Numerical Simulation Analysis of a Mathematical Model of Circadian Pacemaker Neurons ()
1. Introduction
Repetitive spiking activity is an important feature of excitable cells such as neurons and muscle cells. In the field of applied mathematics, this activity can be extensively analyzed using various mathematical models, which are described by a system of nonlinear ordinary differential equations (ODEs). One example is the Hodgkin-Huxley model, which can generate repetitive spiking because of an interaction between the sodium conductance and the potassium conductance (page 25 in [1] ). In this model, the sodium conductance plays an excitatory role, whereas the potassium conductance plays an inhibitory role. The other example is the Morris-Lecar model, which can also generate repetitive spiking because of an interaction between the calcium conductance and the potassium conductance (page 49 in [1] ). In this model, the calcium conductance and not the sodium conductance plays an excitatory role.
Another type of mathematical model of the repetitive spiking activity is the one in which the repetitive spiking is generated by an interaction among sodium, calcium, and potassium conductances. One example is a mathematical model of circadian pacemaker neurons in the suprachiasmatic nucleus (SCN) [2] , which is described by a system of nonlinear ODEs (see the Materials and Methods in [2] as well as the Materials and Methods of this manuscript). Considering the Hodgkin-Huxley and Morris-Lecar models described above, it is assumed that both the sodium and calcium conductances play excitatory roles in the circadian pacemaker neuron model. How- ever, the difference in the contribution to the repetitive spiking between the sodium and calcium conductances was not clarified in detail in a previous study [2] . In particular, the influence of variations in these two conductance values on the repetitive spiking was not studied in detail in that study [2] . Taking into consideration that it is not only interesting but also very important to analyze the ionic conductance in detail (page 26 in [3] ), it is necessary to investigate the influence of variations in these two conductance values and reveal the difference between these two conductances. Therefore, in the present study, we focused on two system parameters of the circadian pacemaker neuron model, namely the maximal conductance of calcium current (gCa) and the maximal conductance of sodium current (gNa), and we performed numerical simulation analysis.
2. Materials and Methods
A mathematical model of the circadian pacemaker neuron, which has been used in the present study, was a model developed by Sim and Forger [2] . This model is described by a system of nonlinear ODEs and contains six state variables: a membrane potential of the circadian pacemaker neuron [V(mV)] and five gating variables of ionic currents (m, h, n, r, and f). The time evolution of the six state variables is described as follows:
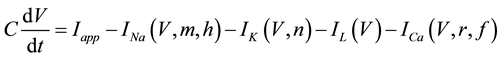 (1)
(1)
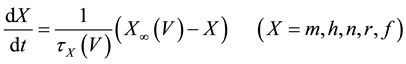 (2)
(2)
where C is membrane capacitance (5.7 pF); INa(V, m, h), IK(V, n), IL(V), and ICa(V, r, f) are a sodium current, a potassium current, a leak current, and a calcium current, respectively, which are defined in Equations (3)-(6) below, in this order; τX(V) (ms) and X∞(V) are time constants of activation/inactivation and steady-state activation/inactivation functions, respectively, which are defined in Equations (7)-(16) below, in this order; and Iapp is applied current.
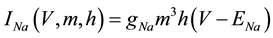 (3)
(3)
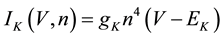 (4)
(4)
 (5)
(5)
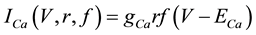 (6)
(6)
where gNa, gK, gL, and gCa are the maximal conductances of a sodium current (the gNa value was varied in the present study; the default value was 229 nS), a potassium current (14 nS), a leak current (1/11 nS), and a calcium current (the gCa value was varied in the present study; the default value was 65 nS), respectively; ENa, EK, EL, and ECa are the reversal potentials of a sodium current (45 mV), a potassium current (−97 mV), a leak current (−29 mV), and a calcium current (61 mV), respectively.
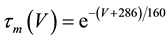 (7)
(7)
 (8)
(8)
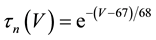 (9)
(9)
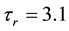 (10)
(10)
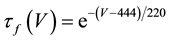 (11)
(11)
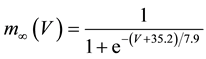 (12)
(12)
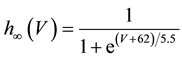 (13)
(13)
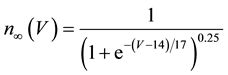 (14)
(14)
 (15)
(15)
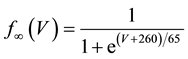 (16)
(16)
Detailed explanations of the model equations have been described previously [2] .
The free and open source software Scilab (http://www.scilab.org/) was used to numerically solve ODEs (initial conditions: V = −80 mV, m = 0.34, h = 0.045, n = 0.54, r = 0.01, f = 0.04).
3. Results
The time courses of the membrane potential of the SCN neuron model under different parameter values are shown in Figure 1. At default parameter values, the model showed spontaneous repetitive spiking (Figure 1(a)), as reported previously (Figure 2(a) in [2] ). When only gCa was completely blocked, the model showed the stable steady state instead of the spontaneous spiking state (Figure 1(b)). When only gNa was completely blocked, the model showed a similar pattern, as illustrated in Figure 1(b) (Figure 1(c)). When both gCa and gNa were set to be values larger than the default values, the model showed the stable steady state (Figure 1(d)). However, the steady state membrane potential value shown in Figure 1(d) was more depolarized than that shown in Figure 1(b) and Figure 1(c).
Figure 2 illustrates the phase diagram of the model in the (gCa, gNa)-parameter space. The dynamical states of the model were classified into three states: the hyperpolarized steady state (white circle), the spontaneous spiking state (black circle), and the depolarized steady state (double circle). A decrease in gNa induced a decrease in the gCa range in which repetitive spiking occurred, and finally, the gCa range disappeared. Therefore, it is concluded that when gNa is zero, repetitive spiking cannot occur, no matter how large the gCa value is. An increase in gNa induced a decrease in the gCa threshold required to induce the transition from the hyperpolarized steady state to the spiking state. For example, even when gCa greatly decreased to 30 nS, repetitive spiking occurred under conditions in which gNa greatly increased to 1603 nS (a seven-fold increase from the default value) (Figure 3(a)). In addition, even when gCa was zero, repetitive spiking occurred under conditions in which gNa was 1603 nS (Figure 3(b)).
4. Discussion
The present study revealed the sensitivity of the model dynamics to variations in gCa and gNa of circadian pacemaker neurons. Although Sim and Forger revealed the difference in time courses between the calcium and sodium currents (Figure 2(c) and Figure 2(d), respectively, in [2] ), they did not clarify whether the calcium and sodium conductances are indispensable to repetitive spiking under the default condition. The present results (Figures 1(a)-(c)) resolved this issue; both gCa and gNa were indispensable to repetitive spiking under the default condition. Interestingly, the present study also revealed the difference between the two conductances. When we considered repetitive spiking under the condition in which the gCa value was smaller than its default value but the gNa value was much larger than its default value, the sodium conductance was indispensable but the
![]()
Figure 1. Time courses of the simulated membrane potential of circadian pacemaker neurons under several (gCa, gNa) conditions. (a) The spontaneous repetitive spiking state at (gCa, gNa) = (65 nS, 229 nS). (b) The hyperpolarized steady state at (gCa, gNa) = (0 nS, 229 nS). (c) The hyperpolarized steady state at (gCa, gNa) = (65 nS, 0 nS). (d) The depolarized steady state at (gCa, gNa) = (80 nS, 350 nS).
![]()
Figure 2. The sensitivity of the dynamical states of the circadian pacemaker neuron model to gCa and gNa variations. Black circle: the spontaneous repetitive spiking state, white circle: the hyper- polarized steady state, white double circle: the depolarized steady state.
![]()
Figure 3. Time courses of the simulated membrane potential of circadian pacemaker neurons under (gCa, gNa) conditions different from those in Figure 1. (a) The spontaneous repetitive spiking state at (gCa, gNa) = (30 nS, 1603 nS). (b) The spontaneous repetitive spiking state at (gCa, gNa) = (0 nS, 1603 nS).
calcium conductance was dispensable to this type of repetitive spiking (Figure 2 and Figure 3(a) and Figure 3(a)).
Previous studies have reported the relationship between ionic conductances and neuronal spiking in various types of mathematical models [4] -[7] . In particular, similar to the circadian pacemaker neuron model analyzed in the present study, a mathematical model of midbrain dopaminergic neurons included the sodium and calcium conductances [4] . In the midbrain dopaminergic neuron model (Figure 4 in [4] ), (1) an increase in gNa induced a decrease in the gCa threshold required to induce the transition from the nonpacemaking state to the pacemaking state and (2) the pacemaking occurred in the absence of the calcium conductance under conditions in which the sodium conductance was set to a large value. The present circadian pacemaker neuron model also exhibited these two characteristics. However, the present results revealed two important differences between these two models: (1) the (gCa, gNa)-parameter space was divided into only two states (the pacemaking and nonpacemaking states) in the midbrain dopaminergic neuron model (Figure 4 in [4] ), whereas the (gCa, gNa)-parameter space was divided into three states (the depolarized steady state, the repetitive spiking state, and the hyperpolarized steady state) in the circadian pacemaker neuron model (Figure 2), and (2) pacemaking occurred in the absence of the sodium conductance under the condition in which the calcium conductance was set to a large value in the midbrain dopaminergic neuron model (Figure 4 in [4] ), whereas pacemaking did not occur in the absence of the sodium conductance even when the calcium conductance was set to a large value in the circadian pacemaker neuron model (Figure 2).
5. Conclusion
The present investigation focused on a mathematical model of circadian pacemaker neurons, performed numerical simulation analysis, and compared this numerical result with that of the previous studies. The important and novel findings of the present study are as follows: 1) there was a difference in the contribution to repetitive spiking under certain conditions between the sodium and calcium conductances: for the generation of repetitive spiking under certain conditions, the sodium conductance played an indispensable role, whereas the calcium conductance was not necessarily essential, and 2) the (gCa, gNa)-parameter space of the circadian pacemaker neuron model showed a different pattern compared with that of the midbrain dopaminergic neuron model. These findings can contribute to our in-depth understanding of the influence of the sodium and calcium conductances on neuronal repetitive spiking.
Acknowledgements
The author would like to thank Enago (www.enago.jp) for the English language review.