Groundwater Level Effect on Redox Potential, on Cadmium Uptake and Yield of Soybean ()
1. Introduction
Mining, manufacturing and the use of synthetic products (e.g. pesticides, paints, batteries, industrial waste, and land application of industrial or domestic sludge) can result in toxic metal contamination of urban and agricultural soils. These metals also occur naturally, but rarely at toxic levels [1] . However, when the amounts exceed a certain level due to pollutants brought from outside, soil contamination occurs and agricultural products become contaminated. Excess of this metal accumulation in soils is toxic to humans and other animals. Exposure to toxic metals is normally chronic (exposure over a long period of time), due to food chain transfer. It poses a great threat to the environment and human health worldwide due to the persistent nature and toxicity and their accumulation in the food chain [2] . Among toxic metals, Cadmium (Cd) has been recognized as one of the most inimical metals in Japan for Itai-itai disease in Toyama prefecture [3] . Cd occurs naturally in the environment in its inorganic form as a result of volcanic emissions and weathering of rocks. Foodstuffs are the main source of Cd exposure for the non-smoking general population. Cd absorption after dietary exposure in humans is relatively low (3% - 5%) but it is efficiently retained in the kidney and liver in the human body, with a very long biological half-life ranging from 10 to 30 years. Cd can also cause bone demineralization, either through direct bone damage or through indirectly as a result of renal dysfunction.
Some methods have been proposed to minimize absorption of Cd by plants from soil, e.g.: 1) soil dressing; 2) chemical cleaning of soil; 3) phytoremediation and 4) use of different varieties and rootstock. However, most of these methods are very costly and time-consuming for effective minimization and have some residual effects also. The redox condition of soil root zone controlled by groundwater level management can be another effective method. Soil is a dynamic and complex system, and any changes in intrinsic physico-chemical properties of toxic metal would surely change the fate of heavy metals in a soil system. Theoretical prediction of toxic metal equilibrium as affected by pH and redox potential has been studied [4] . The influence of redox potential on arsenic speciation and solubility in a contaminated soil was also reported [5] . Furthermore, the environmental and economic benefits of groundwater level management through reduced pollution and increased yields have been documented [6] [7] .
Soybean is a globally important crop providing oil and protein and fastest expanding crops in terms of both calorie and protein intake. Soybeans are a versatile food and one of the central ingredients of Japanese cuisine. They continue the basis of many distinct Japanese flavors, such as soy sauce, miso and tofu, and are processed into countless culinary products. A large-scale domestic agricultural products survey in Japan showed that, 16.7% of soybean contains over 0.2 mg/kg and 49.4% contains over 0.1 mg/kg Cd [8] . However, Codex Alimentarius Commission [9] proposed that the maximum allowable limit of Cd in soybean is 0.2 mg/kg. So, it is essential to minimize the uptake of Cd from polluted soils and increase the seed yield of soybean. As we mentioned previously, the redox condition of soil layer is important and useful for both avoiding heavy metal uptake and increasing soybean yield. The objective of the study is to find out the relation between groundwater level on redox potential on cadmium uptake and soybean yield. This will help to minimize uptake of toxic metals especially Cd in different grain crops.
2. Materials and Methods
2.1. Soil Properties
The experiment was conducted in the greenhouse of Hirosaki University located in Aomori prefecture in Japan. In this study, we used two types of soil collected from two different areas. Cd contaminated soil was sampled from Eastern area of Japan of which contains 1.57 mg·kg−1 Cd, and non-contaminated soil was collected from Kanagi farm, Aomori prefecture. Gravel was collected from near the Iwaki Mountain area. The physical and chemical properties of the soil (Table 1) were measured by the standard methods of analysis [10] . Before setting up the apparatus, first, weeds, small stones and plant debris were removed from the collected soil. After cleaning, soil balls were made of 12 - 15 cm in diameter and dried. Then the soil balls were broken with hammer and the broken clods were sieved in a 4.75 mm opening mesh for making sure that the similar size of the clods could be used in the experiment.
2.2. Experimental design
We made three plastic containers (61 cm × 41 cm × 63 cm) and all of them were filled with gravel (small stone crust; bottom 14 cm), non-polluted soil (15 cm) and polluted soil (top 25 cm) shown in Figure 1. After filling
![]()
Table 1. Soil physical and chemical properties.
Note: soil texture is based on the International Soil Society classification. SL: Sandy Loam; SCL: Sandy Clay Loam. Gravel diameter size 2 - 4 mm.
the soils, Eh and temperature sensors were set up every soil layer of the three boxes. Three different groundwater level treatments were conducted for the three containers: 1) 10 cm (GW-10), 2) 40 cm (GW-40) and 3) 40 cm for the first 50 days, 10 cm for the next 50 days and 40 cm for the last 20 days (GW-40-10-40) (Figure 2).
2.3. Cultivation procedure
In this study Ryuho variety was grown which was recommended for Akita region. After filling the soils in the containers, groundwater level was set up 10 cm for all treatments, 12 hours before seeding to moist the soil. At the time of seeding the soil water content was around 0.42 g·g−1. 12 hours after seeding, the groundwater level was set up as per treatment. At 1st June (2013), 5 - 6 soybean seeds were sown about 2 - 3 cm depth of soil per pit in every box and after 15 days (complete germination) the healthy two remained and others were uprooted. Groundwater level was maintained by Mariotte bottle arrangement which supplied water through the bottom of the plastic box. Groundwater level was measured every day in groundwater level check pipe (4 numbers in each box). Fertilizers (NPK) were applied as per recommended rate for Rhyho variety. In every 4 days, 2 Liter water was supplied in each box that is equal to rainfall of 4 days in Aomori prefecture. Insecticides were periodically sprayed as well.
2.4. Measurement procedure
Oxidation-reduction potential (ORP) of each soil layer was measured by electrometrically using ORP meter (Central Science Co., Ltd., model UC-203) from vegetative to harvesting period. The plant height, number of branches and Soil Plant Analyzer Development (SPAD) of soybean plants were observed continuously from first trifoliate leaf stage. The latest fully developed trifoliate leaf was used for SPAD measurement. After harvesting the seeds, stems and roots were extracted with sulfuric acid and then Cd concentration of each parts of soybeans were measured with Atomic Absorption Spectrophotometer (Model Z-2000, Hitachi Corporation) as described by the Ministry of Agriculture, Forestry and Fisheries of Japan [11] .
3. Statistical analysis
Tukey-Kramer test was performed with 5% significance level to find out difference among Cd uptake in seed, stem and root of soybean plants.
4. Results and discussion
4.1. Redox Potential (Eh) of soil layers
Oxidation-Reduction Potential (ORP) condition is known as a redox potential (Eh). The Eh value in soil higher than 300 mV is an indicator of oxidation condition and lower than 300 mV indicates reduction condition [12] . Eh value was measured less than 300 mV in below 10 cm (from soil surface) in GW-10 treatment for saturated condition by high groundwater level (Figure 3). Only Eh values of 5 cm and 7.5 cm soil depth were higher than 300 mV that means it was in oxidation condition. So, we can say that in GW-10 treatment, reduction condition was occurred in soil layer below 10 cm from the soil surface. In case of GW-40 treatment, Eh value was measured over 300 mV (about 700 mV) for all layer of soil except 40 cm depth Eh value (Figure 4). It represents that oxidation condition occurred from the surface to 40 cm depth in GW-40 treatment. And in GW-40-10-40, first 50 days Eh value of soil (0 - 40 cm) layer was measured about 600 mV, next 50 days below 300 mV and last 20 days Eh value was increasing towards over 300 mV (Figure 5). It showed that vegetative period (first 50 days) it was in oxidation condition and reproductive period (next 50 days) was in reduction condition and last 20 days it was again in oxidation condition.
![]()
Figure 1. Layout of the plastic container used in the experiment.
![]()
Figure 2. Outline of groundwater level treatments.
![]()
Figure 5. Eh value of GW-40-10-40 treatment.
4.2. Cadmium concentration in soybean plants
Cadmium concentration in seeds were significantly higher in GW-40-10-40 (1.16 ± 0.13 mg·kg−1) than other two treatments, 0.96 ± 0.07 mg·kg−1 and 0.8 ± 0.12 mg·kg−1 in GW-10 and GW-40 treatments, respectively (Figure 6). This can be explained with the relationship of Cd solubility and redox potential. The proportion of soluble Cd decreased abruptly with the decrease of soil redox potential [12] . In our experiment, the redox condition of the soil layer from 10 to 40 cm depth was oxidation in GW-40, reduction in GW-10 and reproductive stage, in which soybean needs a lot of water, in GW-40-10-40. Metal solubility increased as redox potential decreased [13] . However, opposite results has also been reported in our first experiment in 2012 [14] . From this two different types of data observed in two consecutive years, we can say that only redox potential is not only important factor but also pH of soil layer have the affect for heavy metal uptake in plants [15] . Among all parameters that might affect transport of heavy metals in soil systems, redox potential is also an important parameter, although pH is usually considered to be a prime importance [13] . As for stem Cd concentration, GW-40 treatment had significantly higher Cd than GW-10 and GW-40-10-40 treatment (Figure 7). Stem Cd concentration was (1.7 ± 0.2 mg·kg−1), (1.3 ± 0.13 mg·kg−1) and (0.96 ± 0.07 mg·kg−1) in GW-40, GW40-10-40 cm and GW-40 treatment, respectively. As for root Cd concentration, there was no significant difference in those three treatments (Figure 8). Cd concentration in the soybean plants was different among three treatments due to difference of oxidation and reduction condition of soil layer. Previous research showed that accumulation of Cd was significantly reduced with diminishing Eh in reductive conditions formed by flooding rice fields [16] . Another researches showed that in soil reduction condition, the presence of sulphate (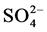 ) ion can be converted to sulfide (
) ion can be converted to sulfide (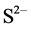 ) ion which reacts with Cd and Cu and produces relatively insoluble Cadmium and Copper sulfide [17] . Concentration of Cd in soybean root was higher than the stem and seed. It might be because the intercellular spaces freely accessible to ions of root tissues were much greater than those of other parts. Some researchers also showed that accumulation of Cd in roots was higher than that in any other parts in plants [18] [19] which are similar to this study.
) ion which reacts with Cd and Cu and produces relatively insoluble Cadmium and Copper sulfide [17] . Concentration of Cd in soybean root was higher than the stem and seed. It might be because the intercellular spaces freely accessible to ions of root tissues were much greater than those of other parts. Some researchers also showed that accumulation of Cd in roots was higher than that in any other parts in plants [18] [19] which are similar to this study.
4.3. Soybean Yield components
While the average main stem length of GW-40 (99.2 ± 3.8) was significantly higher than that of GW-10 (90.9 ±
![]()
Figure 6. Seed Cd concentration of soybean (a, b and c indicate significant difference).
3.2) treatment (Table 2), that of GW-40 and GW-40-10-40 (95.9 ± 4.3) was not significantly different. It means that the main stem length was higher in lower groundwater level than high groundwater level. Main stem length
![]()
Table 2. Soybean yield components of three groundwater level treatments.
Note: Tukey test was performed at 5% level; letters indicates significant difference (n = 8 for GW-10 cm & GW-40-10-40 cm; n = 7 for GW-40 cm after ±value is standard deviation value).
was higher with lower water table treatments within three groundwater (15 cm, 40 cm and 70 cm) treatments [20] . Branch number and good seed weight was also measured the highest in GW-40 treatment among three treatments. Vegetative growth was inhibited following shorter stem [21] by the effect of excess moisture which is supported to the present study. No significant difference was among stem diameter, branch number, and total good seed weight in these 3 treatments.
100 seed weight of GW-40-10-40 treatment was significantly higher than that of GW-10 and it tended to be higher than that of GW-40 (Table 2). We got the higher yield in GW-40 treatment than in GW-10 and GW-40-10-40 treatments, and the lowest was in GW-40-10-40 or changing groundwater level. Effects of groundwater level on soybean yield have been reported by many researchers. Previous researches found 100 seed weight decreases for excess moisture in the soil at the ripening stage [22] . The optimum groundwater table depth for getting high yield differs from researcher to researcher; that is 20 cm [23] , 40 cm [24] [25] , 30 cm to 50 cm [26] and 50 cm [27] . Another research showed that, 10 cm groundwater level gave high soybean yield in the saturated soil culture at semi-arid tropical region in Australia [28] . In Japan most of the high yielded records were obtained on a drained paddy field where the water table maintained at around 50 cm depth from the soil surface. A stable groundwater level is important for obtaining higher yield of soybean and fluctuation of groundwater level reduced the yield [29] [30] . This phenomenon was also observed in our research.
4.4. SPAD (Soil Plant Analyzer Development)
The SPAD meter (KONICA MINOLTA SPAD-502) provides a simple, quick, portable and non-destructive method for estimating leaf SPAD value from which leaf chlorophyll content can be known by calculation. In SPAD value (Figure 9), we can clearly see the effect of groundwater level on SPAD value. In GW-10 treatment, vegetative stage (up to 50 days after sowing) the leaf chlorophyll content was lower (about 31) than that in GW-40 and GW-40-10-40 treatment (about 46) due to excess soil moisture. However, after 50 days it recovers as high as that of the other two treatments. We know the soybean plant has adaptation ability in high range of groundwater level. In previous study [28] reported that temporary chlorosis occurred when saturated-soil-water culture was given to soybean plants and that caused a reduction of leaf nitrogen content [31] . Reference [20] showed that chlorophyll contents decrease as well as seed yield decrease when groundwater level was high (15 cm) compared to lower groundwater level (40 cm and 70 cm).
4.5. Root distribution
While, over 90% of the total root weight was observed from the surface to 10 cm depth in GW-10 and GW- 40-10-40 treatments, some roots penetrated into 40 cm from the surface in GW-40 (Figure 10). There were two peaks of root distribution in the soil profile; the soil surface and a little above the water table [32] [33] which was similar to this experiment. Different root structure was also observed different among the three treatments; primary root system in GW-40 and secondary root (mat like structure) system in GW-10 and GW-40-10-40 treatment.
5. Conclusion
Accumulation of Cd by soybean plant was significantly affected by redox condition of soil layer which can be controlled by groundwater level. Among all parameters, which affect Cd uptake in soybean plants, redox potential should be one of the crucial things. A stable groundwater level is important for obtaining higher yield and
![]()
Figure 9. SPAD value of three groundwater level treatments.
![]()
Figure 10. Root distribution of three groundwater level treatments.
groundwater fluctuation can reduce the yield. This can help in agronomic management in crop cultivation for minimization of Cd and other toxic metals uptake from polluted soils; it can also improve the management of polluted soil especially in developing countries since groundwater level management is relatively easy and does not cost so much. We need more research about groundwater level with other factors in Cd uptake in soybean (especially reproductive stage) and other crops with different Cd concentrations of soil.