Elimination of Pollution of Groundwater by On-Site Sanitation Systems ()
Received 31 October 2015; accepted 14 December 2015; published 17 December 2015


1. Introduction
One of the major challenges faced by the developing countries is that of human waste management and disposal. Nearly half the urban population lack adequate safe waste disposal facilities and a very insignificant portion of rural population do not have proper waste disposal facilities [1] . The sanitation practices that are promoted fall into one of the two broad systems: on-site and offsite sanitation systems. The onsite sanitation system is subdivided into three major types: flush and discharge, ecological (EcoSan toilets) and the traditional drop and store (pit latrine). Human faeces are dangerous to health and detrimental to the environment if not purified or untreated before being disposed into the environment. A WHO Study Group defined sanitation as “the means of collecting and disposing of excreta and community liquid wastes in a hygienic way so as not to endanger the health of individuals and the community as a whole” [2] . Hygienic disposal of human wastes that does not endanger health should be the underlying objective of all sanitation programmes [3] .
2. Pathogen Characteristics
Human excreta contains four types of pathogens; Eggs of helminthes (worms), Protozoa, Bacteria and Viruses. Helminthes and protozoa are relatively large (>25 μ) they are removed efficiently through physical filtration in the soil and it is unlikely they will pollute groundwater. Bacteria and viruses are much smaller. The percolating effluent from sanitation systems can transport bacteria and viruses into the groundwater and these organisms may be ingested and cause infection. Faecal matter contains an average of 109 bacteria per gram, while an infected individual can excrete up to 106 viruses per gram of faeces [4] . The rate of inactivation by natural or artificial processes depends on both the efficiency of removal and the numbers initially present. The distance over which enteric bacteria are traced depends on their death rate, velocity of the groundwater, initial concentration of enteric bacteria and their dispersion within the groundwater body. The maximum linear diffusion appears to be that distance which the groundwater flows in about 10 days. Viruses are much smaller than bacteria. Removal appears to depend almost entirely on adsorption which is not an irreversible process [4] .
3. Faecal Pollution in Drinking Water
Waterborne diseases such as cholera, typhoid, amoebic and bacillary dysentery and other diarrheal diseases are caused by the ingestion of water contaminated by human wastes or animal faeces or urine containing pathogenic bacteria. Worldwide approximately 250 million people are infected with waterborne diseases, which results in 10 to 20 million deaths annually. In Uganda about 21 million (approximately two thirds of the population) lack basic sanitation [5] . The control of these waterborne diseases is performed by monitoring the microbiological quality of underground drinking water by routine assessment [6] . The presence of coliforms such as Escherichia coli (E. coli) in water indicates faecal pollution since coliforms are present in large numbers in the intestinal flora of both humans and warm blooded animals [7] .
The Uganda National Environment Management Authority (NEMA) disposal requirement of treated wastewater is a maximum limit of 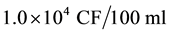 E. coli (faecal coliforms), [8] . Unfortunately on-site sanitation systems receive and dispose human waste directly to the underground streams (aquifer) unpurified at
E. coli (faecal coliforms), [8] . Unfortunately on-site sanitation systems receive and dispose human waste directly to the underground streams (aquifer) unpurified at 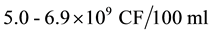 E. coli thus contaminating ground water sources; posing public health threats with water related diseases like typhoid, cholera, and dysentery. This results into high government expenditure on treatment. COD corresponds to the amount of oxygen required to chemically oxidize organic matter by a strong oxidant for example permanganate
E. coli thus contaminating ground water sources; posing public health threats with water related diseases like typhoid, cholera, and dysentery. This results into high government expenditure on treatment. COD corresponds to the amount of oxygen required to chemically oxidize organic matter by a strong oxidant for example permanganate 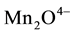 or dichromate
or dichromate 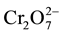 in acid solution. BOD determines the oxygen consumption of microorganisms during biodegradation of organic matter. It is widely used parameter to quantify organic matter pollution in the waste water as it best reflects the actual process taking place in sewage treatment plants. The rate of biodegradation depends on temperature, therefore the standard BOD test is fixed at 20˚C and the incubation period is 5 days. This is expressed as
in acid solution. BOD determines the oxygen consumption of microorganisms during biodegradation of organic matter. It is widely used parameter to quantify organic matter pollution in the waste water as it best reflects the actual process taking place in sewage treatment plants. The rate of biodegradation depends on temperature, therefore the standard BOD test is fixed at 20˚C and the incubation period is 5 days. This is expressed as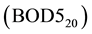 .
.
4. Methodology
The study carried out tests on influents and effluents of experiential setups of layered charcoal and saw dust purifying materials for E. coli or fecal coliforms, Chemical oxygen demand [COD] and Biological oxygen demand [BOD] parameters. There are vast heaps of waste charcoal dust left over from use of charcoal as fuel and excess saw dust from carpentry workshops and saw mills in various urban centers in Uganda with no adequate recycle methods leading to environmental solid waste pollution. The research therefore investigated the efficiency and practical applicability of these materials packed membranes as sewage purifiers for on-site sanitation systems like, septic tanks, cess pool pits, pit latrines, pour-flush pit excreta systems.
4.1. Experimental Setup
The apparatus was erected as shown in Figure 1; Material membranes (charcoal dust and saw dust) were picked from disposal grounds. One 18.5 liter mineral water bottle was filled with 150 mm well compacted layers of charcoal dust at the bottom, followed by 150 mm gravel, 150 mm charcoal dust, 50 mm of stone dust at the top. Then another 18.5 liter mineral water bottle was filled with 150 mm well compacted layers of saw dust at the bottom, followed by 150 mm gravel, 150 mm saw dust, and 50 mm of stone dust at the top. The sewerage influent was then poured and allowed to flow under gravity through arrangement of compacted layers. The resultant effluent water was collected in beakers ready for laboratory tests. The material membrane layers of saw dust and charcoal dust were then varied to different thicknesses of 200 mm and 300 in fresh set of bottles, keeping gravel at 150 mm thickness in each case.
4.2. Waste Water Sampling and Field Study
Waste water samples were collected on daily basis for three days. Central Waste Water Treatment Plant in Kampala for the National Water and Sewerage Corporation (NWSC) sampling point was selected as a source of raw sewerage. The Central Waste Water Treatment Plant was chosen because it provided an experimental room for the setup experiment; it also had well mixed and easily sampled sewage and proximity of laboratories for easy access to perform the prescribed tests within the 3 hour limit set by the national standards.
4.3. Sample Size
The random samples were collected; the samples were grouped into three sets. Each collected per week, preserved, analyzed at the quality control laboratory, values recorded and quantified accordingly. The sampled volumes of the pollution parameters were as in given in Table 1.
4.4. Determination of Characteristics
Physical characteristics were determined by observing the colour of both the influent and effluent from the experimental setup. Chemical characteristics involved determination of BOD and COD. Micro biological characteristics aimed at determining feacal coliforms count of influent and effluent. This was used to indicate the extent of feacal pollution of waste water thus presence of enteric pathogens.
![]()
Table 1. Sampled volumes of pollution parameters.
4.5 Biochemical Oxygen Demand (BOD5)
Samples of wastewater at the inlet channel were collected using well-cleaned, thoroughly rinsed and air dried incubation bottles. Water seals were created by aspirating water into the mouth of the flair bottle and covering the mouth with an Aluminum foil paper or cup to minimize effects of evaporation in the water seal. Then, the samples were prepared to required dilutions. Determination of biochemical oxygen demand (BOD5) was done using Azide modification of wrinkle oxygen electrode method. The method involved filling an airtight bottle with a sample and incubating it at a temperature of 20˚C for 5 days. Dissolved oxygen (DO) was measured before and after incubation and the BOD was measured as a difference of the initial and final DO.
For each test bottle meeting the 2.0-mg/l minimum DO depletion and the 1.0-mg/L residual.
DO, BOD5 was calculated as follows;
When dilution water is not seeded:
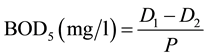 (1)
(1)
When dilution water is seeded:
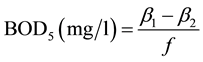 (2)
(2)
where:
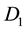 = DO of diluted sample immediately after preparation, mg/l,
= DO of diluted sample immediately after preparation, mg/l,
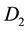 = DO of diluted sample after 5 d incubation at 20˚C, mg/l,
= DO of diluted sample after 5 d incubation at 20˚C, mg/l,
P = decimal volumetric fraction of sample used,
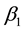 = DO of seed control before incubation, mg/l,
= DO of seed control before incubation, mg/l,
![]() = DO of seed control after incubation mg/l, and
= DO of seed control after incubation mg/l, and
f = ratio of seed in diluted sample to seed in seed control = (% seed in diluted sample)/(% seed in seed control).
If seed material is added directly to sample or to seed control bottles:
f = (volume of seed in diluted sample)/(volume of seed in seed control).
4.6. Chemical Oxygen Demand (COD)
The apparatus included; digestion vessels, heating blocks or ovens to operate at 150˚C ± 2˚C. Samples from specific sampling points were collected in glass bottles or clean plastic bottles as above. Determination of chemical oxygen demand (COD) was done using closed reflux calorimetric method. 50 ml of wastewater sample, diluted was refluxed in a strongly acid solution with a known excess of Potassium dichromate solution [K2Cr2O2]. The amount of dichromate consumed is proportional to the oxygen required to oxidize the oxidizable organic matter in the sample.
4.7. Faecal Coliforms (E. coli Colonies)
Samples were collected in a clean, clear, sterile glass/plastic bottles with a stopper. A septic method of sampling was used to avoid sample contamination. The representative samples were preserved in a cool box containing ice to avoid alteration of the bacteriological status of the sampled wastewater. Equipment and apparatus needed were; a filtering unit, membrane filter pads, autoclave or ovens, incubator, Petri-dishes, forceps, pipettes (granulated type), digital counter, transfer pipette, water still, measuring cylinder, sterilizing banner, hot plate, disinfectant (70% ethanol), weighing balance and thermometer.
Determination of Faecal coliforms was done using membrane filtration method employing Laurel Sulphate broth. A measure of 100 ml was filtered through a membrane filter composed of cellulose esters which retains all the bacteria on the surface of the membrane which was then incubated with the girded side upon a selective media. The Petri-dish was covered with the lid upper most and placed onto the carrier, then placed in the incubator. The incubator was set at 44˚C ± 0.5˚C for 30 mins before the sample was incubated. The thermometer was placed in the incubator to cross check the temperature, then the sample incubated for 24 hours. After incubation the carriers were removed and Petri-dishes allowed cool for 10 mins to allow a false yellow colour to lose and ensure that the yellow was only for the typical E. coli colonies. A low power microscope (10 - 15× magnification lens) and a counting pen were used to count the colonies. All the yellow, pale yellow colonies which are convex, dome-shaped colonies with a reflective surface were counted as true colonies which may be feacal coliform or suspect E. coli but if not incubated at 35˚C then it was considered to be total coliform. The suspended E. coli was confirmed using both brilliant green broth and Tryptone water.
To compute E. coli density, the following empirical formulae were used. Equation (3) was applied for coliform density on a single membrane filter.
![]() (3)
(3)
Equation (4) is applied for average coliform density, if no filter meets the desired minimum colony count. The equation is suited 1) duplicates; 2) Multiple dilutions, or more than one filter/sample.
![]() (4)
(4)
For total solid (dry weight basis), the feacal coliforms per gram dry weight for bio solid analysis was calculated as shown in Equation (5)
![]() (5)
(5)
Data collected was analyzed using empirical formulae above. The results were compared with the NEMA effluent standards (maximum permissible limits) for discharge of waste water in Uganda. After analysis of the results, the discussions included design of an appropriate technology to exploit the result of the experiment.
5. Results
From Table 2, it is seen that the initial flow through charcoal dust is fast and it slows down as the charcoal absorbs water, on the other hand the initial flow in saw dust is low because the saw dust is absorbing the water, after it is saturated with water the flow increases. Charcoal dust is very good in removing colour and suspended solids, whereas saw dust is not good.
From Table 3, the raw sewage samples collected at the inlet of disposal unit gave an average value of ![]() bacterial colonies by density. After filtration treatment/purification on a layers of (150 mm, 150 mm, 150 mm) of (charcoal, gravel, charcoal) and (sawdust, gravel, sawdust), the microbial load of bacteria reduced to
bacterial colonies by density. After filtration treatment/purification on a layers of (150 mm, 150 mm, 150 mm) of (charcoal, gravel, charcoal) and (sawdust, gravel, sawdust), the microbial load of bacteria reduced to![]() , a reduction factor of 4265 for charcoal and
, a reduction factor of 4265 for charcoal and![]() ,a reduction factor of only 181 for saw dust. Despite these high reduction rates, the values obtained are still high above the discharge limit of
,a reduction factor of only 181 for saw dust. Despite these high reduction rates, the values obtained are still high above the discharge limit of ![]() for waste water by National Environment Management Authority (NEMA).
for waste water by National Environment Management Authority (NEMA).
From Table 4, the average contamination density of raw sewage sample is ![]() feacal coliforms (E. coli). Effluent from charcoal and saw dust gave
feacal coliforms (E. coli). Effluent from charcoal and saw dust gave ![]() and
and ![]() fecal coliforms (E. coli) densities giving reduction factors of 5513 and 145 respectively. The purification layers were 200 mm each and gravel kept at 150 mm thickness. This set shows a remarkable improvement in purification of microbial bacteria. But still fecal colonies are slightly above upper recommended discharge limit of
fecal coliforms (E. coli) densities giving reduction factors of 5513 and 145 respectively. The purification layers were 200 mm each and gravel kept at 150 mm thickness. This set shows a remarkable improvement in purification of microbial bacteria. But still fecal colonies are slightly above upper recommended discharge limit of![]() .
.
BOD5 reduced from 647.6 mg/l in the influent to 2 mg/l and 438.5 mg/l for charcoal and saw dust respectively against environmental standards of 50 mg/l. COD reduced from 1345 mg/l in the influent to 7.6 mg/l and 967 mg/l for charcoal and saw dust respectively against environmental standards of 100 mg/l.
From Table 5 for purification layers of 300 mm each and keeping gravel thickness at 150 mm. ![]() /
/ ![]() and
and ![]() fecal coliforms (E. coli) by densities were realized from a raw average sewage
fecal coliforms (E. coli) by densities were realized from a raw average sewage
![]()
Table 2. Observed results from the experimental setup.
![]()
Table 3. Table of analytical results (150 mm membrane).
![]()
Table 4. Table of analytical results (200 mm membranes).
![]()
Table 5. Table of analytical results (300 mm membranes).
density of ![]() giving reduction factors of 44,444 and 375 for charcoal and saw dust respectively. This set shows a remarkable improvement in purification of microbial bacteria with fecal colonies within the upper recommended discharge limit of
giving reduction factors of 44,444 and 375 for charcoal and saw dust respectively. This set shows a remarkable improvement in purification of microbial bacteria with fecal colonies within the upper recommended discharge limit of ![]() for the case of charcoal dust material.
for the case of charcoal dust material.
BOD5 reduced from 698 mg/l in the influent to 0.5 mg/l and 42 mg/l for charcoal and saw dust respectively against environmental standards of 50 mg/l. COD reduced from 1492 mg/l in the influent to 3.0 mg/l and 115 mg/l in effluent for charcoal and saw dust respectively against environmental standards of 100 mg/l.
6. Discussions
The saw dust effluent is not safe to be discharged into water bodies. This would be harmful to aquatic life, because all the oxidation oxygen required for their respiration would instead be used for decomposition of the organic wastes. The COD and BOD from charcoal dust filters in Table 4 and Table 5, were lower than the environmental standards. If discharged into water, it will contain less oxygen debts and can sustain aquatic life.
From Table 4 and Table 5, it is seen that charcoal dust has high efficiencies of 99.44% and 99 corresponding test values of 7.6 mg/l and 2.0 mg/l in reducing COD of effluent compared to 28.37% and 32.25%; corresponding to test values of 439 mg/l and 967 mg/l for saw dust. Also charcoal dust has equally high efficiencies of 99.44% and 99% corresponding to test values of 3.0 mg/l and 0.5 mg/l in reducing BOD of effluent compared to 67% and 93.93%; corresponding to test values of 115 and 42 for saw dust. The test values for charcoal dust are below tolerance limits of 100 and 50 mg/l for COD and BOD for safe discharge to the environment. Charcoal dust as sewage purifier is more efficient or better than saw dust. This is because charcoal dust contains carbon which is riddled with small low volume pores that increase the surface area available for adsorption or chemical reactions/filtration. While, sawdust has starch and other additives that are easily oxidized or broken down to un wanted products in solution.
The efficiency of coliform reduction increases with increasing depth of the charcoal membrane filter as can be seen in Tables 3-5. For greater than 300 mm membrane, the effluent can be discharged to the environment without much threat to stream water and can be suited for recreation and farm irrigation. For ground water resource the technology gives preliminary treatment which when coupled with soil beneath; the resultant water is effectively purified fit for drinking.
Application of Charcoal Filters for Elimination of Groundwater Pollution
Normally waste water from a septic tank is discharged directly into the ground via a soak pit. It is proposed that a lined purification chamber be introduced between the septic tank or cesspool and the soak pit as illustrated in Figure 2 below.
7. Conclusions
・ The experimental tests showed that fecal coliforms (E. coli) are removed by 300 mm thick Charcoal dust filtering media to values slightly above standard discharge limits and its efficiency is directly proportional to the thicknesses of the charcoal dust layers.
・ Charcoal dust as a sewage purifier is better than saw dust; because; it is efficient in reducing BOD and COD contents to considerably very low values satisfying environmental standards and 300 mm thick layer has very high average efficiency of 99.98% elimination level for fecal coliforms in the waste water.
・ Introducing a lined purification chamber with layers of charcoal dust between the septic tank or cesspool and the soak pit will eliminate the pollution of ground water.
![]()
Figure 2. Application of charcoal filters in purifying sewage.
8. Limitation of the Research
・ It was not possible to use septic tank influent because the tests needed to be carried out near the National Water and Sewerage Corporation Laboratory. However raw sewage from sewers which was used has a higher count of pathogens than effluents of the septic tank. Further research is recommended using effluents of septic tanks and increasing the filters until the effluent is satisfies environmental standards.
・ It was noted that the flow in charcoal media is fast at the beginning and reduces with time. There is need to investigate the effect on efficiency of fast soaking the charcoal in water.
Acknowledgements
The management of National Water and Sewerage Corporation; Central Laboratory Bugolobi, P. O. Box 7053, Kampala, for availing their laboratory for the student to conduct the required tests. The Belgian Technical Cooperation for funding this study [9] of which this research is part.