1. Introduction
Progress of world citrus production is being thwarted by some limiting factors
among which Xanthomonas axonopodis pv. citri (Xac) is the most prominent hindrance biotic factor that causes citrus canker disease [1] [2] . Till now, successful management of this disease is not feasible since only few chemicals such as copper containing chemicals were proved as partially effective against the disease [3] [4] . However, in one way, application of these chemicals increases production cost and in another way, it leads to the environmental pollution and induces pathogen resistance [4] [5] . Hence, it is requisite to find an alternative eco-friendly management approach. Induced resistance (IR) is a physiological state of enhanced defensive capacity elicited by specific phytohormone, whereby the plant’s innate defenses are potentiated against subsequent biotic challenges [6] . Phytohormones are natural or synthetic compounds that activate chemical defense in plant [7] . Induce resistance is provoked by phytohormones such as salicylic acid (SA), jasmonic acid (JA), 3-indoleacetonitrile (IAN), nicotinic acid (NA), folic acid (FA) [8] [9] [10] [11] . Salicylic acid plays an important role in induction of plant defense against a variety of biotic and abiotic stresses through morphological, physiological and biochemical mechanisms [12] [13] . It is involved in endogenous signaling, mediating in plant defense against pathogens [14] . Jasmonic acid is a class of lipidic plant hormones, not only involved in plant-microbe’s interactions in defense and symbiosis [15] but also induced secondary plant metabolites, like alkaloids, terpenoids, flavonoids, coumarins and so forth [16] . Besides, IAN prevented bacterial growth by inhibiting biofilm formation [10] . In addition, FA and NA are involved in metabolic functions such as biosynthesis of nucleic acids, amino acids and pantothenate, lignin formation and synthesis of pathogenesis-related (PR) proteins and regulated redox process [17] . So far, some studies have provided the evidence that phytohormones like SA, JA, IAN, NA and FA have the potentiality for mitigating plant disease by modulating redox balance activating enzymes [18] [19] . Above information reveals that phytohormones like SA, JA, IAN, FA and NA create a favorable condition for plant to resist pathogen by modulating physiological and molecular process. In this study, we aimed to understand how exogenous application of phytohormones mitigates citrus canker disease.
2. Materials and Methods
2.1. Isolation and Identification of Pathogen
The causal organism of citrus canker was isolated from infected leaf collected from Citrus Research Institute, Bangladesh using dilution plate techniques described by Goszczynska and Serfontein [20] . Colonies of bacteria were purified on NA and SX media plates. The causal organism of citrus canker was identified by following gram’s staining reaction [21] , potassium hydroxide (3% KOH) test [22] , oxidase test [23] , starch hydrolysis test, catalase test, milk proteolysis test, esculin hydrolysis test, citrate utilization test, Gelatin liquefaction test, Tween 80 lypolysis test [24] and Pathogenicity Test [25] .
2.2. In Vitro Measurement of Antibacterial Properties of the Phytohormone against Xac
In-vitro ability of the phytohormones such as SA (0.3 mM), JA (3 mM), IAN (0.6 mM), NA (5 mM) and FA (0.5 mM) to inhibit the growth of Xac was evaluated using well diffusion method measuring the inhibition zone [26] . 100 μL of the bacterial suspension (108 cfu/mL) was poured on NA plate and spread uniformly [27] . Each well was filled with 20 μL of each elicitor solution and 20 μL distilled water was used as control. The plates were incubated at 30˚C and the inhibition zone was measured at 24 hours, 48 hours and 72 hours after inhibition.
2.3. Cultivation of Planting Material
One year old lemon (Citrus aurantifolia) seedlings were collected from Krishibid nursery Agargaon, Dhaka. The seedlings were vigor, healthy, disease and insect free. Collected lemon seedlings were potted (one plant/pot) in twenty-inch earthen pot and kept in shade for two days. Sterile loamy soil and sand (2:1) were used as potting media. Potted seedlings were irrigated everyday early in the morning with distilled water. No additional nutrient was supplied during the study period.
2.4. Preparation and Application of Phytohormone Solution
0.41 g SA, 0.06 g JA, 0.09 g IAN, 0.61 g NA and 0.22 g FA (MERCK, Germany) weighted and dissolved in one liter sterile distilled water to get the concentration of 3 mM, 0.3 mM, 0.6 mM, 5 mM and 0.5 mM respectively. Prepared elicitor solution was sprayed equally over the three plants. Each plant was sprayed with 330 ml solution approximately. Plants were sprayed on weekly basis and thus the treatments were maintained for seven weeks. The plants got total seven sprays. The above concentration was selected based on preliminary lab test where concentration above 3 mM and 0.3 mM for SA and JA respectively gave same result but caused leaf defoliation. For IAN, NA and FA concentration above 0.6 mM, 5 mM and 0.5 mM gave the similar result as they did.
2.5. Inoculums Preparation and Inoculation for Pathogenicity Test
Bacteria for inoculation were cultured in Nutrient broth (NB) and maintained in 30˚C temperature for 24 hours. Thereafter it was resuspended in sterile distilled water to a concentration of approximately 108 cfu/ml (OD: 0.5650nm). Prepared inoculums were injected in to the lower surface of the healthy leaf with syringe both for pathogenicity. Five leaves were inoculated from each plant and each leaf was inoculated with 0.5 ml bacterial solution.
2.6. Inoculation of Phytohormone Treated Plants with Xac
Inoculums were prepared and inoculated followed by the above procedure. The plants were inoculated with bacteria after seven days of first spray. Data on lesion diameter, disease severity, PAL, TPC and Potassium content were recorded at 15, 30 and 45 days after inoculation. The disease severity was recorded by using the following scale developed by Rai and Mamatha [28] . The 0 - 5 grade disease severity scales was used for recording disease severity data. The grades were, 0 = 0% infection, 1 = 1% infection, 2 = 1% - 10% infection, 3 = 10% - 20% infection, 4 = 20% - 40% infection and 5 = 40% - 100% infection. The following formula was used to determine disease severity.
Percent Disease Index 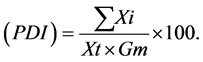 Here,
Here, 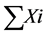 = sum of individual disease rating,
= sum of individual disease rating, 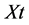 = total number of leaf examined and Gm = maximum grade.
= total number of leaf examined and Gm = maximum grade.
2.7. Measurement of PAL Activity
For extraction of enzymes 0.2 g leaf sample was homogenized (Crl-6010, Kriens, Switzerland) at 4˚C in sodium borate buffer (2 ml of 0.2 mM at pH 8.8) with 1% (w/v) polyvinyl-polypyrrolidone (PVPP) and then 5 mM β-mercaptoethanol was added. The mixture was centrifuged at 15,000 rpm for 30 min and the supernatant was used as crude extract. PAL activity was measured spectrophotometrically according to Wang and Liu [9] and was expressed as unit g−1 of fresh weight. One unit was defined as an increase of the absorbance by 0.01 per minute.
2.8. Extraction and Estimation of Total Phenolic Compounds from Leaves
Phenolic compound was extracted by the following method of Biswas et al., [29] . Initially, 1 g fully dried ground leaf was added with 80% ethanol (1:10 w/v). Secondly, it was centrifuged at 10,000 rpm for 30 minutes at room temperature. Supernatant was separated and saved. This process was repeated for 5 times. Then it was evaporated and 5 ml of distilled water was added. Distilled water in another test tube was used as blank. Afterward, 0.5 ml of FCR was added and after three minutes, 2 ml of 20% Na2CO3 solution was added in each tube. Then the tubes were heated in boiling water for 1 min and cooled at room temperature. Using Ultra Violet Visible (UV-VIS) spectrophotometer the absorbance at 650 nm against blank (water) was measured and the standard curve was prepared using different concentration of catechol. From the standard curve, the phenols concentration was determined in the test sample and expressed as mg∙g−1 of fresh sample materials.
2.9. Extraction and Estimation of Potassium from Leaf
Potassium was extracted by Di-acid mixer (HClO4:HNO3 at 2:1 ratio) method [30] . 20 ml Di-acid mixture was added with one gram oven dried leaves sample. It was then heated at 200˚C until white fume evolved and then cooled. This solution was filtered with whatman filter paper in 100 ml volumetric flask and the volume was made up to the mark with distilled water. Concentration of potassium in the test sample was determined by flame emission spectrophotometer from the standard curve prepared by potassium chloride and expressed as mg∙g−1 of fresh sample materials.
2.10. Statistical Analysis
The experiment was carried out in a complete randomized design (CRD) with three replications. There were 6 treatments combinations such as Control (water), Salicylic acid (3 mM), Jasmonic acid (0.3 mM), 3-indoleacetonytrile (0.6 mM), Nicotinic acid (5 mM) and Folic acid (0.5 mM). Data were subjected to analysis of variance (ANOVA) and significant differences of the means among treatments were analyzed using STATISTIX 10 software (Analytical software, USA). Standard deviation and correlation was calculated using Microsoft excel.
3. Results
3.1. Isolation and Identification of Pathogen
On the basis of morphological, biochemical characters the isolated bacteria was identified as Xanthomonas axonopodis pv. citri. The bacteria yielded yellow, convex, mucoid growth on NA plates and light yellow to slightly blue, small, flattened, growth on SX agar plate after 48 hours of incubation at 30˚C ± 1˚C. The bacterium was rod shaped with rounded ends, cells appeared singly and also in pairs, gram negative (red color) and capsulated under the compound microscope at 100 times magnification with oil immersion. In KOH solution, it produced a mucoid thread when lifted with the loop, showed catalase activity; bubbles were formed after adding 3% H2O2, formed dark purple color on oxidase disk, showed β-glycosidase activity in esculin hydrolysis test. Besides, esterase enzyme was produced in tween 80 lypolysis test, gelatin was liquefied, milk was proteolyzed, citrate was utilized, starch was hydrolyzed and the findings are summarized in Table 1. Besides, in pathogenicity test it gave positive result.
3.2. In Vitro Evaluation of Antibacterial Properties of the Phytohormone against Xac
In-vitro analysis showed that no one chemical was able to produce inhibition
![]()
Table 1. Biochemical characteristics of pathogenic bacteria of citrus canker.
zone on NA plate against Xac. That means the chemicals did not possess any antibacterial properties against the bacteria.
3.3. Citrus Canker Disease Development in Xac Inoculated Plant Treated with or without Phytohormone
Canker development was significantly attenuated by the applied phytohormone compared to control particularly by the SA, JA and IAN. Variation in symptom expression was recorded in phytohormone applied plants when challenged with Xac. SA took 19 days to express symptom after inoculation while JA, IAN both took 16 days, whereas NA, FA and control took 14 days (Figure 1(a)). Besides, at 15 DAI, the treatments varied greatly in disease severity (DS), 21.87% for control and 0% for SA, JA and IAN respectively while FA and NA had a DS of 8.50% and 13.80% (Table 2). DS of both phytohormone and water treated plants increased by degrees prior to the end of experiment but SA treated plants had the lowest DS (7.43% and 15.87%) compare to control (37.33% and 54.20%) at 30 DAI and 45 DAI (Figure 1(b) & Figure 1(c)). Moderate DS (12.80% and 23.80%) were recorded at JA treated plants along with IAN (13.07% and 24.50%) treated plants at 30 DAI and 45 DAI. In addition, FA and NA had lower DS compare to control but higher DS than other elicitor treatments. However, SA showed highest average % reduction of disease over control (79.4%) while JA
![]()
Figure 1. Latent period (a) at different treatment in Xac inoculated plants at 15, 30 and 45 DAI and Symptoms of canker disease at 45 DAI, Control (b) and salicylic acid (c). Different lowercase letters above the bar indicate significant at P < 0.01.
![]()
Table 2. Effect of some selected phytohormone on disease severity of citrus canker.
**Significant at P = 0.01. Different lowercase letters beside the mean value indicate significant at P < 0.01.
![]()
Table 3. Effect of some selected phytohormone on lesion diameter of citrus canker disease.
**Significant at P = 0.01. Different lowercase letters beside the mean value indicate significant at P < 0.01.
and IAN had moderate (67.7% and 66.9%) and FA and NA had lowest (43.3% and 28.8%) average % reduction of disease over control (Table 2). Furthermore, lesion diameter (LD) varied significantly among SA (3.56 mm2), JA (6.90 mm2), IAN (7 mm2), FA (8.13 mm2), NA (9.06 mm2) and Control (9.40 mm2) (Table 3). From the above result, it can be recapped that the applied phytohormone mitigated canker disease development.
3.4. PAL Activity of Inoculated Leaf with or without Phytohormone Treatment
PAL activity at 15 DAI, 30 DAI and 45 DAI were investigated since it is an important enzyme involved in plant resistance. PAL activity was higher in phytohormone treated plants than control at 15 DAI, 30 DAI and 45 DAI respectively (Figure 2(a)). SA treated plants had the highest PAL activity while JA, IAN, FA and NA had moderate PAL activity wherein JA and IAN were close to each other.
3.5. Amount of Total Phenolic Compounds on Leaves at Different Treatments
Total phenolic compounds were investigated in order to scrutinize its impact on canker disease. The accumulation of TPC was notably significant in phytohormone applied plants than control at 15 DAI, 30 DAI and 45 DAI respectively (Figure 2(b)). Among the treatments, SA treated plants had threefold highest TPC content (12.23 mg∙g−1) at 45 DAI compared with control (4.03 mg∙g−1). Besides, JA, IAN, FA and NA treated plants had a moderate and statistically significant TPC content (10.47 mg∙g−1, 8.7 mg∙g−1 and 7.43 mg∙g−1 and 6.56 mg∙g−1). Similar results pattern was found at 15 DAI and 30 DAI. TPC of elicitor treated plants showed continuous increase from 15 DAI to 45 DAI whereas, Control
![]()
Figure 2. PAL activity (a), Total Phenol content (b), Potassium content (c) at different treatments in Xac inoculated plants at 15, 30 and 45 DAI. Different lowercase letters above the bar indicate significant at P < 0.01.
treated plants remained stable. Hence, the results indicating that application of phytohormone augmented TPC in plant leaf that could influence plant resistance.
3.6. Amount of Potassium Contents on Leaves at Different Treatments
Potassium contents were examined to perceive whether applied phytohormone could influence it or not. Potassium accumulation was notably significant in phytohormone applied plants than control at 15 DAI, 30 DAI and 45 DAI respectively (Figure 2(c)). Among the treatments, SA treated plants had the highest potassium content (11.33 mg∙g−1) at 45 DAI compared with control (7.16 mg∙g−1). However, JA, IAN, FA and NA treated plants had a moderate potassium content (10.33 mg∙g−1, 10.47 mg∙g−1 and 9.12 mg∙g−1 and 8.96 mg∙g−1) wherein JA and IAN were close to each other. Similar results pattern was found at 15 DAI and 30 DAI. Potassium content of elicitor treated plants showed continuous increase from 15 DAI to 45 DAI but, Control treated plants remained stable.
3.7. Determination of Correlation Coefficient and Regression Equation
The plants treated with different phytohormones showed attenuated disease severity with increased TPC, potassium content and PAL activity. A negative correlation found between disease severity and TPC (Figure 3(a)). Similarly, the correlation between disease severity and potassium content were also positive (Figure 3(b)). Besides, Disease severity and PAL activity showed negative correlation too (Figure 3(c)). The regression equation also demonstrated the negative correlation among them.
4. Discussion
Plant phytohormone play multidimensional physiological role in biological process of both plant and pathogen by activating of versatile biosynthetic pathway. Xac is signified as a perilous pathogenic stress for citrus so far. Till now, the competence of phytohormone to assuage of plant disease has been successfully confirmed [13] [31] . In our study, initially we isolated the causal organism from infected citrus leaf and identified it as Xanthomonas axonopodis pv. Citri on the basis of morphological and biochemical evidence. Then we found that phytohormones viz. SA, JA, IAN, FA and NA treatment for seven weeks apparently mitigated canker disease development which was undoubtedly manifested by the lower disease severity and lesion diameter. This data agrees with previous investigations [9] [32] . Meanwhile, onset of disease symptom was varied between phytohormone treated and control plants. Canker symptoms were onset earlier in control, FA and NA treated plants and later on SA, JA and IAN treated plants which provide evidence that SA, JA and IAN slowed the infection process of pathogen and led to a belated occurrence of canker disease whereas, FA and NA were in vain. Afterward, in our experiment we found higher PAL activity in
![]()
Figure 3. Correlation between (a) Disease severity and Total phenolic content, (b) Disease severity and Potassium content (c), Disease severity and PAL activity at 45 days after inoculation. The data is significant at P < 0.01.
phytohormone treated plants than control particularly in SA, JA and IAN treatment, in agreements with former study [12] [33] . It has been proved that isoflavones, secondary phenolic compound and lignin are synthesized from PAL [34] . This isoflavones play a vital role in plant microbe interaction and restriction of pathogen multiplication [35] . Again, lignin serves as a matrix on the plant cell wall which provides extra rigidity against pathogen [36] . Interestingly, we found that phytohormone treated plants had higher total phenolic compound than control especially in SA, JA and IAN treated plants which may be influenced by PAL yet other feasibility could exist. It is known that augmentation of TPC alleviated DS [37] [38] . During pathogen attack, inactive bound phenolic in cell turned into active antibiotics by plant hydrolysing enzymes. Besides, phenolic compounds produce reactive oxygen substances such as super oxide anion, hydrogen peroxide which deter pathogenic growth [38] . Lastly, we found that phytohormone application led to an increase of potassium content relative to control notably SA, JA and IAN that may related to attenuation of DS. At this point we could not explicate the explicit explanation for this happening. One reason may be the phenolic compounds that trigger an increase in extra cellular pH and K+ [39] although other possibility was not figure out. Again, Higher K+ concentrations decreased the internal competition of pathogens for nutrient resources that enables plants to allocate more resources to developing stronger cell walls for preventing pathogen infection [40] .
5. Conclusion
In summary, we found that the applied phytohormones viz. SA, JA, IAN, FA and NA mitigated canker severity wherein the former three were more effective against citrus canker and the latter two were less effective. The observed result may be linked with the activation of key enzyme, phenolic compounds and augmentation of potassium content in leaf that created impediments against Xac. The above findings focus on the role of phytohormone especially SA, JA and IAN in mitigating the citrus canker disease, which provide information for development of the alternative diseases management approach.
Acknowledgements
Ministry of Science and Technology, Government of the People’s Republic of Bangladesh is acknowledged for financial support by providing National Science and Information and Communication Technology (NSICT) fellowship, 2014-15. The authors are grateful to Professor Nazneen Sultana for the statistical analysis.