Oral Low-Molecular Weight Hyaluronic Acid in the Treatment of Atrophic Vaginitis ()
1. Introduction
Cross-sectional studies provide strong evidence that sexual activities and sexual function decline with age. Literature evidence demonstrates that the sexual function of women and the frequency of sexual activities start to decline at the aged 20 - 30 years, reaching a peak at the postmenopausal level [1] . Interestingly, Koster and coworkers [2] demonstrated that the decrease in sexual desire correlates significantly with the women’s subjective assessment of being climacteric. During the climacteric period, sex steroid deficiency causes many neuroendocrine changes. As a result, menopausal women frequently experience a variety of symptoms such as hot flushes, sleep and mood disorders, weight gain and sexual dysfunction. Among these symptoms, sexual problems are the most important health concerns of climateric women and are often associated with significant personal distress, thus resulting in diminution of self-worth, self-esteem, and with a significant reduction in life satisfaction and quality of the couple’s relationship [3] [4] . Menopausal symptoms are mainly due to an abrupt decrease in circulating estrogen levels, which is the cause of vaginal atrophy [5] . Vaginal atrophy causes vaginal dryness, soreness and irritation, dysuria, increased vaginal discharge, recurrent urinary tract infections, pain and vaginal bleeding associated with sexual activity. Despite the frequency and effects of vaginal atrophy symptoms, they are often under-reported and, consequently, under-treated because of patient and clinician lack of knowledge of available treatments, embarrassment about initiating a discussion of symptoms and reluctance to initiate hormonal therapy [6] . Diagnosis requires a clinical history and gynecological examination [7] . When vaginal atrophy is correctly diagnosed, therapeutic strategies are divided into non-hormonal and hormonal treatments. Among the hormonal therapy, the intravaginal application of preparations containing estrogens is the most common used treatment. Oral administration of hormones is not recommended because of some evidence that the use of oral estrogen-progestogen therapy beyond 2 - 5 years may increase the risk of breast cancer [8] -[10] . Oral estrogen therapy was considered safer in term of cancer risk [11] . However, a recent evidence indicates a significant increase in breast cancer risk for both estrogen-only and combined estrogen-progestogen therapy among current users with less than 2 years of use [12] . Therefore, although topical estrogenic products have been developed to minimize the systemic adverse effects of the oral hormone replacement therapy, they cannot yet be considered as risk-free alternative in case of prolonged use [13] , thus promoting an active search for different and somehow innovative therapies. Among these, both daily intravaginally administered testosterone or dehydroepiandrosterone (DHEA) have been proposed for the treatment of vaginal atrophy [14] . However, more comprehensive studies are required to confirm the efficacy of this therapy. Last but not least, these therapeutic options still represent a hormone-based therapy, while nowadays there is the need for alternative therapies making women more prone to the use of medications with minor side effects. Although self-medications do not replace hormones, they are associated with fewer adverse effects than prescription medications, and they may be well-suited for several women [6] . The most common used non-hormonal strategies for the treatment of vaginal atrophy symptoms are vaginal lubricants and moisturizers as well as complementary and alternative therapies including vitamin E, olive oil, herbal supplements and isoflavones [15] . Interestingly, since vagina looses collagen and water-retaining ability during menopause, the efficacy of Hyaluronic Acid (HA) in the treatment of atrophic vaginitis has recently been evaluated. The effects of HA include the restore of mucous membranes surface (such as vaginal mucosa), the right level of hydration and the protection against external environment. Two clinical trials proved the efficacy of a gel containing HA, phytoestrogens from Humuluslupulus extract, Vitamin E and liposomes in reducing vaginal dryness and all the other evaluated symptoms in post-menopausal women affected by vaginal atrophy [13] . A successive study was aimed to compare the efficacy of vaginal tablets of either HA or estradiol for the treatment of atrophic vaginitis in climacteric women [16] . Minozzi and colleagues performed an open multicentric longitudinal study to evaluate the efficacy of oral administration of HA, administered in a low molecular weight pharmaceutical form, in postmenopausal patients suffering of atrophic vaginitis [17] . Low molecular weight guarantees a high absorption and bioavailability, and then a greater action on skin and mucosal tissue, such as vaginal mucosa. The results of this study showed that HA administration improved vaginal atrophy already after one month of treatment, and that it slowed the physiological aging of vulvar and vaginal tissues, increasing their hydration rate [17] . On the basis of these preliminary results and in order to reduce the use of hormonal therapy, given the cancer risk, the aim of the present study was to evaluate in a double blind, placebo-controlled trial whether HA, orally administered in a low-molecular weight oral formulation.This alternative therapy could ameliorate vaginal atrophy, improving the symptoms and modifying the vaginal epithelium in postmenopausal women, without side effects and particular risks for the women’s health.
2. Methods
2.1. Subjects
Twelve women, aged 45 - 65 years, with symptoms of atrophic vaginitis were recruited for this double blind, placebo-controlled clinical study. Participants were required to be at least 12 months postmenopausal, and each of them showed mucosal atrophy and the main bothersome related symptoms, such as itching, burning, and dyspareunia. Patients were randomized via computer software to receive either low-molecular weight HA tablets (220 mg hyaluronic acid per tablet, Ialos®, Lo.Li.Pharma Srl, Rome, Italy) per os (two tablets/day for 10 days, and subsequently one tablet/day for three months) or placebo (two tablets/day for 10 days, and subsequently one tablet/day for three months). All the patients gave their written informed consent before entering the study.
Vaginal biopsies were taken from the 2 groups at baseline and after three months of treatment, and vaginal epithelium was analysed using light microscopy, in order to check the mucosal state and the presence of vaginal anomalies. As secondary endpoints, typical symptoms of atrophic vaginitis (itching, burning and dyspareunia) were evaluated in the 2 groups, at baseline and after three months of treatment (t1). The severity of each symptom was self-assessed and received a grade based on a 4-point scale (0 = absent, 1 = mild, 2 = moderate, 3 = severe).
2.2. Light Microscopy
The vaginal biopsies were fixed in 2.5% glutaraldehyde 0.2 M in phosphate buffer (pH 7.4) at +4˚C for 4h, washed overnight with 0.2 M phosphate buffer (pH 7.4), and postfixed in 1% OsO4 in 0.2 M phosphate buffer (pH 7.4) at + 4˚C for 1 h. Samples were dehydrated in graded ethanol and acetone, and flat-embedded in Durcupan. Semithin sections (1 µm) were cut with a LKB Ultrotome V ultramicrotome, stained with an aqueous solution of 1% toluidine blue in 1% borax and 1% pironine [18] and viewed and photographed with a Zeiss Primo Star light microscope (LM).
2.3. Morphometric Analysis
A morphometric analysis of the vaginal epithelium of patients treated with either HA tablets or placebo was carried out by the LM on the number of epithelial layers and the overall epithelial thickness. Light microscopy data were obtained from 10 semithin sections/patient (a total of 60 section/group), collecting 1 semithin section every 100; all micrographs were obtained with a Zeiss Primo Star light microscope at the same magnification of 400× and processed with a Macintosh MacBook, using the Adobe Photoshop 8.0.1 software. On each micrograph two independent observers counted the number of epithelial layers and measured the overall epithelial thickness: both these data were obtained by tracing a straight line perpendicular to the epithelium and by calculating the number of layers and the epithelial thickness expressed in μm along each line. The OptilabGraphtek software was used to measure the epithelial thickness. Results obtained from each group were compared with the respective samples at baseline.
2.4. Statistical Analysis
Statistical analysis of the results was performed using the Student T-test by the S.A.S./Sta 6.0.3 software. p value of ≤0.05 was considered as statistically significant and data were expressed as means ± standard deviation (SD).
3. Results
3.1. Vaginal Biopsy
When the vaginal biopsies obtained from both groups at baseline were observed with the LM, they showed the typical postmenopausal morphology. In fact, the epithelium was formed by a single layer of basal cells, with round or elliptical nuclei, and by many layers of irregularly arranged parabasal cells. The loose lamina propria, which is separated from the epithelium by a well-evident basement membrane, showed stromal cells and blood vessels (Figure 1(A)).
After treatment, in the placebo group, the vaginal epithelium consisted also of a single layer of basal cells of different size and shape, which were connected to the basement membrane, and by many layers of parabasal cells. The most superficial were flattened, with elliptical nuclei and hypochromic cytoplasm. In the loose lamina propria many blood vessels were observed (Figure 1(B)).
In the vaginal biopsies obtained from the HA group, different morphological patterns were observed. In fact, in 3 patients the epithelium was higher compared to the baseline and the placebo groups. It was formed by a single layer of columnar basal cells, oriented parallel to the basement membrane, by many layers of parabasal cells, with round or oval nuclei, and by many layers of large intermediate cells, whose cytoplasm was filled with keratohyaline granules. The lamina propria showed a denser appearance (Figure 1(C)).
In 2 patients of the HA group, the epithelium was taller and it was formed by basal, parabasal, intermediate granular cells, and by many layers of superficial cells with small, pyknotic nuclei and abundant, amorphous cytoplasm (Figure 1(D)).
In a single patient of the HA group the vaginal epithelium reached the highest thickness (about 150 µm) and basal, parabasal, intermediate non granular cells, and a large number of more superficial flattened cells could be observed. The superficial layer, in its deepest part, was formed by cells exhibiting a polygonal shape, with round nuclei and clear perinuclear cytoplasm. Towards the surface, the cells showed at first a tightly packed appearance, with pyknotic nuclei and heavily stained membranes, and finally a keratinized morphology, with rare nuclei and clear cytoplasm (Figure 1(E)).
3.2. Morphometric Analysis
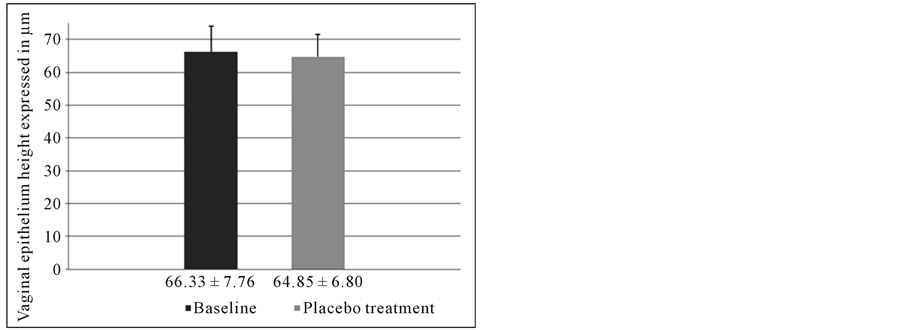
The data obtained from the morphometric analysis of the vaginal epithelium of both groups of patients showed no statistically significant differences between baseline (control) and after treatment with placebo, either for the number of epithelial layers (p = 0.887) or for the overall epithelial thickness (p = 0.269) (Figure 2(A), Figure 2(B)). On the contrary, before and after HA-treatment, statistically significant differences (p < 0.001) were found either for the number of epithelial layers or for the overall epithelial thickness (Figure 2(C), Figure 2(D)).

Figure 1. (A) Vaginal biopsy in both groups at baseline; (B) Vaginal biopsy in the placebo-treated group; (C) Vaginal biopsy in the HA-treated group; (D) Vaginal biopsy in the HA-treated group; (E) Vaginal biopsy in the HA-treated group. Toluidine blue-Pironine stain. Scale bar: 25 µm. Basal cells (b); parabasal cells (pb); blood vessels (v); intermediate cells (i), superficial flattened cells with round nuclei and clear perinuclear cytoplasm (s1), with pyknotic nuclei and heavily stained membranes (s2), with keratinized morphology, rare nuclei and clear cytoplasm (s3).
 (A)
(A) (B)
(B)  (C)(D)
(C)(D)
Figure 2. (A) Number of cell layers of the vaginal epithelium of placebo-treated women related to the basal specimens. No statistically significant difference was found between the groups; (B) Mean height of the vaginal epithelium of placebotreated women related to the basal specimens. No statistically significant difference was found between the groups; (C) Number of cellular layers of the vaginal epithelium of HA-treated women related to the basal specimens. A statistically significant difference (p < 0.001) was found between the two groups; (D) Mean height of the vaginal epithelium of HA-treated women related to the basal specimens. A statistically significant difference (p < 0.001) was found between the two groups.
3.3. Symptoms
The evaluation of the main symptoms related to vaginal atrophy showed a strong improvement after three months treatment with HA oral tablets (Table 1): mild itching and dyspaurenia was still present only in 2 patients. In the placebo group, no particular improvement of symptoms was observed at T1 compared to baseline.
4. Discussion
Recent clinical studies have shown that during the menopausal transition and after menopause, up to 45% of women will develop vaginal atrophy as a consequence of decreased levels of circulating estrogens [6] . Symptoms include vaginal dryness, itching, soreness, bleeding, increased susceptibility to infection and pain with sexual intercourse. These vulvovaginal atrophic changes have a negative impact on postmenopausal woman’s sexual activity and quality of life. Indeed, while other postmenopausal symptoms, such as hot flushes and night sweats, resolve spontaneously in time, atrophic symptoms affecting the vagina and lower urinary tract are often progressive and frequently require a treatment. In occasion of the World Menopause Day 2010, the International Menopause Society Writing Group estimated that only 25% of postmenopausal women in the Western society will seek medical advice for vulvovaginal atrophic symptoms [19] . This reluctance is partially due to the fact that in recent years the hormone replacement therapy, that is effective in treating vaginal atrophy, has been related to

Table 1. Self-assessed evaluation of symptoms in the two study-groups at baseline and after three months (T1) of treatment.
increased risk of breast cancer, heart disease and stroke. Despite it is nowadays accepted that local hormonal treatment of vaginal atrophy is not associated with these possible risks of systemic of hormone replacement therapy, cultural reasons make still most women reluctant to discuss treatment options with clinicians. During menopause, vagina loses collagen and water retaining ability. For this reason, lubricants like hyaluronic acid can be used as adjuvant to counteract the vaginal atrophy also due to estrogen deficiency. In the present study, we tested in a double blind, placebo-controlled trial whether HA, orally administered in a new low-molecular weight oral formulation, could ameliorate vaginal atrophy and modify vaginal epithelium in postmenopausal women. The results of our study show that the postmenopausal women administered with oral HA had reduced epithelial atrophy after three months of treatment compared to placebo-treated women. In particular, at the end of the treatment HA-treated patients had higher epithelium thickness and a higher number of epithelial layers than placebo-treated women. Furthermore, treatment with oral HA was able to ameliorate the symptomatic condition of women affected by vaginal atrophy, with a consequent improvement in the quality of life. These results confirm and extend previous findings about the efficacy of HA preparations in vaginal atrophy. Indeed, Morali and coworkers showed that a gel containing HA, liposomes, phytoestrogens from Humuluslupulus extract, and Vitamin E, applied on the external genitals or intravaginally in postmenopausal women with genital atrophy, improved vaginal dryness and related symptoms since the first week of treatment, with no adverse effects and high compliance by the test subjects [13] . Similarly, Tea and colleagues found a significant reduction in symptoms of atrophic vaginitis in breast cancer patients suffering from hormone or chemotherapy-induced atrophic vaginitis and treated with HA vaginal tablets [20] . Few years later, Ekin and colleagues compared the effectiveness of the HA acid vaginal tablets with estradiol vaginal tablets for the treatment of atrophic vaginitis [16] . The results of their study showed that both treatments provided relief of vaginal symptoms, improved epithelial atrophy, decreased vaginal pH, and increased maturation of the vaginal epithelium. On the basis of these results, the authors concluded that HA vaginal tablets can be used in patients with atrophic vaginitis who do not want to or cannot take a local hormonal treatment [16] . However, because of cultural, personal, and hygienic reasons, some patients show low compliance towards the vaginal route of administration. In these patients, an ideal treatment would be represented by the oral administration of hydrants devoid of important side effects, such as HA. The absorption and the bioavailability of orally administered HA can be improved by new pharmaceutical formulations containing low molecular weight HA [21] [22] .
5. Conclusion
The present study is therefore the first double blind, placebo-controlled trial that showed that HA, orally administered in a low-molecular weight oral formulation, can ameliorate vaginal atrophy and modify vaginal epithelium in postmenopausal women. This is due to the direct action oh HA on the vaginal mucosa, restoring its physiological state and counteracting inflammation. Thus, even though the small number of patients and the need to perform additional studies in order to confirm our preliminary findings, we have shown the evidence that low molecular weight HA tablets meet all the requirements of an optimal treatment for vaginal atrophy management: reversal of atrophic anatomic changes, symptom relief and good patients’ compliance.
Source of Funding
Study not funded by any grant.
NOTES
*Corresponding author.