Received 8 January 2016; accepted 12 March 2016; published 15 March 2016

1. Introduction
C. aurantifolia the family Rutaceae is rich in volatile oils and terpenoids, glycosides and alkaloids. Many of them are with remarkable antimicrobial activities. The seeds of C. Aurantifolia species showed a wide range of biological activities [1] .
The total extracts of C. aurantifolia seeds were tested against Gram positive and Gram negative bacteria, and the three extracts of methanol, chloroform and ethanol exhibited significant activity and the methanolic extract gave the highest activity and was selected for further phytochemical investigations.
The results of antibacterial activity, phytochemical screening and chemical analysis of C. aurantifolia seeds are reported in the present work.
2. Materials and Methods
The C. aurantifolia seeds were collected from fruits purchased from the local market and were identified at the medicinal and Aromatic Plants Institute, the National Centre for Research, Khartoum, Sudan.
The following microorganisms were used for assessment of antimicrobial activity:
1) Bacillus subtilis (Gram positive) NCTC 8236.
2) Staphylococcus aureus (Gram positive) ATCC 25923.
3) Escherichia coli (Gram negative) ATCC 25922.
4) Proteus vulgaris (Gram negative) ATCC 6380.
5) Klebsiella spp. (Gram positive) ATCC 53657.
6) Shigella spp. (Gram positive) NCTC 4837.
Media: Agar, Nutrient Borth.
2.1. Qualitative Analysis of Chemical Constituents
The diethyl ether, methanol and water extracts were used for the phytochemical screening.
2.1.1. Preparation of Extracts
Twenty five gram of powdered seeds were extracted separately in a Soxhlet apparatus and after removal of solvents. The percentage yields were determined the extracts were used for phytochemical screening, antimicrobial activity, chromatographic analysis for isolation and identification of active constituents [2] .
2.1.2. Sterols and Triterpenes
The diethyl ether extract (10 ml) was dissolved in 0.5 ml acetic anhydride, chloroform (0.5 ml) and concentrated sulphuric acid (2 ml) was placed at the bottom of the test tube by means of a pipette [2] .
2.1.3. Carotenoids
The ether extract (10 ml) was evaporated to dryness and saturated solution of antimony trichloride in chloroform (3 drops) was added (Carr price’s reactions) [2] .
2.1.4. Coumarins
3 ml of extract added water. After cooling was divided is two test tubes contained the reference, and the aqueous solution of the second tube was made alkaline with 0.5 ml of ammonia solution (10%) [2] .
2.1.5. Tannins
The methanol and water extracts (0.5 - 1 ml) were diluted water (1 - 2 ml) and 2 - 3 drops of diluted solution of ferric chloride were added [2] .
2.1.6. Reducing Sugars
The methanol extract (1 ml) was diluted with distilled water (1.2 ml) and Fehling’s (1 and 2) reagent (0.5 - 1 ml) solutions were added and heated [2] .
2.1.7. Alkaloid Salts
20 ml of the methanol extract were transferred to a beaker and evaporated on boiling water-bath. 5 - 10 ml of hydrochloric acid (10%) were added to the residue. The alkaloids became salts of mineral acids from the aqueous solution. The alkaloids were liberated as free bases with the help of ammonia solution (10%) and then extracted with ether or chloroform. The solution 1.5 ml 2% hydrochloric acid solution. The acid solution was then divided into three test tubes: one was the reference and in the other two test tubes, 2 - 3 drops of Mayer’s or Bertrand’s reagent were added [2] .
2.1.8. Saponins
2 ml of the aqueous extract added 2 ml of distilled water and shaken, vigorously [2] .
2.2. Chromatographic Analysis
The extracts were screened in different solvent systems by thin layer chromatography and the active extracts were fractionated by liquid solid chromatography.
2.2.1. Column Chromatography
The column was packed with about 100 g of silica gel using the wet packing procedure with chloroform. The three grams of chloroform extract were placed on top of column and eluted with the solvent chloroform, followed by increasing amounts of methanol in chloroform and finally with pure methanol [3] .
2.2.2. Thin Layer Chromatography
The compositions of fractions collected from the column were monitored by the following solvent systems:
Chloroform/methanol (8.5:1.5) and
Toluene/ethyl acetate/formic acid (5:4:1).
The fractions were further purified by preparative TLC and detection with UV-light and 1% vanaillin in conc. sulphuric acid [3] .
2.3. Assessment of Antimicrobial Activity
Microbiological techniques followed were those described by Atlas [4] and [5] .
2.4. Organic Matter
Two-grams of each sample were put in a previously weighed porcelain crucible. The crucible was placed in a muffle furnace at 550˚C for six hours, transferred to a desiccator and then weighed. The ash weight was determined and its percentage was calculated as follows:
Weight (g) of (crucible + sample) before ignition = x
Weight (g) of (crucible + sample) after ignition = y
Organic matter% = [(x − y)/(Sample weight (g))] × 100
2.5. Nitrogen and Crude Protein Content
2.5.1. Nitrogen
The procedure used is a two-stage process in which the released gum samples are digested in hot concentrated sulfuric acid and the ammonia using sodium hydroxide, is neutralized using standard acid (AOAC; 1984) [6] .
A. Digestion
Sample + (H2SO4 conc. + catalyst)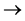 (NH4)2SO4
(NH4)2SO4
(NH4)2SO4 + (1) Alakali (2) steam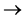 2NH3 + H2BO3
2NH3 + H2BO3
B. Neutralization
NH3 + B3BO3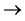 NH4 + H2BO3
NH4 + H2BO3
The borate anion equivalent to the ammonia produced is back titrated with standard HCl (0.02 mol∙dm−3).
B2BO3 + HCL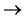 H3BO3 + Cl−
H3BO3 + Cl−
Accurately weighed 0.2 g of gum sample, One gram of powdered was added to mixture of potassium sulfate and cupric sulfate (10:1). The contents transferred to a steam distillation unit, 20 ml of 40% sodium hydroxide solution were added, and distillation was carried out with steam. The distillate was collected in 10 ml of boric acid solution (2%) to which methyl red indicator were added and titrated against O.OIN-BCL. The same procedure was carried out for a blank.
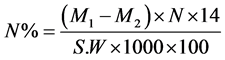

where:
M1: ml of HCl that neutralized the sample distillate.
M2: ml of HCl that neutralized the blank distillate.
N: the normality of HCl titrate (0.01).
14: each ml of HCl is equivalent to 14 mg nitrogen.
S.W: sample weight (0.2 g).
1000: conversion factor from gram to mg [6] .
2.5.2. Protein
Crude protein % = N% × 6.6 [6] .
3. Results and Discussion
The extract of C. aurantifolia seeds with chloroform, methanol and ethanol was screened against Gram positive and Gram negative bacteria. The antimicrobial activity was significant and gave grounds to the extract in Table 1. Phytochemical screening of extracts is presented in Table 2.
![]()
Table 1. Results of antimicrobial activity of the chloroform, methanolic and ethanolic extracts of C. aurantifolia seeds.
The chloroform extracts of C. aurantifolia seeds fractionation by using a column packed with silica gel and elution with chloroform and methanol. Biological monitoring of activity gave several active fractions of six fractions which have been isolated in a pure state by preparative TLC. Fraction 1 and 2 elution were with chloroform pure, fraction 3 elution by chloroform:methanol (95:5), fraction 4 elution by chloroform:methanol (90:10), fraction 5 elution by chloroform:methanol (75:25) and fraction 6 elution by chloroform:methanol (50:50). The result was shown in Table 3.
Chemical analyses of the seeds have been done such as determination of ash content (0.27%), nitrogen content (3.22), and protein content (21.25%). All trials were restored three times, according to the average result in Table 4.
In Figure 1, TLC of six fractions chloroform extract of C. aurantifolia Stationary phase Silica gel 60 precoated plates, thickness 20 mm the mobile phase by used N-butanol:glacial acidic acid:water (5:4:1) Detection
![]()
Figure 1. TLC of column fractionation of the chloroform extract of C. aurantifolia seeds: Stationary phase = Silica gel 60 precoated plates, thickness 0.20 mm. Mobile phase: Toluene/ethyl acetate/formic acid (5:4:1). Detection: UV-light (366 nm); 1% vanillin in conc. H2SO4.
![]()
Table 2. Results of the general phytochemical screening of the C. aurantifolia seeds.
Positive result: (+); Negative result: (−).
![]()
Table 3. Evaluation of antimicrobial activity of column fractionation of the chloroform extract of C. aurantifolia seeds.
![]()
Table 4. Chemical analysis of C. aurantifolia seeds.
by UV-light (366 nm) and 1% vanillin in conc. H2SO4.
4. Conclusion
Some recent studies reported important uses of seed extracts of C. aurantifolia in complementary medicine and we recommend more studies on the seeds composition of the Sudanese varieties.