections in the pattern shift systematically to higher d-spacings than those of RuO2. It’s indicated that tin is replaced by ruthenium according to the smaller ionic radius of the Ru4+ ion (0.76 Å) with respect to Sn4+ (0.83 Å), which is in agreement with the formation of solid solution phases with (Sn, Ru)O2 compositions. No metallic Ru phase was detected but one peak at about 40˚ which was corresponding to the Ti substrate (Figure 1).
3.2. SEM Analysis
SEM images of the coating surface at two magnifications are shown in Figure 2. It can be found that the electrodes consist of mud-cracks structure and flat area. The area of each mud was also in the same size. The widths of the crack are about 0.2 µm. Cracks can provide the space for the reaction of active RuO2 and SnO2. It also provided the channel for the oxygen to result in the passivation of Ti substrate. Therefore, the cracks can deteriorate the electrochemical performance of as-prepared electrode. The flat areas of all the electrodes are composed of numerous nanosize particles which are not well-grown. Most of theses particles are agglomerated to form flat area.
3.3. TEM Analysis
The HRTEM image is shown in Figure 3. The selected area diffraction (SAD) pattern shows that there is no any bright spot under illumination which confirms that formation of amorphous mixed oxide coating. But some small crystalline particles which are homogeneously distributed in the coatings can be seen clearly in Figure 3(a). The sizes of these particles are about 1 - 3 nm. The EDS results show that this kind of particles consists of Ru, Sn, and O element (not shown here). Such type of morphology is different from not only the amorphous structure but also the crystalline structure. The amor phous structure is expected to have a high surface area which is

Figure 1. The XRD pattern of the Ti/Ru0.4Sn0.6O2 electrode.

Figure 2. The SEM images of the Ti/Ru0.4Sn0.6O2 electrode.
 (a)
(a) (b)
(b)
Figure 3. The HRTEM and SAD images of the electrode.
beneficial for supercapacitor application because it can be rearranged when ions such as H+ and OH– are inserted while the lattice of crystalline is rigid and is difficult to expand. But in this paper not the absolute amorphism but the very poorly crystalline is in favor of the capacitance.
3.4. Electrochemical Properties
The charge-discharge potential windows play an important role for supercapacitor performance. Figure 4 shows the CV curves of the electrodes at 25 mv/s scan rate with different potential ranges. It’s seen that different potential ranges have almost similar curve shapes. The cyclic voltammograms are almost rectangular and symmetrical. This indicates that the oxidation and reduction processes at the electrode interface are highly reversible. Every curve exhibited a capacitive behavior. We also can observe the response current of the electrode almost reverses instantaneously when the scan direction changes whether from anodic to cathodic scan or from cathodic to anodic scan. The changes exhibit almost vertical lines which reflect little impedance of the electrodes [5].
The specific capacitance (Csp) of the Ti/Ru0.4Sn0.6O2 electrode is obtained to explore the dependence of the electrodes’specific capacitance on the potential ranges. Table 1 shows the values of specific capacitance. The Csp of the Ti/Ru0.4Sn0.6O2 electrode obtained from Q/(2ΔV·W), where Q is the CV charge, ΔV is the potential range of CV and W is the weight of ruthenium oxides. The Csp of the electrodes increases with the broadened potential window. The increase in capacitance at broader windows is attributed to the presence of inner active sites, which can involve in the redox transitions completely, probably due to the diffusion effect of proton within the electrode [10]. The increasing trend of the capacitance suggests that the parts of the surface of electrode are accessible at high charging-discharging rate. Hence, the specific capacitance obtained at the broadest window is believed to be close to that of full utilization of the electrode material.
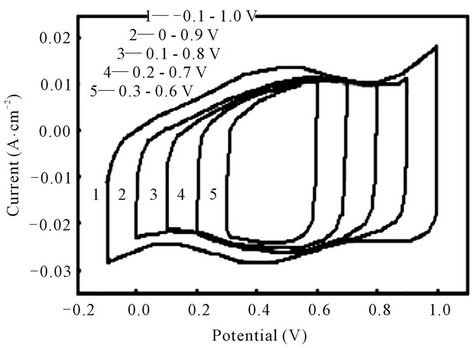
Figure 4. Cyclic voltammogram of the Ti/Ru0.4Sn0.6O2 electrode at different potential windows.

Table 1. The specific capacitance of the Ti/Ru0.4Sn0.6O2 electrode at different potential ranges.
Compared with the CV experiment, the galvanostatic charge-discharge experiment can further reflect the supercapacitance characteristics. Figure 5 shows the galvanostatic charge-discharge curve of Ti/Ru0.4Sn0.6O2 electrodes at different current densities. The curves show linear change with the time changing and exhibit good capacitance characteristics. The charge curve and the discharge curve are symmetrical, which show a better reversibility. With current density increasing, the discharge time of the electrodes shortens, and the corresponding specific capacitance also decreases gradually. The reason is the charge-discharge process is related with not only the moving speed of ions in the electrolyte, but also the adsorption process on the electrode surface. In a smaller current density, ion moving speed and adsorption process is developed completely. So the electrode reaction not only occurs in the electrode surface, but occurs in body phases. The active sites in the electrode are fully utilized, which lead to the charge-discharge process completely. Similar results have been reported [12].
Figure 6 shows the relationship between cycle number and specific capacitance of the Ti/Ru0.4Sn0.6O2 electrode in 0.5 mol/L H2SO4 at the scan rate of 25 mV/s and the potential window of –0.1 - 1.0 V. The specific capacitance decreases gradually with increasing cycle number. The specific capacitance declines about 18.8% after 1000 scan cycles. This suggests that the electrode has a good stability and is useful for supercapacitor application.