The Use of New Chemically Modified Cellulose for Heavy Metal Ion Adsorption and Antimicrobial Activities ()
1. Introduction
Heavy metal pollution has become a serious problem with the rapid increase of global industrial activities. Industrial uses of metals and other domestic processes have introduced substantial amounts of potentially toxic heavy metals into the atmosphere and into the aquatic and terrestrial environments. The contamination of the aquatic systems with toxic heavy metal ions is a problem of global concern. Among the heavy metals, lead causes encephalopathy, cognitive impairment, behavioral disturbances, kidney damage, anemia and toxicity to the reproductive system [1] . The excessive copper concentrations can lead to weakness, lethargy and anorexia, as well as damage to the gastrointestinal tract [2] . Removal of copper and lead heavy metal ions from waste waters is essential from the standpoint of environmental pollution control. Numerous methods have been used to remove heavy metals from waste waters which principally include chemical precipitation, ion-exchange, reverse osmosis, coagulation and flocculation, membrane separation, biosorption, and adsorption [3] .
In recent years, increasing costs and environmental considerations associated with the use of commercial adsorbents, have led to a significant body of research work aimed at developing new low-cost adsorbents derived from renewable resources. In this context, the advantages of using cellulose as the basis for new adsorbent design lie primarily in its high abundance, low cost and the relative ease with which it can be modified chemically [4] . Approaches to cellulose modification have been based on either direct chemical modification approaches [5] -[8] or the grafting of suitable polymer exchange to the cellulose back bone followed by fictionalizations [9] -[12] .
Amongst all the treatment processes mentioned, adsorption using sorbents is one of the most popular and effective processes for the removal of heavy metals from waste water. The adsorption process offers flexibility in design and operation and in many cases produces treated effluent suitable for re-use, free of color and odor. In addition, because adsorption is sometimes reversible, the regeneration of the adsorbent with resultant economy of operation may be possible [13] .
With the growing of public health awareness of disease transmissions and cross-infection caused by the microorganisms, the use of antimicrobial materials has been increased in many applications. The continuous search for potential antimicrobial agents has lead to identification of antimicrobial biomaterials that are based on polymers or their composites. In recent years, antibacterial textile fibers have gained an increasing attention because they offer several interesting properties. It could be either bactericidal (to kill bacteria) or bacteriostatic (to prevent the bacterial proliferation) and in the two cases it protects the human body [14] . Cellulose, which is a naturally occurring complex polysaccharide, is biodegradable and the most abundant renewable organic raw material at low costs in the world. Modification of cellulose by graft copolymerization and direct chemical modification techniques allows one to chemically change the cellulose chain by introducing functional groups, which leads to new cellulose products with new properties [15] .
In this work, we report the chemical modification of cellulose using sodium metaperiodate (NaIO4) oxidation followed by condensation with p-toluidine. The modified cellulose contains methyl formylimino groups which can both act as chelating group with metal ions and possess antimicrobial activities.
2. Materials and Methods
2.1. Materials
Cellulose (Loba), p-toluidine (Alfa Aesar), sodium metaperiodate (Sigma-Aldrich) was used as received. Copper and lead salts were procured from Sigma-Aldrich chemicals. All other chemicals and solvents used were either of analytical grades or purified according to standard procedures.
2.2. Metal Solutions
Metal salts CuSO4∙5H2O, Pb(NO3)2 were used for preparing stock solutions. Stock solutions of 1000 mg/L of standardized Cu2+, Pb2+ ions were prepared by dissolving the exact amount of the salts in double distilled water. The stock solutions were diluted to the required experimental concentration for the batch adsorption experiments.
2.3. Preparation of Chemically Modified Cellulose (DTD)
The oxidation reaction using sodium metaperiodate was carried out onto cellulose before the coupling process. Sodium metaperiodate oxidation is a highly specific reaction that cleaves the bond between C2-C3 of the glucosidic ring and converts into the 2,3-dialdehydic groups, following the mechanism of Malaprade reaction, without significant side reactions [16] [17] . Cellulose powder suspended in distilled water was mixed with sodium metaperiodate solution and stirred at room temperature in dark. After specified reaction time dialdehyde cellulose (DAC) formed was filtered and washed with deionised water until neutral conditions are achieved. During oxidation when the concentration of NaIO4 increases, the number of carbonyl groups per 100 glucose units also increases. To achieve approximately 30 carbonyl groups per 100 glucose units, the concentration of NaIO4 was kept at 0.04 M with a reaction time of 4 hr [18] [19] . The dialdehyde cellulose samples were dried under vacuum at room temperature to a constant weight. The C2-C3 dialdehyde groups of the cellulose are condensed with the aromatic amine p-toluidine to form the pendent Schiff bases in the cellulose chain. Typically 2 g of dialdehyde cellulose in double distilled water was stirred with 3 g of p-toluidine catalyzed by HCl at 70˚C for 3 h. The pale yellow color of dialdehyde cellulose slowly changes to reddish brown. At the end of the reaction period, chemically modified cellulose (DTD) was filtered, washed with hot water, ethanol and then dried under vacuum.
2.4. Characterization Methods
The heavy metal ion concentration of the solutions before and after equilibrium was determined by Atomic Absorption Spectrometer AA6300 (Shimadzu, Japan). The pH of solution was measured using a Hanna pH meter using glass electrode. FT-IR analysis was carried out using Shimadzu Spectrophotometer with KBr pellets. The SEM images of the DTD and metal loaded DTD were analyzed using a Leo Gemini1530 scanning electron microscope. Thermo gravimetric analysis (TGA) was recorded using a Perkin-Elmer analyzer in static air at a heating rate of 10˚C/min. Solid-state 13C CP-MAS NMR spectra were performed at 100.52 MHz on a Bruker AMX-200 spectrometer.
3. Adsorption Studies
Batch adsorption experiments were carried out by shaking the flasks using a horizontal bench shaker (Orbitek- Teqip-ACT/EQ/454) at 200 rpm. The experimental data obtained in batch studies were used to calculate the percentage removal of heavy metal ions by using mass balance equation.
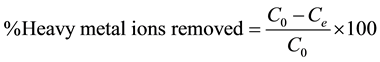 (1)
(1)
where C0 and Ce are initial and equilibrium final concentrations (mg/L) of the metal solutions respectively.
3.1. Effect of Solution pH on Adsorption
The effect of pH on the adsorption of DTD was carried out in the pH range of 2.0 to 10.0 at 30˚C. The samples were then shaken in a horizontal bench shaker at 200 rpm at a different solution pH for 60 min and then filtered through Whatman 42 filter paper. The filtrate was analyzed using AASC.
3.2. Effect of Adsorbent Dosage
Batch adsorption experiments were carried out at different adsorbent dosages of DTD from 5 to 25 mg at a pH of 6.0, by keeping the contact time and temperature constant.
3.3. Effect of Contact Time
Batch adsorption experiments were carried out by varying contact time of 20 - 120 min by keeping all other parameters constant.
3.4. Effect of Metal Ion Concentration
Initial metal ion concentrations were investigated in the range 50 to 300 mg/L at a pH 6.0.
3.5. Adsorption Isotherms and Kinetics
The adsorbed metal amount qe (mg/g) was determined by using the following mass balance relationship:
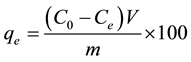 (2)
(2)
where V is the volume of the solution (L); and m is the adsorbent mass (g).
The amount of metal adsorbed at time (t), qt (mg/g), was calculated using the following equation:
 (3)
(3)
Ct―the concentration of metal solution at any time t (mg/L).
3.6. Antimicrobial Activity Test
The antimicrobial activities of modified cellulose DTD against Escherichia coli, Staphylococcus aureus and Enterococcus faecalis were examined using the agar well diffusion assay method. Diluted bacterial cultures were spread on sterile Mueller-Hinton agar plates, after which modified cellulose (50 µl) were placed on impregnated discs with 6 mm diameter for testing. The plates were incubated for 24 h at 37˚C under aerobic conditions and the diameter of the inhibition zones of each disc were measured and recorded [20] .
4. Results and Discussion
4.1. Characterization
Synthesis of chemically modified cellulose containing pendent methoxy benzalanilene group in the polymer chain is presented in Figure 1. The reaction involved a nucleophilic addition of the amino group to the cellulose dialdehyde carbonyl followed by an acid catalyzed dehydration.
The FTIR spectra of native cellulose (RA) and DTD are presented in Figure 2. The IR spectrum of cellulose exibited the main characterisict cellulose peaks. Absorbence at 3348 cm−1 (-OH strecthing), 2903 cm−1 (C-H stretching), 1664 cm−1 (C-C ring stretching and ?OH in plane bending), 1430 cm−1 (-CH2 bending), 1371 cm−1 (-CH bending) and 1058 cm−1 (C-O-C stretching) are in good agreement with the reported values. In the chemically modified cellulose the -OH stretching frequency appeared at 3345 cm−1 while that of the imine -CH stret- ching frequency appeared at 2903 cm−1. On oxidation with NaIO4 the pyranose ring is cleaved at C2-C3 and
![]()
Figure 1. Synthesis of chemically modified cellulose.
![]()
Figure 2. FT-IR spectra of native cellulose (RA), chemically mo- dified cellulose (DTD) and Pb2+ metal ion adsorbed modified cellulose DTD-Pb.
hence the C-C ring stretching frequency is not observed in the modified cellulose. A new peak at 1624 cm−1 is clearely due to the -N=CH- stretching frequency and the C-N stretching frequency appeared at 1517 cm−1 which supports the formation of methyl benzalaniline pendent groups in the chemically modified cellulose. To further establish the structure of chemically modified cellulose, solid state 13C-NMR spectroscopy was performed on both native and modified cellulose. 13C CP-MAS NMR spectra of natural cellulose and DTD are given in Figure 3 and Figure 4, respectively. In natural cellulose the C1-C6 signals appeared between δ values of 62.4 - 102.4 ppm. However, chemically modified cellulose DTD clearly showed the presence of pendent methyl benzalaniline groups in the cellulose chain (Figure 4). Apart from the usual signals, broad signals at 201.7 ppm are due to the azomethine carbon C7. Aromatic carbons C8 appeared at 175.9 ppm, C9 and C10 showed signal at 126.8 ppm and 151.1 ppm respectively. The C11 of CH3 groups appeared at 18.1 ppm.
4.2. Thermal Stability
TG traces of cellulose and DTD are shown in Figure 5. It is clear from the figure that natural cellulose has better thermal stability than chemically modified cellulose DTD. Initial decomposition temperatures of natural cellulose and DTD occur around 270˚C and 150˚C respectively. This indicates that DTD can be used as an adsorbent up to 150˚C. The decrease in thermal stability of DTD is probably due to the breaking up of pyranose ring at C2 and C3.
4.3. Metal Ions Uptake Studies
4.3.1. SEM Analysis
Scanning electron micrographs of DTD and metal loaded DTD are shown in Figures 6(a)-(c). Modified cellulose (DTD) surface is more irregular, rough and has open porous structure. The presence of pores in DTD suggests the possibility of the metal ions to be trapped and adsorbed onto the surface. These cavities are large enough to allow the metal ions to penetrate into the surface, and interact therein with the surface chelating groups. Surface morphology of the metal ion adsorbed DTD shows layers of metal ions on to porus surface. The
![]()
Figure 3. 13C CP-MAS NMR spectrum of native cellulose.
![]()
Figure 4. 13C CP-MAS NMR spectrum of DTD.
![]()
Figure 5. TG traces of natural cellulose and DTD.
![]()
Figure 6. Scanning electron micrographs of (a) DTD, (b) Pb2+ loaded DTD and (c) Cu2+ adsorbed DTD.
particle size was measured by particle size analyser and was found to be 448.3 nm. The value of the average particle size of the adsorbent provides more surface area for the removal of Cu2+ and Pb2+ ions from the aqueous media.
Figure 7 present the EDX spectra of DTD, Cu2+ treated DTD and Pb2+ treated DTD respectively. It is obvious that the DTD spectrum (Figure 7(DTD)) showed only carbon, oxygen and nitrogen peaks. However, Cu2+ and Pb2+ metal ion solution treated DTD (Figure 7(DTD-Cu) & Figure 7(DTD-Pb)) showed in addition to C, O and N clear peaks for Cu and Pb, thus confirming the metal ion uptake ability of DTD from aquous solutions.
4.3.2. Influence of Initial pH Value
The pH of the solution affects the adsorptive process through protonation and deprotonation of functional groups of the active sites of the adsorbent surface. Initial pH values were varied from 2 - 10 and the corresponding % adsorption is given in Figure 8. The adsorption capacity increases with increasing pH values up to 6 and decreases beyond this value. The present removal of metal ions exhibited a significant increase at pH 6 is attributed to the low H+ concentration which may compete with metal ions for coordination with active azomethine groups and subsequently lower the percent removal at low pH values. However at a lower pH value of 2 the percent removal of metal ions is >70% which suggests that the adsorbent is an effective one even at lower pH values which is an important requirement in the industrial applications.
4.3.3. Effect of Adsorbent Dose
Adsorbent dose was varied from 5 mg - 25 mg and the percent removal is shown in Figure 9. As the adsorbent dose increases the percent removal of metal ion increases and reaches a saturated value at the dosage of 20 mg. With increase in the adsorbent dose, the active sites available for coordination with metal ion increases and hence the percent removal increases. At 20 mg dosage maximum metal ions are removed and any further increase beyond this value, the percent removal remains constant. One of the interesting aspects of this study is that even at a low dosage of the adsorbent (10 mg) the percent removal is >70% suggesting the high concentra-
![]()
Figure 7. EDX spectra of DTD, Cu2+ treated DTD and Pb2+ treated DTD.
tion of the coordinating sites in the cellulose chain.
4.3.4. Adsorption Isotherms
The initial concentration of metal ions is an important factor for effective adsorption. The percentage removal of metal ions Cu(II), Pb(II) at different metal ion concentration (10 - 30 mg/L) were performed by keeping all other parameters constant. An adsorption isotherm can be used to characterize interaction of the adsorbates with adsorbents and optimizing the use of adsorbents. Adsorption isotherms are the basic requirements for designing any adsorption system. The distribution of metal ions between the liquid phase and the adsorbent is a measure of the position of equilibrium in the adsorption process and can be expressed by a series of isotherm models. The non-linear forms of the Langmuir [21] and Freundlich [22] adsorption isotherm models were used to evaluate the adsorption experimental data, using MATLAB R2009a. The non-linear forms of the Langmuir and Freun-
![]()
Figure 8. Effet of initial pH values on adsorption efficiency.
![]()
Figure 9. Effect of adsorbent dose on percentage removal.
dlich adsorption isotherms based on experimental observations are given in Figure 10.
4.3.5. Langmuir Isotherm Model
Langmuir isotherm models the monolayer coverage of adsorption surface. This model assumes that the maximum adsorption occurs at specific structurally homogeneous adsorption sites within the adsorbent and intermolecular forces decreases rapidly with the distance from adsorption surface. The non-linear equation of Langmuir isotherm model is expressed as;
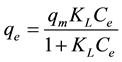 (4)
(4)
where Ce―the equilibrium concentration of the metal ions in the solution (mg/L), qe―the adsorbed value of the metal ion at equilibrium concentration (mg/g),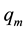 ―the maximum adsorption capacity (mg/g) and KL is the
―the maximum adsorption capacity (mg/g) and KL is the
Langmuir binding constant which is related to the energy of adsorption. The data obtained qe, KL and correlation coefficient (R2) values are shown in Table 1. The suitability of the adsorption process could be evaluated by calculating the separation factor constant (RL): RL > 1.0. unsuitable; RL = 1.0. linear; 0 < RL < 1, suitable; RL = 0, irreversible. The RL value can be calculated from the following equation.
![]() (5)
(5)
where b―the Langmuir adsorption equilibrium constant and Co―the initial metal ion concentration. The values of RL lie between 0.0081 and 0.0069, indicating suitability of the chemically modified cellulose as adsorbent for Cu(II) and Pb(II) from aqueous solution.
4.3.6. Freundlich Isotherm Model
The Freundlich isotherm model is related to multilayer adsorption, heterogeneous surface and interaction between adsorbed molecules. The nonlinear form of the Freundlich equation is given by
![]() (6)
(6)
where![]() ―the Freundlich constant ((mg/g) (L/mg)1/2) which represents the adsorption capacity and the strength of the adsorption bond and n―the heterogeneity factor which represents the bond distribution. The values of n, between 1 and 10 indicate favorable adsorption. In the present study, the n value of DTD adsorbent-metal ion system of Cu+2 (3.60) and Pb+2 (3.74) was found to be greater than 1, which indicates that the adsorption system is a favorable one and suggesting physical adsorption. According to the correlation coefficients, the experimental results exhibited the best fit with Langmuir model suggesting a mono layer homogeneous adsorption of the metal ions on to the chemically modified cellulose chelating DTD. The maximum adsorption capacity of Cu(II) and Pb(II) metal ions were found to be 157.3 and 153.5 mg/g, respectively, indicating the high potentiality of the adsorbent DTD.
―the Freundlich constant ((mg/g) (L/mg)1/2) which represents the adsorption capacity and the strength of the adsorption bond and n―the heterogeneity factor which represents the bond distribution. The values of n, between 1 and 10 indicate favorable adsorption. In the present study, the n value of DTD adsorbent-metal ion system of Cu+2 (3.60) and Pb+2 (3.74) was found to be greater than 1, which indicates that the adsorption system is a favorable one and suggesting physical adsorption. According to the correlation coefficients, the experimental results exhibited the best fit with Langmuir model suggesting a mono layer homogeneous adsorption of the metal ions on to the chemically modified cellulose chelating DTD. The maximum adsorption capacity of Cu(II) and Pb(II) metal ions were found to be 157.3 and 153.5 mg/g, respectively, indicating the high potentiality of the adsorbent DTD.
The adsorption capacity of the present DTD adsorbent has been compared with other adsorbents reported for copper and lead.
![]()
Table 1. Isotherm parameters for the adsorption of Pb2+ and Cu2+ over DTD.
When compared to other cellulose based adsorbents, the present chemically modified adsorbent is a highly efficient one for the removal of Pb2+ and Cu2+ ions from aqueous solution.
4.3.7. Kinetic Studies
Adsorption kinetics provides valuable information about the controlling mechanism of the adsorption process, rate of the adsorbate uptake and optimum operating conditions for the full-scale batch process. Adsorption kinetic models, such as the pseudo-first-order [36] and pseudo-second-order [37] models were used for the experimental data.
The effect of adsorption time on the removal of Pb2+ and Cu2+ by modified chelating DTD is presented in Figure 11. As can be seen, the removal showed a rapid rate for the first 60 min where the percent removal reached about 93% and 90% for Pb2+ and Cu2+ respectively.
For better understanding of the kinetic mechanism which governs the whole process, the experimental data obtained were fitted with the well known kinetic pseudo-first-order and pseudo-second-order models according to the following equations:
![]() (7)
(7)
where ki is the pseudo-first-order rate constant (min−1) of adsorption and qe and qt (mg/g) are the amounts of
![]()
Figure 11. Effect of contact time on percentage removal.
metal ion adsorbed at equilibrium and time t (min) respectively. The linear form of pseudo-second-order equation can be written as
![]() (8)
(8)
where k2 is the pseudo-second order rate constant of adsorption (g/(mg∙min)).
All the kinetic parameters of the removal process are summarized in Table 2. As can be seen the correlation coefficients (R2), the experimental data exhibited the best fit with the pseudo-second-order kinetic model. This indicates that the chemical coordination step is considered as the rate determining step without the involvement of a mass transfer in solution [38] [39] . The modified cellulose DTD is characterized for its high concentration of the pendent methyl benzalaniline groups, which contains azomethine moieties which can act as Lewis base on coordinating with heavy metal ions.
4.3.8. Adsorption Thermodynamics
The thermodynamic studies were conducted at various temperatures (300 - 335 K). It provides information on energetic changes that are associated with adsorption and adsorption process is spontaneous or not. The thermodynamic parameters for the adsorption including the Gibbs free energy change![]() , the enthalpy change
, the enthalpy change![]() , and the entropy change
, and the entropy change![]() , were calculated using the following equations,
, were calculated using the following equations,
![]() (9)
(9)
where ![]() -standard free energy changes (J/mol), R is the universal gas constant 8.314 J/mol/K and T is the absolute temperature, respectively.
-standard free energy changes (J/mol), R is the universal gas constant 8.314 J/mol/K and T is the absolute temperature, respectively. ![]() was calculated and
was calculated and ![]() and
and ![]() were obtained from the slope and intercept of the plot logK versus 1/T. The values of
were obtained from the slope and intercept of the plot logK versus 1/T. The values of![]() ,
, ![]() and
and ![]() for the adsorption onto DTD are given in Table 3. The negative values of
for the adsorption onto DTD are given in Table 3. The negative values of ![]() at all temperatures and
at all temperatures and ![]() indicates that metal-DTD adsorption system is a spontaneous and exothermic in nature. The change of Gibbs free energy decreased with increasing temperature indicates a more efficient adsorption has occurred at higher temperatures. The negative entropy
indicates that metal-DTD adsorption system is a spontaneous and exothermic in nature. The change of Gibbs free energy decreased with increasing temperature indicates a more efficient adsorption has occurred at higher temperatures. The negative entropy ![]() of adsorption confirms the decreased randomness at the solid-solution interface during adsorption which reflects the affinity of the adsorbent (DTD) toward metal ions.
of adsorption confirms the decreased randomness at the solid-solution interface during adsorption which reflects the affinity of the adsorbent (DTD) toward metal ions.
4.3.9. Desorption Studies
A most important problem is the recyclability of adsorbent and multi-usability. The regenerated adsorbent was used up to five adsorption-desorption cycles with Cu(II) and Pb(II) ions and the results are given in Figure 12.
![]()
Table 2. Kinetic parameters of removal of Cu and Pb onto DTD.
![]()
Table 3. Thermodynamic parameters for the adsorption of Cu2+ and Pb2+ onto DTD.
The acid solutions of 0.1 M of H2SO4, HCl and CH3COOH were used as eluents. On comparison 0.1 M solutions of H2SO4 and HCl has equal efficiency for the regeneration of Cu(II) and Pb(II) metal ions than 0.1 M solution of CH3COOH. This is due to the weak acidic nature of CH3COOH. Even after five cycles of adsorption- desorption, the efficiency of DTD did not exhibit a significant decrease. The polymeric resins with azomethine group are acid resistant and the adsorbents had a good potential for reuse. The results shows adsorption-desorp- tion process is a reversible process which specifies the formation of coordinate bond between the chelating groups and the metals.
4.4. Antimicrobial Activity
The modified cellulose DTD was tested by standard disc diffusion method with E. coli, E. faecalis and S. aures and the results are presented in Table 4. The results showed that the untreated cellulose is not active against the
![]()
Table 4. Antimicrobial activity of DTD.
selected microorganisms while the modified cellulose DTD showed activity against the same microorganisms. The significant antimicrobial activity of the modified cellulose is due to the presence of methyl benzalaniline pendent groups in the cellulose chain. These new modified cellulosic materials have good antimicrobial properties and can be used in many medicinal applications.
5. Conclusion
Removal of Pb2+, Cu2+ and antimicrobial activities was carried out using novel cellulose adsorbent bearing pendent methyl benzalaniline groups (DTD). Adsorbent capacity of the chemically modified cellulose towards Cu2+ and Pb2+ is 157.3 and 153.5 mg/g respectively. The adsorbent is an active one over a wide range of pH values. The new modified cellulose also shows promising antibacterial activity. The adsorption kinetic studies revealed that the adsorption process fits with the pseudo-second-order model while the adsorption isotherm studies confirmed that the experimental results follow the Langmuir model. Thermodynamic studies showed that the adsorption process is feasible and exothermic in nature.
NOTES
*Corresponding author.