Do Plants Have a Choice of Traits to Be Modulated? Evidence from an Invasive Plant Mikania micrantha Kunth in Different Urban Environments ()
1. Introduction
Mikania micrantha Kunth, commonly known as “mile-a-minute” weed, is considered to be one of the top 10 worst weeds of the world [1] . This herbaceous creeper with a pantropical distribution has now spread to Oceania and most of the Asian countries [2] . Its detrimental presence in India has been reported mainly from forests and plantations of southwest and northeast India [3] [4] . Since the past few years, luxuriant growth of M. micrantha has also been noted along roadsides, abandoned plots, managed plantations, and on the margins of water bodies in the metropolitan city of Kolkata, although these were mostly restricted to small ground cover patches [5] . Urban areas often act as immigration gateways for invasive species to enter through transportation networks [6] where they initially reside in small isolated patches leading to their lower visibility and lesser chance of detection. These small patches then become propagule sources for spread to natural areas [7] [8] thereby increasing their chance of successful establishment in a novel environment [9] . After establishment, invasive plants can alter the soil profile through their direct and indirect effects [10] . Native species often fail to cope up with the altered soil conditions in disturbed urban environments, thereby paving the way for successful colonization of non-native species.
Studies on phenotypic plasticity, or the ability of the same genotype to express different phenotypes in response to different environmental conditions [11] , provide key ecological understanding of the underlying mechanisms of an invasive plant’s success in a novel environment [12] [13] . Modulation of phenotypic plastic traits provide invasive species a competitive advantage over the native species in the community by overcoming a series of biotic and abiotic barriers for successful establishment, broadening its habitat niche in the invaded range [14] [15] , and enabling it to grow and reproduce in a wide range of environmental conditions [14] [16] [17] [18] . Being plastic may allow a population of invaders (even one as small as 10 - 20 individuals) to quickly adapt to the new environment and to maintain higher fitness relative to the native taxa, thereby establishing dominance in the community [14] .
Leaf traits are widely studied since they are the key functional traits that are linked to plant responses to environmental variability [19] [20] [21] and stress tolerance [22] . The majority of these studies focus on differences between species, using species average trait values (e.g. [23] [24] [25] ). However, plant functional traits can vary substantially within species [26] [27] and therefore taking intraspecific trait variation into account may improve the accuracy and resolution of such studies [28] . The importance of reproductive traits such as the germination behaviour of a species has recently been highlighted to be a critical factor for a species to become invasive in novel environments [29] [30] . A short-term germination advantage provides invasive species with a strong competitive advantage over native species [31] which can strongly affect the fitness, growth and survival of a species in a community [30] [32] .
There are a few studies that have explored trait modulation strategies for individual plants growing in an urban environment in the Indian sub-continent (e.g., Ricinus communis [33] , Calotropis procera [34] ). Precise locally collected data available from different regions and environments [35] , especially for the most serious invaders, would aid in obtaining meaningful results. Most world-wide trait based experimental studies on M. micrantha have primarily focused on its vegetative traits. The species has been reported to have higher photosynthetic activity, greater water and photosynthetic nitrogen use efficiency [36] , low leaf construction cost [37] , and high regeneration capacity from clonal fragments [38] to explain its competitive advantage in the community by using comparisons of congeners. However, much less is known about the plasticity of the sexual reproductive traits of M. micrantha as well as its adaptive potential in a heterogeneous urban environment. Along with its year-long vegetative growth, M. micrantha also reproduces sexually during the winter season [39] . In spite of producing self-incompatible flowers, M. micrantha has been found to be an extremely successful colonizer [40] , and appears to be an exception to the Baker’s law [41] . The plasticity in mating systems has been found to increase the long-distance colonizing ability of an invasive species, and assuring reproduction at low population densities [42] which could be crucial for invaders to colonize urban areas. In view of increasing urbanization, which is now considered a major driving force of plant biodiversity loss and community homogenization [43] , integrated studies involving both vegetative and reproductive traits that affect the ability of a population to tolerate environmental heterogeneity [44] , are necessary for understanding the plasticity patterns in M. micrantha.
This study was, therefore, formulated to evaluate the importance of phenotypic plasticity in vegetative and reproductive traits involved in M. micrantha’s tolerance to different habitat conditions in an urban environment. A basic understanding of the soil characteristics on growth of M. micrantha in different disturbance prone areas could also help undermine the difference in trait plasticity responsible for their fast adaptability in urban areas. The hypothesis to be tested was that the survival ability of M. micrantha is a function of plasticity of its fitness-related traits (in terms of both growth and reproduction) to heterogeneous habitat conditions induced by anthropogenic disturbances prevalent within an urban locality.
2. Materials and Methods
2.1. Site Description and Sampling Design
The Greater Kolkata region (88˚22'15.66''E, 22˚39'20.48''N), with a wide mosaic of contrasting habitats, was chosen as the study area (Figure 1(a)). The area was divided into four regions (north, south, east, west) within which three habitats from each region were selected with profuse M. micrantha growth, considered to be representative of the disturbance factors prevalent in an urban locality. The three habitat types were roadsides (Figure 1(b)) with vehicular disturbance, natural areas (Figure 1(c)) with maximum anthropogenic disturbance, and abandoned plots (Figure 1(d)) with minimal disturbance. Some of the factors which influence disturbance regimes in roadsides, natural area and abandoned plots have been listed in Table 1.
The study was conducted during the winter season (January to February, 2015) with temperatures ranging from 13˚C - 16˚C with no or negligible precipitation. In this season, M. micrantha reproduces sexually by forming seeds along with its year-long vegetative growth. This season ensured availability of both vegetative as well as sexual reproductive traits, presumably involved in local adaptation and closely related to fitness. In each habitat type, a quadrat based sampling method [45] was chosen for recording and collecting plant and soil samples from three representative sites. The above ground biomass and individual samples for plant traits were collected from two different quadrats, each of size 0.5 × 0.5 sq. m. (Figure 1(b1), Figure 1(c1), Figure 1(d1)). A smaller quadrat of size 0.25 × 0.25 sq. m was used to sample and estimate the seed related traits (Figure 1(b2), Figure 1(c2), Figure 1(d2)). After harvesting the above ground biomass, soil samples were collected from within the same quadrat (Figure 1(b3), Figure 1(c3), Figure 1(d3)).
At each site, prior to plant sample collection, the community composition within the quadrat (0.5 × 0.5 sq. m) was also recorded. The associated plants occurring with M. micrantha were identified and recorded while some of the unknown species were sampled and identified later from the Botanical Survey of India (BSI), Shibpur, Howrah.
2.2. Estimation of Plant above Ground Biomass and Traits
Ten randomly selected 8-noded individual plants were sampled from the first quadrat of size 0.5 × 0.5 sq. m for measurement of leaf related traits and ten other plants were additionally harvested for measurement of proline content. Since it is otherwise not possible to select plants of uniform age due to its vegetative mode of reproduction, the 8th nodal leaf from the tip of each plant was selected to overcome any age-related variation. The average height of the plant population was estimated from 10 random observations recorded within the second quadrat (0.5 × 0.5 sq. m) following which the total above ground biomass (a measure chosen for quantifying the productivity of the sites) was harvested.
All individual plants and biomass samples were brought to the laboratory in polythene bags. Proline was estimated from fresh plant samples following Bates et al. [46] , and expressed on a fresh weight basis. The leaf related trait measurements along with laminar nitrogen, phosphorus, and potassium content were estimated following standard protocols (Table 2), and expressed on a leaf area basis. The vegetative and reproductive biomass was measured separately and expressed on a dry weight basis along with the nitrogen content of the plant parts.
![]()
Figure 1. Site characteristics of the three habitats with M. micrantha growth. (a) Location of the three habitats in the four regions of the Greater Kolkata area; (b) M. micrantha patch along roadsides, (b1) 0.5 × 0.5 sq. m. sampling of above ground biomass, note the dust deposition on the leaves, (b2) seed sampling from within a 0.25 × 0.25 sq. m. quadrat, (b3) soil sampling; (c) M. micrantha patch in a natural habitat, (c1) above ground biomass sampling from within a 0.5 × 0.5 sq. m. quadrat, (c2) seed sampling, (c3) soil sampling; (d) M. micrantha patch in an abandoned plot, (d1) 0.5 × 0.5 sq. m. sampling of the above ground biomass, (d2) seed sampling, (d3) soil sampling.
![]()
Table 1. Disturbance characteristics prevalent in the three habitats within the study area.
![]()
Table 2. Details of vegetative and reproductive traits of M. micrantha studied in the three urban habitat types.
2.3. Estimation of Seed Traits
Mature capitulums were harvested from within a 0.25 × 0.25 sq. m. quadrat (Figure 1(b2), Figure 1(c2), Figure 1(d2)). The seeds were removed from the mature capitulum and 100 seed weight estimated for each site. For germination studies, 50 seeds per site, with 3 replicates each, were set in petri dishes and maintained in a controlled environment of a Growth Chamber (Temperature-27˚C, Humidity-75%, 16 h light 8 h dark). The seeds were considered as germinated with the onset of 2 leaves. The germinated seeds were counted daily and removed from the Petri dish to avoid the repeat counting error. The experiment lasted for one month, and the following germination parameters were estimated following standard methodologies [47] :
Final Germination percentage (FGP) =
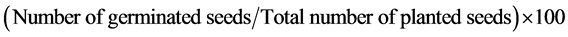
Mean germination time (MGT) 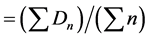
where, n = number of seeds germinated on day D, and
D = number of days counted from the beginning of the germination.
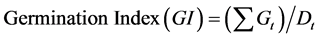
where, Gt is the number of germinants at day t and
Dt is the number of days that have elapsed.
Median germination time (T50) =
Time taken for 50% of the seeds to germinate.
All the above parameters involve only those seeds that were able to germinate in the allotted time essentially regarded as Restricted MGTs and T50s [52] . These severely underestimate the true MGT and T50 of the seed lot because they do not adjust for the larger germination times which remain unobserved due to termination of the experiment. Thus, it is also necessary to consider viable seeds that have not germinated/emerged at the end of an experiment (also known as censored observations). These censored observations were also taken into account to estimate the germination survival functions [53] .
2.4. Soil Sampling and Estimation of Soil Parameters
After harvesting the above ground plant biomass, soil samples from each site were collected from a depth of 0 - 10 cm from a 0.5 × 0.5 sq. m. quadrat by using soil-grab-sampling method (Figure 1(b3), Figure 1(c3), Figure 1(d3)). The soil samples were divided into two fractions, put in polythene bags and brought to the laboratory. One fraction of soil was air-dried and sieved using a 2 mm sieve to remove plant materials, soil micro fauna, debris and stones. The air-dried samples were analyzed for available nitrogen (N) following the Kjeldahl method [54] , available phosphorus (P) following colorimetric molybdenum-blue-method [55] , available potassium (K) following neutral ammonium acetate extract method determined by flame photometer [56] and organic carbon (OC) using K2Cr2O7-H2SO4 oxidation method following Nelson and Sommers [57] . The concentrations of heavy metals like copper, zinc, iron and manganese, all of which are considered to be essential micronutrients for plants, were also estimated following standard methods of Lindsay and Norwell [58] . Major sources of these metals in urban soils include vehicular emission and industrial waste materials [59] .
The remaining fraction of the fresh soil sample (3 replications for each site) was analysed to estimate microbial biomass carbon (Cmic), a biological and eco-physiological indicator of soil quality for sustaining plant growth [60] , using the chloroform fumigation-extraction method [61] . In this method, each sample was divided into two sub-samples (10 g dry weight equivalent) of which one was fumigated with ethanol free chloroform for 24 h in a vacuum desiccator, while the other was treated as a control. Both the fumigated as well as the control samples were extracted with 0.5 M K2SO4 for 30 minutes (300 rpm) in an oscillator [62] and the resulting extracts filtered. Cmic in filtrates was determined by potassium dichromate method and computed using the difference between fumigated and control samples with a conversion factor of 0.38 for Cmic [61] . Cmic values were finally expressed as soil microbial quotient Cmic/Corg (the ratio of the microbial biomass C to organic C) following Li et al. [63] .
2.5. Statistical Analysis
All data were checked for their compliance with assumptions of normality and non-normal data subjected to various transformations based on their frequency distribution. For normally distributed data, comparison of plant (vegetative and reproductive) and soil traits among different habitats was done using one-way ANOVA followed by post-hoc Tukey’s HSD test, whereas non-parametric alternatives of between groups’ comparison tests, namely Kruskal-Wallis H and post-hoc Mann-Whitney U test, were applied to the data that was not normally distributed. For comparing plant and soil parameters between the three habitats, there were 12 (3 sites × 4 regions) observations for each habitat. For all cases, the significance level was set at 5% level of probability.
To estimate germination survival functions, the ‘germination time’ was used to calculate the germination probability, i.e. the probability that an individual seed germinates after a specific time‘t’, considering the initial event at t = 0. The germination probability was estimated non-parametrically by using the Kaplan-Meier method for every seed lot (i.e. seeds sampled from the population of roadsides, natural areas, and abandoned plots) including the censored observations. The null hypothesis i.e. no difference in the overall germination probability between groups (i.e. different habitats) was tested using the Log-rank (Mantel-Cox) test [64] [65] which calculates a Chi-square (χ2) statistic that is compared to a χ2-distribution with two degrees of freedom. The survival plot displays the time course of germination probabilities (or the germination functions) which takes the form of a non-increasing step function, starting from 1 at time 0 and approaching 0 as time increases [53] .
All the statistical analyses were carried out using SPSS version 18.0 and R version 3.2 while data and outputs were visualized by SigmaPlot Version 12.0.
3. Results
3.1. Vegetative Traits of M. micrantha
There was no significant difference in the productivity of M. micrantha populations, as is evident from the total above ground biomass values for the three habitats in Table 3. However, the roadside population produced significantly higher amount of reproductive biomass (mean biomass = 9.9 gm ± 2.6) in comparison to natural areas (1.2 gm ± 0.5; p = 0.002) and abandoned plots (1.5 gm ± 0.5; p = 0.008).
The vegetative and reproductive trait values of M. micrantha in the three habitat types are also presented in Table 3. The roadside plants were found to produce significantly higher LA (29.37.3 mm2 ± 524.6; p = 0.047) and have higher LDMC values (0.18 mg∙g−1 ± 0.01; p = 0.008) in comparison to the plants of natural areas. The proline content was found to be significantly high in plants of natural areas (19.2 n∙mol∙g−1 ± 4.4; p = 0.001) and roadside populations
![]()
Table 3. Vegetative and reproductive traits of M. micrantha in the three urban habitat types.
Different lower case letters indicate significant differences between the seasons, as revealed by Tukey’s HSD test and Mann-Whitney test at p = 0.05 level.
(12.2 n∙mol∙g−1 ± 2.1; p = 0.005) than those in abandoned plots (5.4 n∙mol∙g−1 ± 1.2). The plants in the natural areas showed maximum SLA (14854.9 mm2∙g−1) in comparison to the other two habitats. The abandoned plots had the maximum number of associated species (5.25 ± 0.4) and M. micrantha was found to attain maximum height (6.2 cm ± 0.8) in this habitat.
3.2. Reproductive Traits of M. micrantha
Significantly high amount of reproductive N (2.2% N∙mg−1 ± 0.2) was found in the roadside population of M. micrantha when compared to that of abandoned plots (1.6% N∙mg−1 ± 0.03; p = 0.011). However, the final germination percentage of the seeds from the roadside population was found to be the lowest (71.8% ± 3.5) among the three habitat types. The lightest seeds of M. micrantha population in natural habitats (0.0059 gm ± 0.0002) showed fastest germination capability in terms of having the lowest MGT and T50 values (Table 3). In the abandoned plots, M. micrantha population produced heavier seeds (0.0086 gm ± 0.0012) and took a significantly longer time to germinate (MGT = 6.2 days ± 0.8) compared to those from roadside population (5.1 days ± 0.5; p = 0.038) and natural areas (4.5 days ± 0.1; p = 0.009).
3.3. Seed Survival Functions of M. micrantha
The analysis of seed germination survival functions using the log-rank test, presented in the Kaplan-Meier survival plots (Figure 2), revealed a significant difference in germination survival functions between the seeds of the three different habitats (χ2 = 35.1; p < 0.001). The seeds of M. micrantha from natural areas showed significantly better germination survival function than the seeds from roadside population (χ2 = 29.3; p < 0.001) and those from abandoned plots (χ2 = 18.4; p < 0.001). As evident from the survival plots (Figure 2), the seeds sampled from the abandoned plots (represented by blue line) required more number of days for the germination process to be completed (as evident from more number of step functions) in comparison to that of the seeds sampled from roadsides as well as natural areas.
The probability of having censored seeds (not having germinated) after 7 days
![]()
Figure 2. Kaplan-Meier survival plots of M. micrantha seeds sampled from three different habitats. The final cross represents censored observations.
(t = 7) was maximum in abandoned plots [P (7) = 0.36 ± 0.02], followed by those in roadsides [P (7) = 0.33 ± 0.02] and natural habitats [P (7) = 0.2 ± 0.016] (Table 4). It took 12 days for the germination process to be completed in seeds sampled from M. micrantha population growing in natural habitats with the lowest probability of having the censored seeds [P (12) = 0.178 ± 0.016] compared to 15 days in those from roadside population [P (15) = 0.282 ± 0.018] and 21 days in those from abandoned plots [P (21) = 0.19 ± 0.016].
3.4. Analysis of Soil Parameters in the Three Habitat Types
Soil nitrogen content was found to be significantly low along roadsides (0.0109% ± 0.0012) in comparison to natural areas (0.0207% ± 0.0026; p = 0.008) and abandoned plots (0.0239% ± 0.0019; p < 0.001) (Table 5). The OC content was the lowest in roadside soils, though not significant. MBC values (501.4 µg/gm ± 61.6) were significantly lower along roadsides when compared to natural areas (1101.6 µg/gm ± 120.6; p < 0.001) and abandoned plots (1267.6 µg/gm ± 226.5; p = 0.005). Among the heavy metals, manganese (Mn) content was significantly
![]()
Table 4. Estimated probability of having censored seeds at time t (varying from 4 to 21 days) for the seed lots sampled from the three different habitats of the study area.
“-” in a cell indicates no germination event recorded on that day, and thus the probability of having censored seeds remains unchanged from the previous day.
![]()
Table 5. Estimates of soil parameters in the three habitats of the study area.
Different lower case letters indicate significant differences between the seasons, as revealed by Tukey’s HSD test and Mann-Whitney U test at p = 0.05 level.
low in roadside soils (27.1 ppm ± 4.3) as compared to natural areas (65.7 ppm ± 3.0; p < 0.001) and abandoned plots (63.8 ppm ± 4.0; p < 0.001).
4. Discussion
The ability of the plant to grow in all the studied habitats of an urban locality was evident from the non-significant difference in the productivity of M. micrantha population (vegetative biomass) across the habitat types. However, a significant high amount of reproductive biomass of M. micrantha in the roadside population possibly indicated the ability of the plant to modulate its reproductive trait in response to disturbance prevalent along roadsides. Similar kinds of trait modulation in disturbance gradients of urban habitat have been reported in other plants, e.g. Ricinus communis [33] and Calotropis procera [34] .
4.1. Modulation of Plant Traits along Roadsides
The soil along roadsides appeared to be resource poor compared to the other two habitats, as evident from a significantly low soil N, Mn content and MBC values. While soil nitrogen is critical for plant growth [66] , soil Mn is an essential micro-nutrient and is the key to the entire status of soil redox [67] . A potentially low redox is known to negatively affect soil microorganisms [68] , which is probably reflected from the low MBC values in the roadside soils.
A high probability of germination failure in this habitat could have resulted from the low seed resource content as evident from its lighter seeds. However, the roadside M. micrantha population was found to produce a large number of seeds in order to ensure population sustainability. The roadside plants are often exposed to high vehicular disturbance which enables the seeds to be carried over long distances [69] [70] . To ensure survivability of seeds, the plants possibly allocated more resources to its reproductive part, as evident from the significantly high amount of reproductive N content. The capacity to produce a large number of seeds at the expense of vegetative structures is one of the key strategies of the invasive species to maintain a steady dynamic population [33] .
This strategic adaptation of M. micrantha was also evident in the laminar characteristics of its roadside population through its high LDMC values, signifying high content of sclerenchyma and vascular tissues [71] , and lower amount of laminar N content. More-over, large amount of dust deposition on the leaf surface of the roadside population could result in blockage of stomatal conductance [72] [73] which might decrease photosynthetic efficiency. To counteract this and to compensate for the preferential resource allocation to reproductive parts, the roadside plants were found to produce significantly large LA for better acquisition of sunlight and increased photosynthetic efficiency.
It is interesting to note that in this low resource environment M. micrantha, being a disturbance tolerant species [74] , could exist in monocultures along the roadsides. A high uptake rate of mineral N by M. micrantha for its monospecific growth could also be responsible for the low soil N concentration observed along roadsides [36] [75] . Swamy and Ramakrishnan [76] reported high phosphorus concentration in shoots of M. micrantha which indicates its capacity to mobilize and store soil P. High levels of available P in M. micrantha invaded roadside plots could also influence its growth induced by a rapid phosphorus cycle [75] .
The ground cover form of M. micrantha, as found in this study, is very likely to form a canopy over the resident species, causing their death and subsequent decay which could be another major strategy of M. micrantha to achieve dominance in the community [79] . Moreover, this life form could be directly detrimental to native vegetation since it covers the soil surface and could smother other low-growing plants by inhibiting native seedling regeneration [77] . The low number of average species richness accounts for the loss of other associated species in this habitat.
4.2. Modulation of Plant Traits in Natural Areas
A significantly high amount of proline in the leaves of M. micrantha could be indicative of the higher stress that the population in natural areas are exposed to compared to the other two habitats possibly due to the influence of more anthropogenic disturbance. Proline acts as a marker of stress and its accumulation may be part of a stress signal which might influence adaptive responses [78] in plants by modulating both its vegetative as well as reproductive traits. Among the vegetative traits, possession of high SLA allows efficient capture of solar energy which enables opportunistic resource acquisition for growth and reproduction in invasive plants. This appears to be a key mechanism that makes SLA so important for invasive species [79] . SLA is closely correlated with other plant traits such as photosynthetic capacity, leaf N content, leaf life-span, and importantly, relative growth rate [80] . In this non-limiting resource environment of natural areas with comparatively higher soil N values, high SLA reflected the expected return on assimilated resources [81] , thereby maintaining the productivity of the population. The ability to produce leaves with an increased surface area rapidly and to avoid significant investment of biomass in long lasting structures is critical in disturbed natural habitats where fast growth is supreme.
Among the reproductive traits, the population produced light seeds with fast germination ability. The presence of low amount of storage food reserves in the seeds, as evident from its low seed weight, could have resulted in faster (low MGT and T50) and more efficient germination of seeds. This is also evident from the low probability of having censored seeds at the end of the germination experiment. A capacity for rapid germination is a useful strategy to mitigate the effects of inter-specific competition [82] [83] , as it can suppress the germination or establishment of later germinating seedlings of neighboring species [84] . The seed germination efficiency has been considered as one of the key traits of plant invasiveness [30] . Increased disturbance enhances seed production [85] and copious production of light, wind-dispersed seeds is generally correlated with the ability to respond to large disturbances [86] .
4.3. Modulation of Plant Traits in Abandoned Plots
The absence or minimal presence of disturbance in the abandoned plots was evident from a significantly low amount of proline in M. micrantha leaves, and the increased availability of soil nutrients, resulting in a higher species richness. In this habitat, with more number of resident species in the community, M. micrantha was found to increase its average population height to be able to harvest more light energy, and increase its photosynthetic efficiency. The height of a population has been recognized to be one of the key strategies for invasive plant success in a competitive environment [87] .
Less disturbed abandoned plots facilitate soil microbes to utilize organic carbon more efficiently than in natural areas. Consequently, an increase in microbial biomass was observed in this habitat. A high microbial biomass in soils with low organic carbon probably resulted in a higher soil microbial quotient in abandoned plots when compared to those from natural areas. This quotient is an important index by which the ability of soil micro-organisms in the recycling of carbon, nitrogen, and phosphorus in soils can be assessed [88] . High values of the microbial quotient, as observed in this habitat, reflects the efficiency of microbial incorporation and stabilization of soil organic carbon by the soil mineral fractions [89] . It, therefore, indicates that the average availability of OC is greatly improved and turnover on soil carbon pool is enhanced. This increase of bioavailability of soil nutrients may be another factor for M. micrantha in becoming a successful invader [63] . A well-developed root system of M. micrantha also ensures a high nutrient competitive ability compared to other associated plants with lower root biomass [74] , enabling it to grow rapidly, and to suppress other plants in the process.
M. micrantha population were found to have the highest seed weight, thereby implying more food reserves which might help in establishing its progenies in a community where a large number of species compete for space and resources. The high amount of storage material in the seeds enable seedlings to compete better with superior survivability [33] , eventually leading to a significantly high MGT of the seeds. However, despite having the longest duration of germination process, as found in the survival analysis, the probability of having censored seeds is less than 20%. This finding indicates the efficiency of the M. micrantha population to allocate resources in production of seeds with enough food storage to sustain the germination process in a competitive environment and to assure efficient germination as well.
5. Conclusions
This study, for the first time, demonstrates the phenotypic plasticity of M. micrantha traits (both vegetative and sexual reproductive traits) in a disturbed urban environment. The production of more reproductive biomass in the roadside population, modulation of both vegetative and reproductive traits in anthropogenically disturbed natural areas and increased population height with a longer span of seed germination in abandoned plots enabled the plant to maintain its population in the different habitat types.
Patch of M. micrantha was found close to road networks in Kolkata in our earlier study [5] which is indicative of its increasing chance of dispersal to uninvaded interior areas where the plant can successfully grow and establish its population. Moreover, the plant has also been observed to grow luxuriantly along the margins of the waterbodies [90] . With its highly adaptive ability, the plant is therefore capable of increasing its range from conventional terrestrial to littoral habitats also, thereby escalating the problem many folds. Given the fact that M. micrantha is difficult to control once it attains dominance in the community, a continuous monitoring, irrespective of habitat types, should be prioritized to detect and timely respond to a rapid increase in its population.
Acknowledgements
The authors would like to thank Indian Statistical Institute for necessary funding of this research. The authors are thankful to the Botanical Survey of India for their assistance in identification of the plant species and Soil, Water and Manure Testing Laboratory of Vivekananda Institute of Biotechnology, Nimpith, West Bengal (India) for their assistance in analysis of soil parameters. The authors are grateful to Prof. Anup Dewanji of Applied Statistics Unit for statistical advice, and Mrs. Arpita Ganguly, Mr. Sandip Chatterjee and Mr. Susant Mahankur (AERU, ISI) for their technical and field assistance. We would like to thank the anonymous reviewers for their helpful suggestions.