1. Introduction
The word allelopathy is derived from two separate Greek words: allelon which means “of each other”, and pathos means “to suffer” [1] . It refers to the chemical inhibition of one plant species by another. Allelopathy is biological phenomenon that maintains balance among the various plant communities and in natural ecosystems. Some plants release chemicals referred to allelochemicals that often affect the growth, development, survival and reproduction of neighboring plants [2] [3] . Allelochemicals are mostly plant secondary metabolites (PSM) of either acetate or shikimate metabolic pathways. These chemicals include long-chain fatty acids, phenolic compounds, alkaloids, steroids, and derivatives of coumarin, quinines, flavonoids, tannins, terpenes and water soluble organic acids usually with a broad spectrum of activity [4] .
The toxicity of these compounds is a function of concentration, flux rate, age and physiological stage of the plant, climate and environment. These PSMs may be found or extracted from all parts of the plant including roots, rhizomes, stems, leaves, flowers and seeds. They are in vacuoles of cells as glycosides, polymers and/or crystals so that they do not affect the plant producing them [5] [6] . Allelochemicals are released into the environment as leachates, volatilized compounds, exudates, and decomposed plant material [2] [7] .
The mode of action of allelochemicals may be direct or indirect. Direct action includes effects on plant growth and metabolism and indirect effects are within purview of alteration of soil properties, soil nutrition and changes in beneficial and harmful soil microbial populations. Allelochemicals may interfere with various important processes such as seed germination, photosynthesis, respiration, water relations, ion uptake and growth, cell ultrastructure and oxidative stress [1] [8] .
Some microorganisms and different chemical processes in the soil environment may inactivate these PSMs or break them into new toxic allelochemicals [9] [10] . Tannins are reported to be microbial inhibitors while phenolics were growth substrates [11] . Some phenolics in forests soils were used by fungi and cellulose hydrolyzers whereas growth of heterotrophic bacteria was inhibited [12] . Thus alleloochemicals do not only affect changes in plant communities but also microbial populations in the soil.
The plant species, Conocarpus lancifolius, was introduced into Kuwait as part of the “Greenery Program” and has become the dominant perennial ornamental plant. It is a fast growing plant and produces large amounts of biomass particularly in summer (≥40˚C) with drip irrigation. The areas directly beneath or within the vicinity of C. lancifolius do not promote or support growth of other plants. This observation is within the purview of alleolopathy. Currently, no serious herbivores attack the species and it appears to be totally devoid of plant pathogens. The absence of pests probably enables this plant species to reallocate resources previously dedicated to its defense, to growth and reproduction. These observations indicate that the species may have either resistance or tolerance mechanisms that are operative when it is attacked by herbivores or pathogens. The plant may have biochemical reactions that produce compounds that are toxic to the growth and development of pests particularly pathogens. Some of these inhibitory compounds may be preexisting or induced as plant secondary metabolites (PSM) or allelochemicals.
There are no allelopathetic studies on C. lancifolius. Preliminary analysis of the leaf extract in our laboratory showed the presence of phenolics, tannins, flavonoids, steroids, terpenoids. Some of these chemicals could be allelochemicals but this has not been investigated.
The objectives of this research were: 1) to evaluate the allelopathic effects of C. lancifolius leaf extract on the growth and physiological traits in corn (Zea mays) and bean (Vigna sinensis); 2) to determine the chemical constituents of leaf extract and 3) the effects of the leaf extract on some soil borne fungi in vitro.
2. Materials and Methods
2.1. Donor Plant Material and Preparation of Leaf Extract
Fresh leaves of C. lancifolius were collected and rinsed thoroughly with sterile distilled water and dried at 30˚C for two weeks in an oven. Aqueous extracts of 2.5%, 5.0% and 7.5% (w/v) were prepared with ground leaf tissue in deionized distilled water at 25˚C. The mixtures were filtered through a double layer muslin cloth and with whatman no:#1 filter paper and extracts were stored in the dark kept at 4˚C as stock solution for further studies.
2.2. Test or Target Plants
Corn (Zea mays) and bean (Vigna sinensis) seeds purchased locally were used as test or target plants in the experiments. Germination tests were performed on each seed lot to ensure viability was above 95%. Prior to each experiment seeds were surfaced sterilized with 90% ethanol for 2 min and in 2.0% sodium hypochloride (NaOCl) for 5 min, then thoroughly rinsed with sterile distilled water.
2.3. Laboratory Seed Bioassay
The alleolopathic effect of the leaf extract on seed germination was performed with fifty corn and bean seeds of approximately the same size in Petri dishes lined with a double layer of sterile filter paper moistened with 5 ml of each concentration of leaf extract. Distilled water was applied as the control. All treatments were replicated five times and placed in a completely randomized design under cool fluorescent light at 25˚C. Seed germination was determined daily; and hypocotyl and radicle lengths were determined 7 days after treatment.
2.4. Seed Water Uptake or Imbibition
Ten corn and bean seeds (approx. 300 mg) were placed in beakers with 100 ml of aqueous leaf extract (2.5%, 5.0%, and 7.5% w/v) of C. lancifolius for 6, 24, 48 and 72 h. At these time intervals the seeds for each treatment were taken out, blotted on folds of sterile filter paper and weighed. Sterile distilled and deionized water was used a control. The osmotic or solute potential of leaf extracts were measured at 22˚C ± 1˚C, with a Wescor Vapor Pressure Osmometer 5520 (Wescor Inc. Logan Utah, USA) using 0.1 ml aliquot of the each extract. Osmolality measurements were converted to osmotic pressures using Van’t Hoff’s equation: Osmotic pressure (MPa) = RTc, where RT = 2.454 kg∙MPa∙mol−1 at 22˚C, and c = osmolality in mol∙kg−1. The water imbibed was determined by subtracting the initial seed weight from the daily seed weight after the time intervals. The experiment was repeated and each treatment was replicated four times.
2.5. GC-MS Analysis of Leaf Extract
GC MS analysis of extract for phenolics and steroids [13] , was performed using a Thermo Finnigan GC MS equipped with a HP-5MS fused silica capillary column (30 m × 0.25 mm i.d., film thickness 0.25 µm) an electron ionization system with ionization energy 70e was used. The identification of compounds was based on matching the mass spectral and retention times with those of the NIST 21 and Wiley 229 libraries spectral databases.
2.6. Allelopathic Effects of Leaf Tissue as Soil Amendment
Dried ground leaf tissue (0%, 2.5%, 5.0%, 7.5%, w/w) was mixed with sterilized local sandy-loam soil in plastic pots to assess the allelopathic effects of leaf tissue. Corn and bean seeds were sown in each pot with soil at field capacity. Every two days the soil was sprayed with deionized, distilled and sterilized water to maintain the soil at field capacity. The treatments were randomized in the greenhouse at 30˚C for 21 days and the experiment was repeated twice with four replicates per treatment. Germinated plants were evaluated on the basis of percent seed germination; number of leaves, leaf area, shoot height, fresh and dry weights of plants. The chlorophyll content index (CCI), chlorophyll fluorescence, transpiration (E), stomatal conductance (gs) and the net photosynthesis (A) were also determined after 21 days.
Chlorophyll content was measured with a portable chlorophyll meter (CCM-200, OPTI-SCIENCES, Tyngsboro, MA, USA); a portable laser leaf area meter (CI 203 Laser Leaf Area Meter, Camas, WA, USA) was used to scan the leaf area of mature leaves attached to corn and bean plants; and LCi portable photosynthesis meter (ADC Bioscientific Ltd. Hoddesdon, UK) was used to determine transpiration (E), stomatal conductance (gs) and the net photosynthesis (A). Measurements were taken from three mature leaves of each plant.
2.7. Determination of Chlorophyll Fluorescence
Corn and bean plants grown in local sandy soil amended with different concentrations of leaf tissue were used to evaluate leachate dosage-response on chlorophyll fluorescence. Chlorophyll fluorescence emission was measured using portable pulse amplitude modulated leaf chlorophyll fluorometer OS5-FL, (OPTI-SCIENCES, Hudson, USA) [14] . The fluorescence parameters measured in the kinetic test mode were: the minimal fluorescence yield (Fo), the maximal fluorescence yield (Fm), variable fluorescence (Fv), the ratio of the variable to maximal fluorescence or photochemical efficiency of PSII (Fv/Fm), and the apparent photosynthetic electron transport rate (ETR).
2.8. Effect of Leaf Extract on Nutrient Uptake
Ten grams of agar with half strength Hoagland’s solution was sterilized; amended with aqueous leaf extracts at 2.5%, 5.0%, and 7.5% (w/v) and allowed to solidify in sterilized test tubes. Surface sterilized corn and bean seeds were germinated in Petri dishes and those that began to germinate on the third day were selected and placed in the test tubes with the medium; which were then incubated at 25˚C under cool white fluorescent lights for 21 day. Plants were evaluated on basis of shoot and root development; and the development of symptoms of nitrogen and phosphorus deficiencies.
2.9. Biocontrol Experiment
Inhibition of Mycelia Growth Rate
Three plant pathogens: Fusarium f. sp. lycopersici (Fol), Rhizoctonia, Sclerotinia sclerotiorum and one biological control agent (Trichoderma harzianum) were used in this study. The fungal test organisms were isolated locally from host plants, soil and are part of our fungal collection.
The leaf extracts used above and sterile water as a control were added to PDA and water agar media aseptically at the following concentrations 2.5%, 5.0%, 7.5%, and 10.0% to determine the mycelial growth rate of each fungus. Actively growing mycelia plugs (5 mm diameter) of F. oxysporum f. sp. lycopersici (Fol), R. solani, S. sclerotiorum and one biological control agent Trichoderma harzianum were placed inverted at the center of amended medium in 9 cm diameter Petri dishes. Each test was replicated five times and the inoculated plates were incubated at 25˚C ± 1˚C and radial growth was recorded daily for 5 days. Mycelia plugs that showed no visible fungal growth were transferred to fresh PDA medium to ascertain viability of each fungus on the plug.
2.10. Statistical Analysis
Seed germination seedling growth bioassays were conducted in a complete randomized design (CRD) with four replications. The experiments were conducted at least twice and the data pooled and analyzed by analysis of variance (ANOVA) and the mean values were separated where applicable using Duncan’s Multiple Range test at a probability level of 0.05.
3. Results
3.1. Seed Germination
The leaf extract of C. lancifolius significantly inhibited the germination of corn and bean seeds (Figure 1 & Figure 2). The onset of germination in both corn and bean was delayed by 3 - 4 days with increased concentration of leaf extract. Both crops showed relatively similar germination and growth curves 10 days after treatment. Percentage germination decreased as the leaf extract concentration increased from 2.5% to 7.5% (Table 1). Seeds in 10% leaf extract did not germinate and in 7.5%, germination was reduced by 89.0%. The highest concentration (7.5%) of leaf extract resulted in the greatest reduction in the radicle and plumule lengths of both crops after 10 days. Plumule and radicle lengths of bean in 7.5% extract was reduced by 90.2% and 89.7%, respectively. In corn, 2.5% leaf extract enhanced or promoted the development of fibrous root system.
3.2. Solute Potential of Extract and Water Uptake by Seeds
The solute potential of aqueous leaf extract increased significantly with increased extract concentration. Increased leaf extract concentration resulted in a reduction in the amount of water imbibed by both bean and corn seeds (Figure 3). The least water uptake or imbibition was in the 7.5% leaf extract. Bean seeds imbibed significantly more water compared to corn seeds in all treatments. Water uptake in corn seeds was relatively the same in all three concentrations of leaf extract.
3.3. GC-MS Analysis
The GC-MS chemical constituents of C. lancifolius aqueous winter leaf extract is shown in Table 2 and the
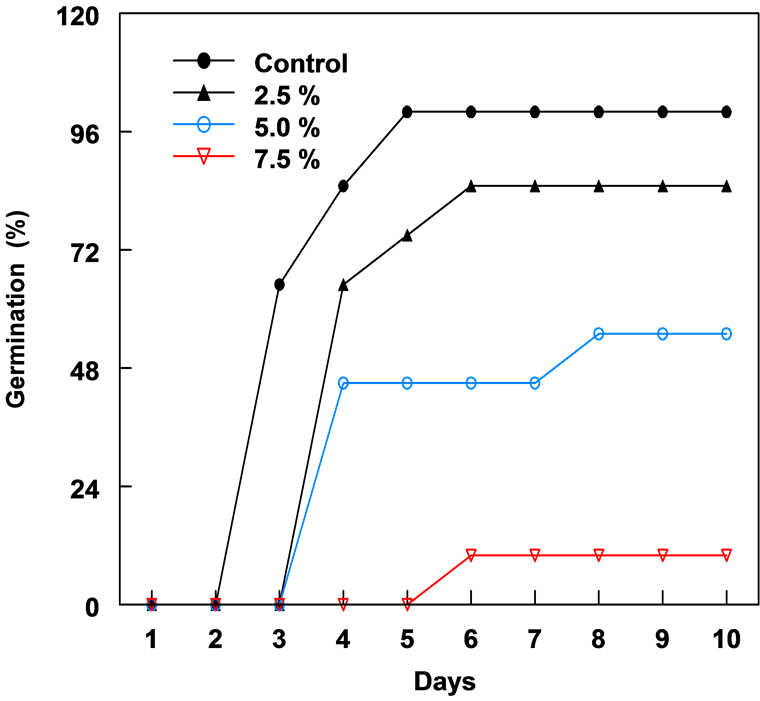
Figure 1. The percent germination of corn seeds in different concentrations of leaf extracts of C. lancifolius, 10 days after treatment.
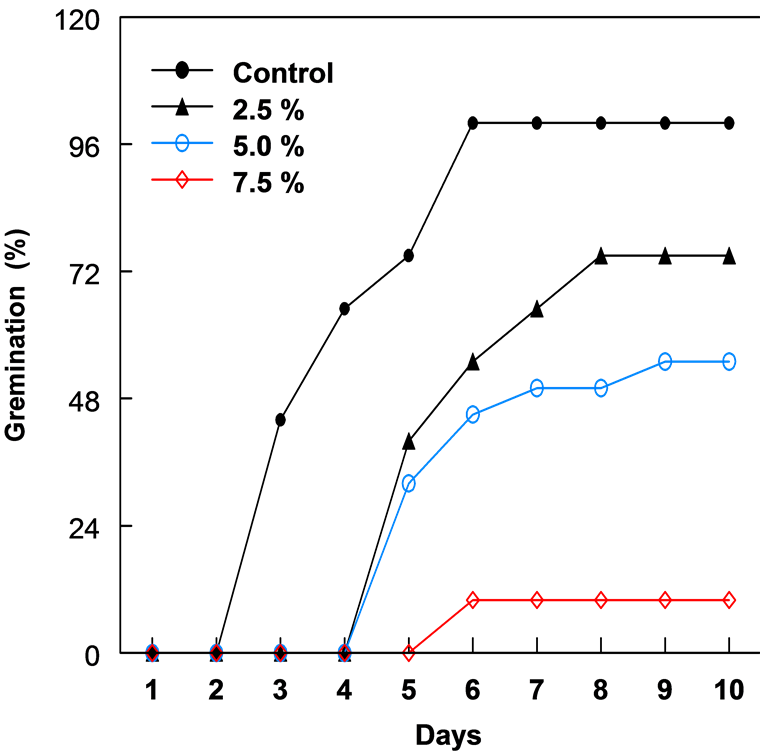
Figure 2. The percent germination of bean seeds in different concentrations of leaf extracts of C. lancifolius, 10 days after treatment.

Table 1. The effect of leaf extract of C. lancifolius on percent germination, plumule and radicle elongation. Each value represents the mean of 60 corn and bean seeds bioassay at 25˚C. Values in the same row followed by the same letter are not significantly different at P = 0.05 based on Duncan’s multiple range test (DMRT).

Table 2. Chemical constituents and retention times of each compound detected in the GC-MS analysis of aqueous leaf extract of C. lancifolius.
chromatogram of the compounds is shown in (Figure 4). Compounds identified included phenolics, terpenes, alkaloid, and fatty acids. The major constituents were β-phenylethylamine (11.14%), dotriacontane (9.91%), docosane (9.01%), hydroxyl phenyl acetic acid (6.6%), 6 tetracosane (5.61%), proceroside (5.14%), pyrogallol (4.90) and bicyclogermacrene (4.22). Overlapping peaks in the chromatogram indicated the complex nature of the leaf extract. The compounds 2,6,10,14,18 pentamethyl licosane and cyanidin-3-glucoside were detected in the summer samples but not in the in leaf samples harvested in the cool winter months.
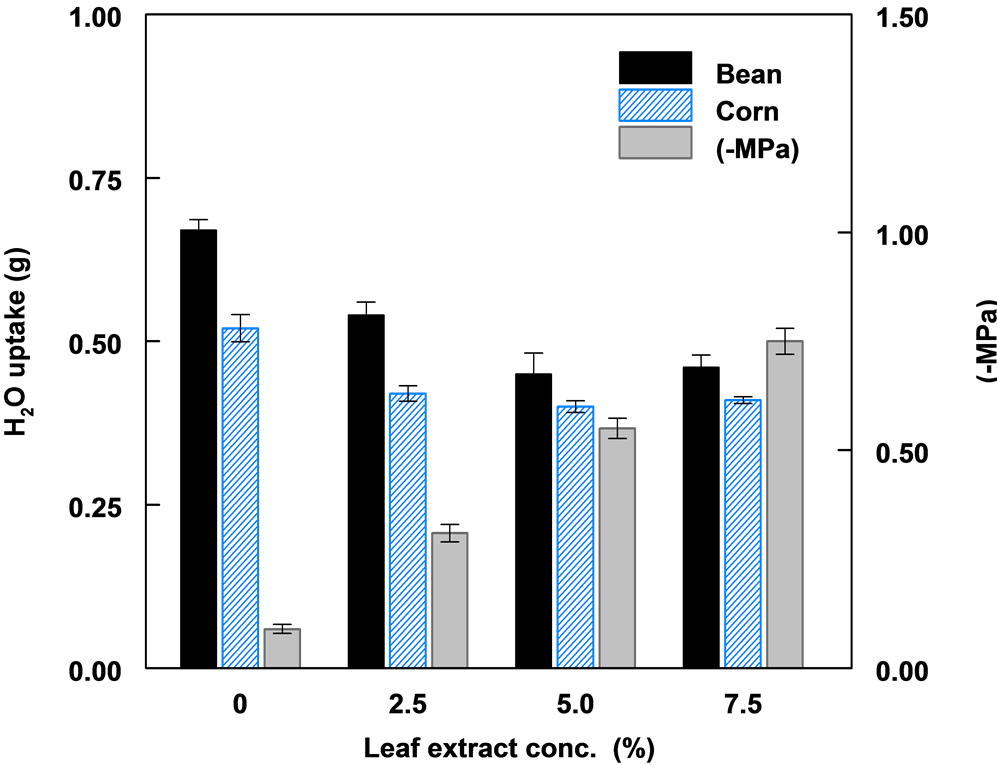
Figure 3. The effect of leaf extract concentration, corresponding solute potential (−MPa) of extract and water uptake by corn and bean seeds.
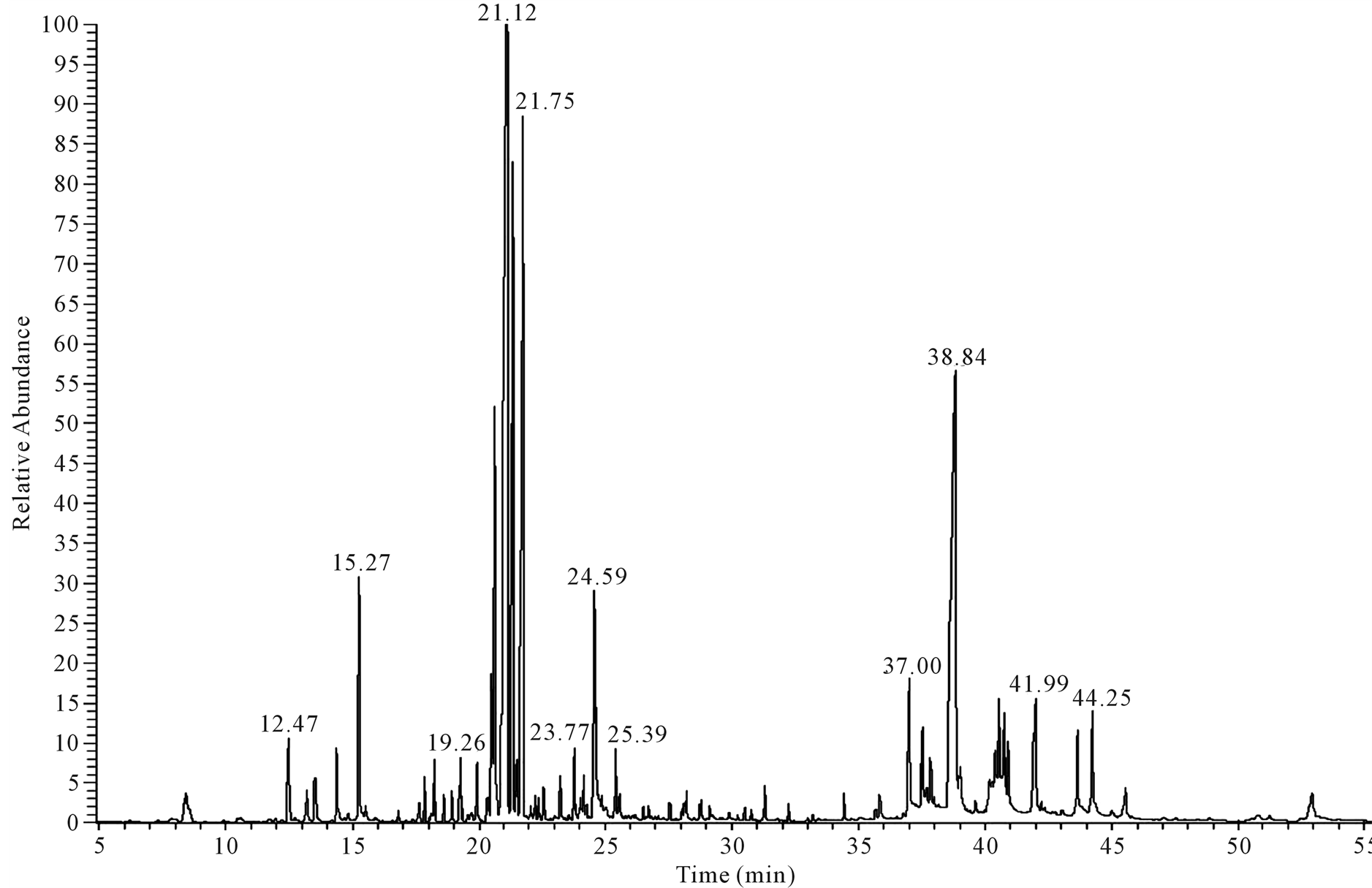
Figure 4. GC-MS chromatogram of leaf extract constituents of C. lancifolius using NIST 21 and Wiley 229 libraries spectral databases.
3.4. Leaf Tissue as Soil Amendment
Incorporation of 7.5% leaf tissue into local soil resulted in significant reduction in seed germination, growth traits (Table 3), transpiration and photosynthetic parameters in both corn and bean plants (Table 4). The reduction in growth traits chlorophyll content and net photosynthesis in 2.5% leaf extract was not significantly different from control. Transpiration and stomatal conductance were only significantly reduced in plants grown in

Table 3 . Percent germination and growth traits of corn and bean plants grown in sterilized (st) and nonsterilized (nst) local soil amended with different concentrations of ground leaf tissue of C. lancifolius. Values in the same row followed by the same letter are not significantly different at P = 0.05 based on Duncan’s multiple range test (DMRT).

Table 4. Photosynthetic rate (A), Transpiration (E), stomatal conductance (gs) and chlorophyll content index (CCI) in corn and bean plants grown in sterilized (st) and nonsterilized (nst) local soil amended with different concentrations of ground leaf tissue of C. lancifolius. Values in the same row followed by the same letter are not significantly different at P = 0.05 based on Duncan’s multiple range test (DMRT).
7.5% treatment. The growth inhibition and reduction in photosynthesis in both crops was also not significantly different in plants in sterilized and nonsterilized soils.
There was a significant difference in photochemical efficiency (Fv/Fm) between treatments in corn and bean plants (Figure 5). Similarly, electron transport rates (ETR) in both plants was significantly different between treatments of extracts (Figure 6). There was a linear relationship between leaf extract concentration and Fv/Fm with r values of 0.96 and 0.98 for corn and bean, respectively. The relationship between extract and ETR was curvilinear. Compared to corn, bean plants showed lower Fv/Fm and ETR values.
3.5. Effect of Leaf Extract on Nutrient Uptake
Corn and bean plants showed significant reductions in shoot and root growth in soil amended with ground leaf tissue (Figure 7). Inhibition of roots growth of both crops was greater than reduction in shoot growth. Bean plants showed nitrogen deficiency but corn plant showed nitrogen and phosphorus deficiencies. Plant with nitrogen deficiency were slightly chlorotic or yellowish green but phosphate deficient plants showed purple to red leaves early in the growth stage with dark brown leaf tips as the plants aged.
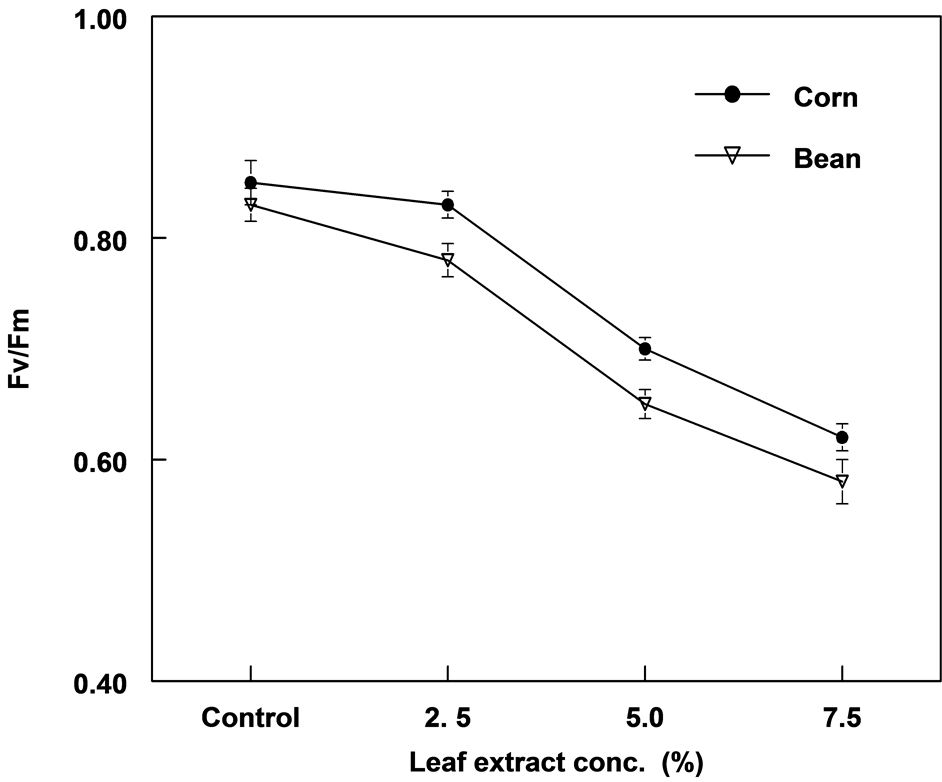
Figure 5. The effect of leaf extract of C. lancifolius on the maximum photochemical efficiency (Fv/Fm) in corn and bean plants.

Figure 6. The effect of leaf extract of C. lancifolius on the electron transport rate (ETR) in corn and bean plants.
Corn and bean seedlings grown in agar with half strength Hoaglands solution plus leaf extract particularly in 7.5% extract showed significant reductions in shoot and root development. The effects of leaf extract on shoot, root, fresh and dry weight are shown in Table5 The fresh and dry weights of plants were significantly affected by 7.5% leaf extract. Corn seedlings in 7.5% extract showed a slight phosphate and nitrogen deficiencies and the CCI of leaves was 6.9 in 7.5% leaf extract compared to 10.2 in the control plants.
3.6. Effect of Leaf Extract on Fungal Growth Rate
Mycelia growth of the pathogenic fungi was greatly inhibited as the concentration of leaf extract was increased (Figure 8). In the media amended with 10.0% leaf extract Fusarium oxysporum f. sp. lycopersici, Rhizoctonia solani and Sclerotinia sclerotiorum did not grow. The viability of these mycelia tested on the 10.0% treatment on freshly prepared PDA without extract showed no growth. A lot more sclerotia of S. sclerotiorum were formed in the 7.5 % extract amended PDA compared to the control (Figure 9). Sclerotia developed in R. solani did not increase in any of the treatments but the mycelia appeared light brown or pale in color in 7.5% treatment.
Mycelia growth in the controls and 2.5% treatment was not significantly different, but was significantly different in 5.0% and 7.5% at P = 0.05, (Figure 10). The growth of T. harzianum was not inhibited in the medium amended even in 10.0% leaf extract.

Figure 7. Response of plants grown in soil amended with ground leaf tissue of C. lancifolius; (a) = corn plants; left = control and right = 7.5%; (b) = bean plants; left = control and right = 7.5% leaf extract.

Table 5. Growth traits of corn seedlings grown in agar amended half strength Hoagland’s solution in different concentrations of leaf extract of C. lancifolius. Values in the same column followed by the same letter are not significantly different at P = 0.05 based on Duncan’s multiple range test (DMRT).
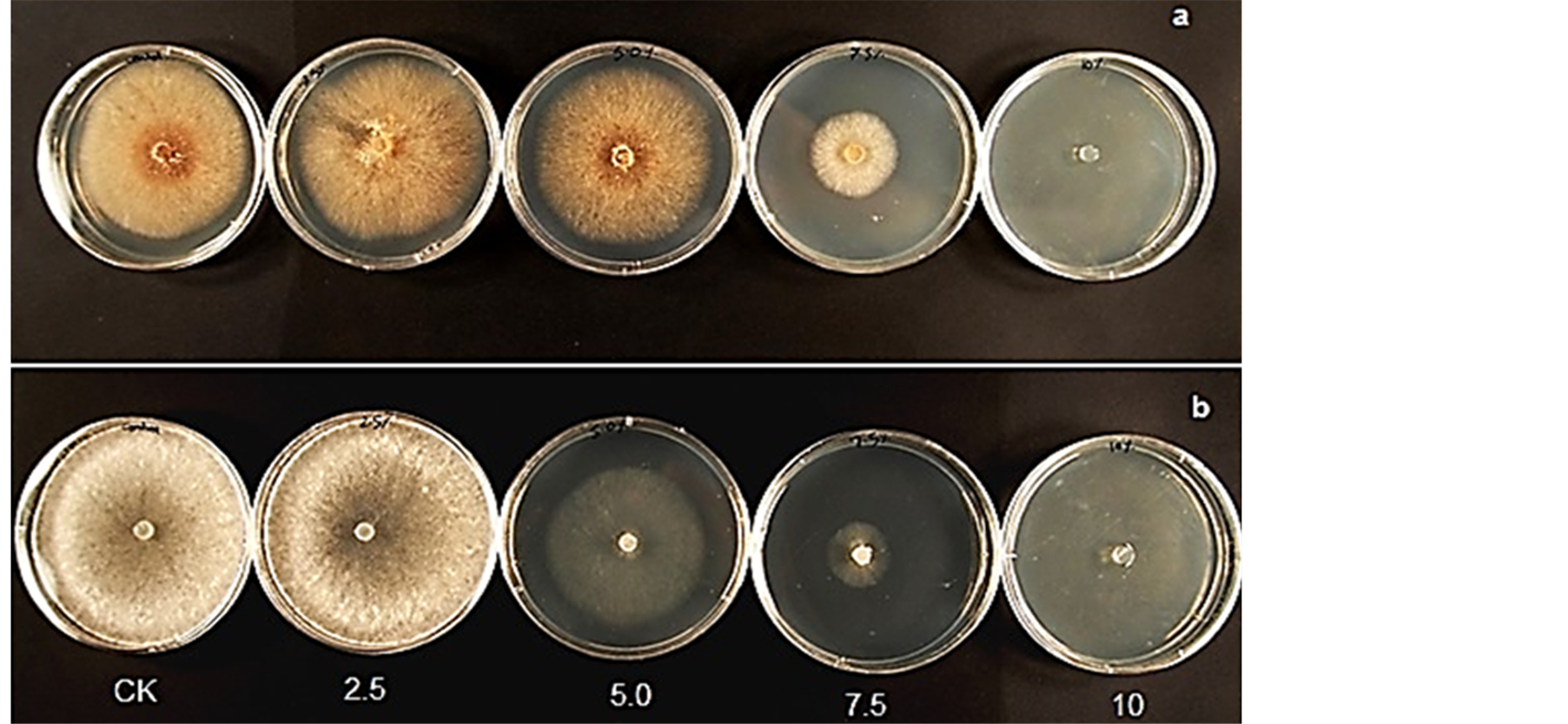
Figure 8. Mycelia growth of (a) Rhizoctonia solani and (b) Sclerotinia sclerotiorum on PDA amended with different concentrations of leaf extract of C. lancifolius.
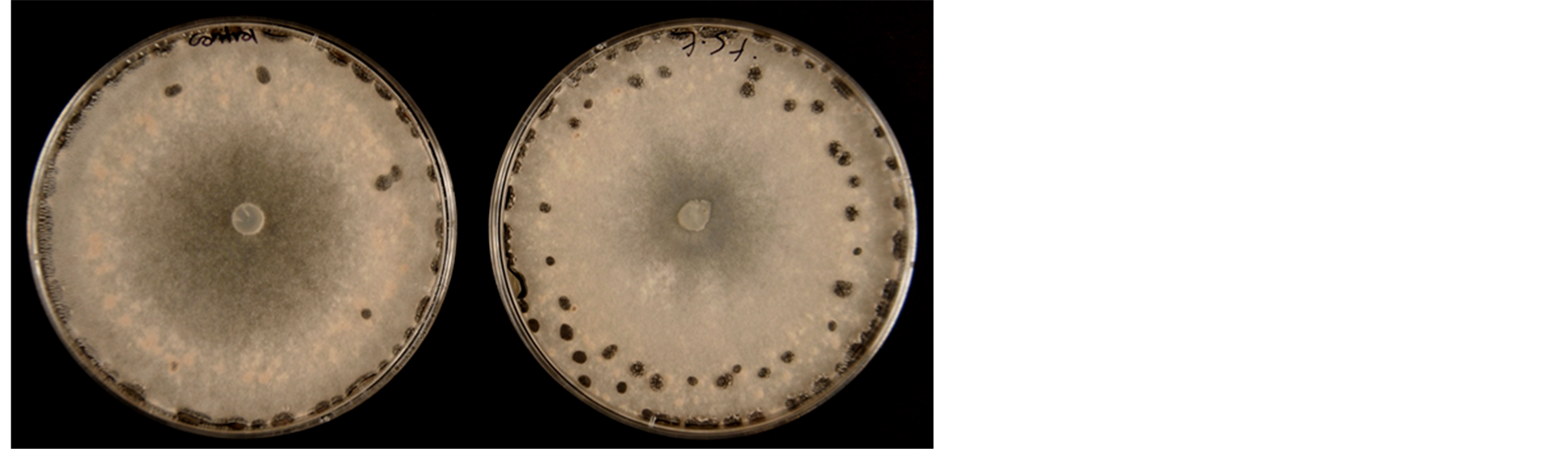
Figure 9. Mycelia growth of Sclerotinia sclerotiorum on PDA amended with leaf extract of C. lancifolius; left culture = control; right = 7.5% leaf extract.
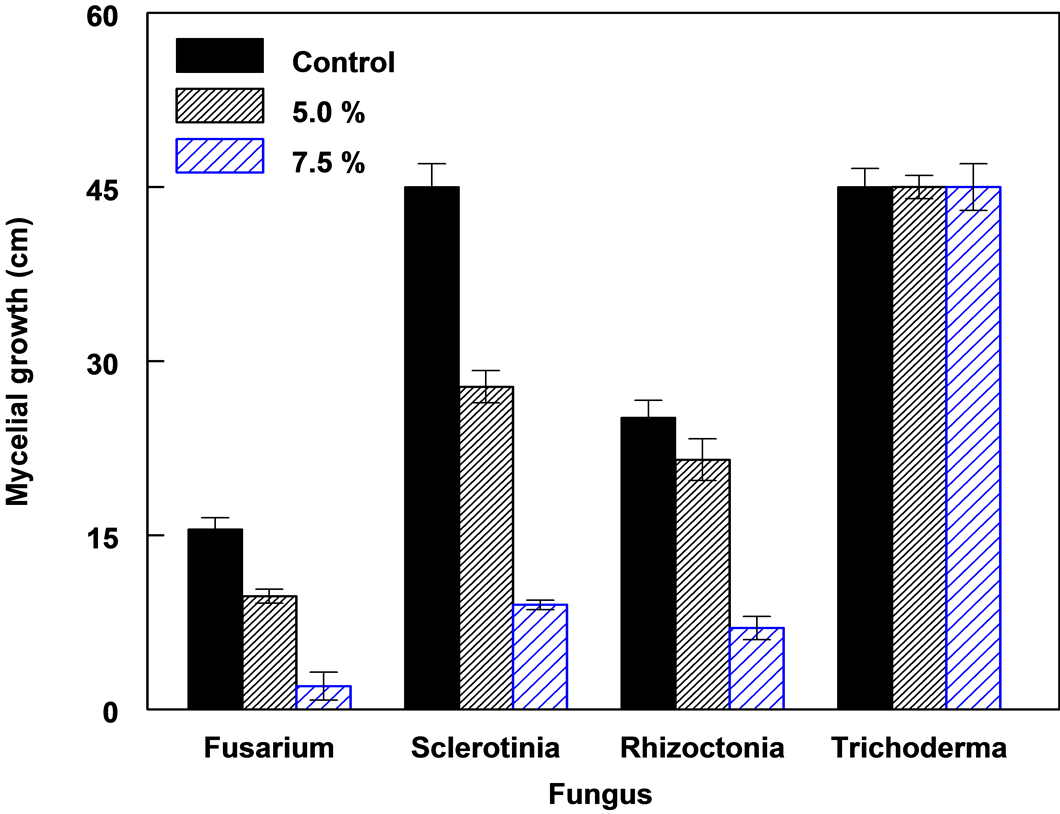
Figure 10. The growth of four fungal species on PDA amended with different concentrations of leaf extract of C. lancifolius, 3 DAT.
4. Discussion
Leaf extracts of C. lancifolius showed allelopathic effects in corn and bean seeds and plants. Analysis of the leaf extracts showed the presence of phenolics, terpenoids, alkaloids, and fatty acids. Phenolic compounds were predominant in the extracts and may have played a major role alone and/or collectively with other groups of compounds in inhibiting germination, growth and development of corn and bean seedlings. The leaf extract caused a delay seed germination as well as inhibition of growth of corn and bean plants. Germination decreased with increased leaf extract concentration and at 10.0% the seeds of both crops did not germinate. These observations are in agreement or similar to the inhibitory effects of leaf extracts of Brassica nigra [15] , Eucalyptus camandulensis [16] , Acacia auriculiformis [17] and alfalfa [18] in which germination and growth of some agricultural crops were proportional the concentration of extracts. Regression analysis between seed germination and extract concentration showed R2 = 0.98 in both corn and bean. Elongation of radicles and plumules were significantly inhibited in 7.5% leaf extract this may be related to high concentration of allelochemicals, phenolic acids terpenes and alkaloids in the extract [19] . Radicles were more sensitive to allelochemicals of C. lancifolius than the plumules. Similar sensitives of radicle lengths compared to plumule and shoot lengths have been reported [15] [18] . The inhibitory effects of increased extract concentration might be due to synergistic or additive effects of the constituents of the extract rather than a single compound. The lowest concentration of leaf extract (2.5%) promoted or enhanced the development of fibrous root system of corn, similar to observations reported with leachates of Eucalyptus which had varying degree of inhibitory and stimulatory effects on germination and growth of crop plants [20] [21] .
Aqueous leaf extract concentration also caused a significant reduction in water uptake necessary for seed germination in both crops. The nature of testa of bean and corn seeds could have been a factor in water uptake required for active metabolism during germination. Differences in water uptake between bean and corn seeds could also be due to hygroscopic tissue of micropyle-hilium fissure of bean seeds which can passively allow water uptake in seeds of some legumes [22] or related to in osmotic potential of the leaf extracts [23] . Germination was higher in the control and lower concentrations of leaf extracts and water uptake could have played a role in the process. During seed germination, a number of metabolic and physiological processes could be attributed to water uptake [24] . Although biochemical events that trigger germination are unknown; increase in respiration is followed by hydrolytic enzyme activity, cell division and enlargement in the embryo; all of which could have been affected by allelochemicals in the leaf extract [25] .
GC-MS analysis identified at least thirty one compounds; 11 of them were phenolic compounds. This is the first report of the chemical constituents of aqueous leaf extract of C. lancifolius. Phenolics are known to interfere with energy metabolism, cell division, mineral uptake and other biosynthetic processes such as net photosynthesis, respiration and enzymatic activities [2] . Phenolics in C. lancifolius leaf extract may have contributed significantly to suppress germination, growth and biomass production of corn and bean plants.
In this study microbial activity was not a contributing factor because the amended sterilized and nonsterilized soils showed relatively the same inhibitory effects in corn and bean plants.
The results also showed chlorophyll content was significantly reduced in in 5.0% and 7.5% leaf extracts which is accordance with other observations [26] . The reduction in chlorophyll content ultimately resulted in the reduction or decline in photosynthesis in both crop plants [8] [27] .
Chlorophyll fluorescence data of both treated corn and bean plants indicated physiological stress on photosynthesis particularly PSII attributable to reductions in Fv/Fm and ETR. Corn and bean plants, 7.5% treatment showed 27.1% and 30.1% inhibition in FV/Fm; and 46.1% and 45.3% inhibition in ETR, respectively. Fv/Fm has been shown to be constant (0.83 - 0.84) in non stress plant species [28] ; and any slight decrease is an indication of diminishing photosynthetic efficiency. The photosynthetic efficiency of both bean and corn plants were significantly affected in this study but bean was slightly more susceptible than corn because of the greater reduction in Fv/Fm.
The application of half-strength Hoagland’s solution did not completely overcome the reduction in growth and nutrient deficiencies particularly in the 7.5% leaf extract. This is in agreement with the observations [29] , who observed that the application of nitrogen and phosphorus did not overcome the effects of ferulic acid.
Plants with allelopathic properties can be exploited for biological control of weeds and plant pathogens. In this study the antifungal activity of C. lancifolius leaf extract was tested against five fungi. The leaf extract showed antifungal activity against S. sclerotiorum, F. oxyposporum f. sp lycopersici and R. solanii which are plant pathogens. The antifungal effect of the leaf extract was concentration dependent with negative linear relationship between the concentration of extract and mycelia growth. Phenolic compounds which were the most abundant group of compounds in the leaf extract including pyrogallol have been shown to have antimicrobial and allelopathic activity [30] -[33] . The leaf extract C. lancifolius did not inhibit the growth of T. harzianum species. This indicates that T. harzianum probably has the capacity to detoxify, metabolize or lacked the receptors sites for the antifungal components in the extracts. The ineffectiveness of leaf extracts in T. harzianum species; to act as contact fungicides [34] , disrupt fungal cell membranes [35] or to inactivate the enzymes and metabolic processes [36] implies that the extracts could be used to complement or enhance the activity of T. harzianum in the control of soil-borne pathogens. The local isolate of T. harzianum has been shown to be a good biological control agent of soil-borne pathogens [37] .
The allelochemicals in the C. lancifolius leaf extract showed antifungal activity on the three pathogens and herbicidal effects on two crops. Additional studies on the long term and decomposing effects of the plant litter on soil-borne pathogens and other microorganisms would enhance our understanding on the allelopathic effects of C. lancifolius. As C. lancifolius is propagated nation-wide, additional studies on the invasive potential of the species need to be conducted to evaluate the effects of leachates from leaves, fruits and seeds; and root exudates.
5. Conclusion
The results of this research showed the negative effects of leaf tissue and extracts of C. lancifolius in the germination, the growth traits, physiological and biochemical processes of corn and bean plants. Corn (monocot) was more tolerant to the leaf extract than bean (dicot) plant. The inhibitory effects correlated with increased concentration of leaf extract. In 10% leaf extract, seeds of both crops did not germinate. Thirty one compounds were detected by GC-MS in the leaf extract and most of them were phenolic compounds. The allelopathic effects of the leaf extract with regards to germination were associated with imbibition by the seeds as well as the solute potential of extracts. Growth inhibition of the seedling and plants correlated significantly with chlorophyll content, photosynthesis by limiting photochemical efficiency and electron transport rate of PS II; inhibition of nutrient uptake that induced nitrogen and phosphate deficiencies. Leaf extract inhibited mycelia growth of Fusarium oxysporum f. sp. lycopersicon, Sclerotinia sclerotiorum, and Rhizoctonia solani but not Trichoderma harzianum. Accordingly, the leaf extract of the C. lancifolius has the potential as herbicide but cannot be recommended to be used as a biological control agent of soil borne fungal plant pathogens because it has the potential to cause biochemical and physiological damage to cultivated crops.
Acknowledgements
The Authors express their gratitude to the Kuwait Foundation for the Advancement of Sciences (KFAS # 2011- 1207-08), College of Graduate Studies and Research Administration, Kuwait University for funding this research. We also acknowledge the assistance of Divya Saju as the Research assistant.
NOTES
*Corresponding author.