Changes in Plasma Levels of Vasoactive Factors in Patients Undergoing Abdominal Surgery ()
1. Introduction
Patients undergoing abdominal surgery under general anesthesia present substantial risks of post-surgery venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE) [1] [2] . Without effective thromboprophylaxis, the reported incidences of DVT and proximal DVT, as assessed by fibrinogen uptake test, are 25% and 7%, respectively. Incidences of PE and fatal PE are 1.6% and 0.9%, respectively [2] . In Japan, VTE is a common post-surgery complication in patients undergoing major abdominal surgery [3] .
In recent years, it has been shown that vascular endothelial cells (VEC) not only are a barrier between blood and surrounding tissues, but also have important secreting functions. They can secrete vasoactive factors, such as endothelin-1 (ET-1), nitric oxide (NO) and protacyclin (PGI2), to regulate vascular tonus and to inhibit platelet aggregation, thus playing an important role in different cardiovascular diseases [4] . In addition, the platelet- derived thromboxane A2 (TXA2) is a potent agonist of platelet aggregation and a potent vascular smooth muscles cells constricting agent [5] . Imbalanced ratios of these factors may result in VTE after abdominal surgeries. Although it has been well established that surgery is one of the most common risk factors for VTE [6] [7] and that abdominal surgery leads to a hypercoagulation state (and thus to an increased DVT risk) [8] , little is known about the molecular mechanisms of VET after abdominal surgery and about temporal changes in vasoactive factors before and after surgery. Insights into these temporal changes might bring light into this pathogenesis and then may provide insights for a better thromboprophylaxis management before, during and after surgery by targeting specific changes in vasoactive factors.
The aim of the present study was to prospectively assess plasma levels of vasoactive factors (ET-1, NO, PGI2 and TXA2) in patients undergoing abdominal surgery under general anesthesia, using high performance liquid chromatography (HPLC) and radioimmunoassay (RIA). We also investigated the changes in vasoactive factors in the few days following surgery.
2. Materials and Methods
2.1. Subjects
This study was approved by the Hebei Medical University’s ethical review board and written informed consent was obtained from each subject before study procedures were undertaken. Inclusion criterion was the need for an abdominal surgery for an acute abdominal disease. Patients were excluded if: 1) they were taking indomethacin, nitroglycerin, cortisone or any kind of anticoagulant drug before surgery; and 2) if they experienced any complication after surgery.
A total of 30 cases (14 males, 16 females; average age 48 ± 11 years, ranging from 24 to 70) were included in final analyses, including: 24 cases of cholelithiasis and cholecystitis, 2 cases of peptic ulcer and 4 cases of portal vein hypertension. All cases underwent open abdominal surgery under general anesthesia.
2.2. Blood Sampling and Vasoactive Factors Assessment
Venous blood samples were obtained from each patient according to these time points: within 1 - 3 days after hospitalization (T1), at beginning of surgery and after anesthesia (T2) and during the first (T3), third (T4) and fifth day (T5) after operation. Plasma was obtained by centrifugation.
According to the manufacturers’ protocols, HPLC was used to measure plasma levels of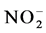 ,
, 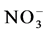 , ET-1 and 6-keto-PGF1α (Waters Corporation, Milford, MA, USA). TXB2 was measured using RIA (Immunological Institute, Bejing, China).
, ET-1 and 6-keto-PGF1α (Waters Corporation, Milford, MA, USA). TXB2 was measured using RIA (Immunological Institute, Bejing, China). 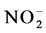 and
and 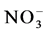 were used as surrogate markers for NO; 6-keto-PGF1α was a prostacyclin surrogate and TXB2 was a TXA2 surrogate.
were used as surrogate markers for NO; 6-keto-PGF1α was a prostacyclin surrogate and TXB2 was a TXA2 surrogate.
Ratios representing vasoconstriction/vasodilatation balance were assessed (NO/ET-1 and TXB2/6-keto- PGF1α).
2.3. Statistical Analysis
Statistical analysis was conducted using SAS version 8.0 (SAS Institute, Cary, NC, USA). Continuous variables were analyzed using descriptive statistics and means ± SD are presented. ANOVA was used to compare the different time points for each marker. Differences were considered statistically significant if p < 0.05.
3. Results
3.1. Changes in Levels of Vasoconstriction Factors (ET-1 and TXA2)
ET-1 plasma levels significantly increased at the beginning of surgery and after operation (T1 = 69.2 ± 10.7 vs. T2 = 82.4 ± 14.7 vs. T3 = 96.6 ± 22.8 pg/ml, p < 0.05), reaching its highest value at the 3rd day after operation (T4 = 105.3 ± 26.4 pg/ml), with ET-1 levels 1.5-fold higher than before surgery (p < 0.05). There was no difference in ET-1 levels following surgery and they remained high 5 days after surgery (Table 1).
TXB2 plasma levels before operation were significantly lower than following surgery (T1 = 30.6 ± 18.2 vs. T3 = 157.6 ± 121.78 pg/ml, p < 0.05). At the beginning of surgery, TXB2 levels were increased by 2.2-fold compared to baseline levels, but these changes were not statistically significant. TXB2 levels reached a peak on the 3rd following surgery, being increased by 5.8-fold compared to baseline (T1 = 30.6 ± 18.2 vs. T4 = 178.9 ± 21.4 pg/ml, p < 0.05) (Table 1).
3.2. Changes in Levels of Vasodilatory Factors (NO and PGI2)
Baseline NO levels were significantly higher than at the beginning of surgery and after (T1 = 2575 ± 550 vs. T2 = 19.22 ± 44 vs. T3 = 1692 ± 39 ng/ml, p < 0.05 for T1 vs. T2 and T3). NO levels were decreased by about 35% after surgery and remained stable 5 days post-surgery (Table 1).
Baseline 6-keto-PGF1α levels were significantly higher compared to the beginning of surgery and to the 1st day after operation (T1 = 180.5 ± 17.8 vs. T2 = 132.1 ± 32.6 vs. T3 = 110.9 ± 31.9 pg/ml, p < 0.05 for T1 vs. T2 and T3). Plasma 6-keto-PGF1α levels continued to decrease on the 3rd and 5th day post-surgery (p < 0.05) (Table 1).
3.3. Changes in Vasoactive Ratios (NO/ET-1 and TXB2/6-Keto-PGF1α)
Mean baseline value of the NO/ET-1 ratio was higher compared to values obtained before surgery and in the days following surgery (T1 = 37.7 ± 9.0 vs. T2 = 23.8 ± 6.0 vs. T3 = 18.2 ± 5.0, p < 0.05). NO/ET-1 ratio remained low in the days following surgery (Table 2).
Post-surgery ratio of TXA2/6-keto-PGF1α was significantly higher compared to baseline and to the beginning of surgery (T1 = 0.18 ± 0.10 vs. T2 = 0.51 ± 0.35 vs. T3 = 1.39 ± 0.96, p < 0.05 for T1 and T2 vs. T3). TXA2/6- keto-PGF1α ratio reached a peak on the 3rd day following surgery, being increased by 11.1 fold compared to baseline (Table 2).
4. Discussion
In the present study, plasma levels of ET-1, NO, PGI2 and TXA2 were assessed at five different time points before and after abdominal surgery. Results showed that vasodilatory factors (NO and PGI2) significantly decreased after operation, whereas vasoconstricting factors (ET-1 and TXA2) significantly increased. The imbal-
![]()
Table 1. Changes in plasma levels of ET-1, TXB2,  /
/ and 6-keto-PGI1αin patients undergoing abdominal surgery (means ± SD, n = 30).
and 6-keto-PGI1αin patients undergoing abdominal surgery (means ± SD, n = 30).
T1 = 1 - 3 days after hospitalization; T2 = at surgery beginning, after anesthesia; T3 = first day after surgery; T4 = third day after surgery; T5 = fifth day after surgery; ET = endothelin; NO = nitric oxide; PGI2 = prostacyclin; TXB2 = thromboxane B2; *, **, *** = statistical groupings, p < 0.05 between groups.
![]()
Table 2. Changes in plasma NO/ET-1 and TXA2/PGI2 ratios in patients undergoing abdominal surgery (means ± SD, n = 30).
T1 = 1 - 3 days after hospitalization; T2 = at surgery beginning, after anesthesia; T3 = first day after surgery; T4 = third day after surgery; T5 = fifth day after surgery; ET = endothelin; NO = nitric oxide; PGI2 = prostacyclin; TXB2 = thromboxane B2; *, **, *** = statistical groupings, p < 0.05 between groups.
ance between vasodilators and vasoconstrictors may be helpful in providing information about VTE Onset in patients undergoing abdominal surgery. A better understanding of surgery-related VTE might provide better prophylactic management in these patients.
ET was isolated in 1988 by Yanagisawa et al. from pig aortic VEC. It is a short 21-amino acid peptide and a potent vasoconstrictor [9] -[12] . ET has three isomers, namely ET-1, ET-2 and ET-3, ET-1 having the strongest vasoconstriction capabilities. ET’s vasoconstricting action results mainly from increasing concentration of calcium ion (Ca2+) in smooth muscle cell lining the artery. Activation of ET receptors by ET results in immediate transport of Ca2+ in the sarcoplasmic reticulum, greatly increasing Ca2+ levels and finally resulting in vasoconstriction. The present study showed that ET-1 levels were significantly higher post-surgery compared to baseline. These results are in agreement with other studies [9] [13] [14] . The reason for this increase in ET-1 levels may be that as the vena cava is injured, ET-1 release increases in circulation as a direct injury’s effect [15] ; a combined decrease in NO release is also observed, and NO is an ET-1 antagonist [16] . It may also be hypothesized that any vein injury during the course of a surgery increases ET levels. ET is a potent vasoconstrictor, making the blood flow slower and contributing to blood coagulation in the area of surgery, thus increasing the probability for VTE onset.
NO is secreted by the VEC and is the most important known endogenous vasodilator [17] . NO protects vessel walls by inhibiting platelet aggregation, [18] - [21] cell adhesion [22] and secretion of cell-recruiting factors [23] . In both vascular smooth muscle cells and platelets, these effects of NO are known to be mediated by cGMP. NO is synthesized from L-arginine by the action of the nitric oxide synthase (NOS). NO is highly reactive and thus unstable, and its half-life is very short, between 1 and 5 seconds. NO reacts quickly with water, O2 and various oxygen ions existing in circulation, forming 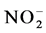 and
and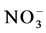 . The sum of the plasma levels of
. The sum of the plasma levels of 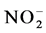 and of
and of 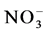 provides an indirect measurement of NO levels in the body. NO increases cellular levels of cGMP by activating soluble GMP enzyme in the smooth muscle cells in vascular walls, thus increasing cGMP levels. Then, cGMP activates an enzyme cascade, relaxing smooth muscle cell [24] [25] . Also, via cGMP-dependent mechanisms, NO inhibits platelet aggregation and adhesion [26] . The present study suggests that the sum of NO2- and NO3-levels at decreased significantly after surgery, compared to baseline. This decrease may be the result of a decrease in NO secretion and/or of an increase in the NO antagonistic agent, ET-1, as a result of injured vessels during operation [27] [28] . Decreased NO levels decreases vasodilation and anticoagulation of platelets, which contributes to vasoconstriction, platelets adhesion and convergence and, finally, to thrombosis.
provides an indirect measurement of NO levels in the body. NO increases cellular levels of cGMP by activating soluble GMP enzyme in the smooth muscle cells in vascular walls, thus increasing cGMP levels. Then, cGMP activates an enzyme cascade, relaxing smooth muscle cell [24] [25] . Also, via cGMP-dependent mechanisms, NO inhibits platelet aggregation and adhesion [26] . The present study suggests that the sum of NO2- and NO3-levels at decreased significantly after surgery, compared to baseline. This decrease may be the result of a decrease in NO secretion and/or of an increase in the NO antagonistic agent, ET-1, as a result of injured vessels during operation [27] [28] . Decreased NO levels decreases vasodilation and anticoagulation of platelets, which contributes to vasoconstriction, platelets adhesion and convergence and, finally, to thrombosis.
PGI2 is a product of arachidonic acid metabolism generated by the vessel wall of all mammalian species studied, including human. After a half-life of 2 - 3 minutes, it returns to the form of inactive 6-keto-PGI1α. PGI2 relaxes smooth muscle cells by increasing the amounts of cellular cAMP. By increasing cAMP levels in platelets, PGI2 also inhibits platelet adhesion and convergence [29] . PGI2 decreases ET production and weakens ET vasoconstrictive effects [28] [30] . In addition, PGI2 decreases the production of its antagonist agent, TXA2. The present study showed that postoperative PGI2 levels are lower than at baseline. This decrease may be the result of a vascular endothelium injury during operation, causing a decrease in PGI2 synthesis [31] and an increase in the secretion of the antagonist agent TXA2. PGI2 and TXA2 are produced from the same precursor (arachidonic acid). VEC can induce formation of PGI2 using PGI2 synthase, but platelets only synthesize TXA2 using TXA2 synthase, the two being in a homeostasis state. A decrease in PGI2 causes an increase in TXB2, then promoting platelet adhesion and convergence, increasing thrombosis.
TXA2 is mainly produced in platelets and is a potent vasoconstrictor and platelets aggregating agent [31] [32] . It can also be synthesized by other cell types [33] [34] . Inhibition of TXA2 synthesis decreased ischemic events in clinical trials, suggesting an important role for TXA2 in in vivo regulation of hemostasis and thrombosis [35] . With a half life of 30 seconds, it returns quickly to inactive TXB2. The present study suggest that postoperative TXB2 levels were higher than before surgery and may be the result of injured vascular wall and of activated platelets, the latter resulting in the release of great amounts of TXA2 and in the decrease of NO and PGI2 synthesis. Decrease in PGI2 levels breaks the homoeostasis between TXA2 and PGI2, resulting in TXA2 increase. Increased TXA2 levels thus promote coagulation processes and thrombosis.
NO and ET are the most important local vasoactive factors. They have inverse bioactivities and their combined effect finely regulates vascular tonus, growth and repair. In case of vascular diseases, the breaking in their homeostasis may contribute to further exacerbate the disease [35] . Some studies indicated that NO is an endothelial-derived thrombus regulator, decreasing platelet adhesion on VEC surface [36] . The present study showed that the NO/ET-1 ratio decreased significantly after operation, indicating that the platelets are in an activted state.
Both PGI2 and TXA2 are metabolites of arachidonic acid. PGI2 is produced by vascular endothelium by the PGI2 synthase, while TXA2 is produced by blood platelets by the TXA2 synthase. PGI2 and TXA2 are the opposite poles of a homeostatic mechanism for the regulation of platelet aggregability in vivo. Thus, a balance between PGI2 and TXA2 production regulates the circulation hemostatic state and has been the target of intensive investigation on the role of PGI2 in vascular disease processes. It has been suggested that a number of vascular diseases are related to an imbalance in the PGI2 and TXA2 system, including arterial and venous thrombosis, atherosclerosis and diabetes. A possible explanation for diseases with a tendency for thrombosis is that PGI2 production is reduced and/or TXA2 production is increased [37] . It has been reported that reduction of PGI2 levels and the increase of the PGI2/TXA2 ratio observed after ethanol perfusion in umbilical veins may cause vascular disruption in the umbilical-placental circulation [38] . In the present study, PGI2 production was significantly decreased, but TXA2 significantly increased after operation, resulting in a decreased PGI2/TXA2 ratio, promoting VTE onset.
The present study suffers from a number of limitations. First, only 30 patients were included; however, this relatively small number of patients did not preclude us to observe significant changes in the levels of vasoactive factors. Also, patients underwent abdominal surgery for different diseases, mostly hepatic, but also gastric and vascular. But since blood vessels are often injured in the course of abdominal organ surgery, it is difficult to assess if VTE incidence in abdominal organ surgery is different from pure abdominal vascular surgery. We must assume that blood vessels are injured in all cases and that vasoactive factors are affected in the same way. Some studies report that an endoscopic approach for abdominal surgery reduces VTE risk [39] - [41] . However, we were not able to make any conclusion on this point in the present study. Values from the T2 time point (before the beginning of surgery but after anesthesia) must be taken with care, since changes in vasoactive factors are observed before any surgical procedure is undertaken. These changes may be due to deterioration in health condition in some patients between their hospital admission and their surgery. These changes might also be due to the anesthesia per se. Indeed, an animal study suggested that increases in TXB2 during surgery were due to the anesthesia, not to the surgery [42] , which would be consistent to the observed increase in TXB2 levels at T2 time point, even if it did not reach statistical significance. Finally, we recruited patients in a wide age range. Even if the present study was not designed to assess VTE incidence, it has been shown that age is an important VTE predictor and might have an impact on vasoactive levels [43] .
Level I evidences support the use of thromboprophylaxis in patients undergoing major abdominal surgery [44] . However, a study reported that patients admitted with an abdominal condition with high VTE risk did not all receive adequate prophylaxis [45] . Results from the present study show that surgery induces important pro- thrombosis mechanisms in patients undergoing abdominal surgery. Our results thus support the concept of using adequate thromboprophylaxis in these patients.
5. Conclusion
In conclusion, the present study clearly shows that two groups of endogenous vasoactive factors (NO/ET-1 and PGI2/TXA2) are significantly affected by abdominal surgery. Although the precise mechanisms remain unclear, these changes may play a role in VTE pathogenesis. These findings may contribute to the improvement in surgical techniques, thromboprophylaxis use and advances in perioperative and postoperative care to reduce the risk for VTE.
Abbreviation
VTE = Venous Thromboembolism
DVT = Deep Venous Thrombosis
VEC = Vascular Endothelial Cell
ET-1 = Endothelin-1
NO = Nitric Oxide
PGI2 = Prostacyclin
TXA2 = Thromboxane A2
TXB2 = Thromboxane B2
NOTES
*Corresponding author.
#These two authors contribute equally to this work.