1. Introduction
Methionine, an essential amino acid was first isolated by Mueller [1] . High levels of methionine can be found in nuts, meat and some other plant foods. However, in humans and animals, this amino acid cannot be produced and is therefore an important nutrient additive in their feeding. The impact of L-methionine on animal nutrition and the consequences of its absence as a nutritive feed additive have been investigated. It was observed for poultry that the stability of shells decreases just as the milk production in cow does [2] .
Currently, methionine is produced either by chemical synthesis or hydrolyzing proteins. These processes are expensive. Chemical synthesis produces a mixture of D-and L-methionine [3] whereas hydrolysis of proteins leads to a complex mixture from which methionine must be separated. Biologically active L-methionine can be produced either by enzymatic synthesis (bioconversion of precursors), or by submerged fermentation using microorganisms [3] . Because fermentation processes have been able to inexpensively provide many other amino acids, there is a significant interest in developing a microbial process for commercial production of methionine [4] [5] .
A number of microorganisms capable of producing amino acids have been isolated and amino acids such as lysine, threonine, isoleucine and histidine have been produced successfully by fermentation [6] [7] . Attempts have been made to overproduce biologically active L-methionine using fermentation [8] -[10] .
This study was conducted to examine the influence of cultural conditions on methionine accumulation by Bacillus thuringiensis EC1.
2. Materials and Methods
2.1. Microorganism
Bacillus thuringiensis EC1, isolated from fermented oil bean seeds, Pentachletra macrophila Benthan and identified based on 16S rRNA sequencing at Macrogen Incop., Republic of Korea was used. The organism was maintained on Nutrient Agar (Lab M) slant at 4˚C.
Fermentation in Submerged Medium
Fermentation was carried out following the method described by Ozulu et al. [11] . A loopful of a 24 h culture of Bacillus thuringiensis EC1was inoculated into 1ml of a sterile seed medium (peptone, 10.0 g; yeast extract, 10.0 g; NaCl, 5.0 g; H2O, 1 L; pH 7.2) in a test tube and incubated at 30˚C on a VWR DS2-500-2 orbital shaker at 160 rpm for 24 h. A 100 ml Erlenmeyer flask containing 20 ml of the sterile fermentation medium {KH2PO4, 0.05 g; K2HPO4, 0.05 g; MgSO4∙7H2O, 0.1 g; MnSO4∙4H2O, 0.001 g; FeSO4∙7H2O, 0.001 g; CaCO3, 20.0 g; (basal medium), glucose, 20.0 g, (NH4)2SO4, 10.0 g; H2O, 1 L; pH 7.2} was inoculated with 1ml (ca. 3.15 × 106 cell/ml) of the seed medium. The flasks prepared in duplicates were incubated at 30˚C on a shaker (160 rpm) and the broth culture assayed for methionine accumulation after 72 h. Uninoculated flasks served as control.
Methionine Assay
Methionine was assayed for, following the method described by Greenstein and Wintz [12] . The broth culture was centrifuged at 1500 × g for 15 min and 5 ml of the supernatant in a test tube was added 1ml of 5 N NaOH and 0.1 ml of 10% sodium nitroprusside solution. The tube was thoroughly shaken and the mixture allowed to stand for 10 min. Then 2 ml of 3% aqueous solution of glycine was added to the reaction mixture with frequent shaking over a period of 10min. After an additional 10 min interval, 2 ml of concentrated orthophosphoric acid was added drop wise to the mixture and the test tube properly shaken. Colour development was allowed to proceed for 5 min and colour intensity measured at 540 nm in a spectrophotometer (PerkinElmer Lambda 35 UV-VIS). The methionine yield was extrapolated from a standard methionine curve.
2.2. Optimization of Fermentation Conditions for Methionine Production
2.2.1. Effect of Medium/Fermenter Volume Ratio and Inoculum Size on Methionine Production
The effects of medium/fermenter volume ratio (volume of the culture medium: volume of fermentation flask) and inoculum size on methionine production by Bacillus thuringiensis EC1 were examined. Erlenmeyer flasks (100 ml) with different volumes (20, 25, 30 ml) of the sterile fermentation medium were each inoculated with 1 ml of seed inoculum. Each 1 ml contains a loopful (ca. 3.15 × 106 cell/ml) or two loopfuls (ca. 7.56 × 108 cell/ml). The flasks were incubated and methionine accumulation determined as previously described.
2.2.2. Effect of Carbon Source
Different carbon sources (Glucose, Maltose, Mannitol, Sucrose, Lactose) were studied for their effects on methionine accumulation by B. thuringiensis EC1. A 100 ml Erlenmeyer flask containing 20 ml of the basal medium, carbon source 20.0 g, (NH4)2SO4, 10.0 g, pH 7.2 was inoculated with 1 ml of a 24 h culture of the seed inoculum Fermentation processes and methionine determination were as earlier described. The carbon source with the highest methionine yield was used for further studies.
Effect of varying concentrations (20, 40, 60, 80, 100 g/l) of maltose was also investigated.
2.2.3. Effect of Nitrogen Source
Effect of different nitrogen sources (ammonium sulphate, ammonium chloride and urea) on methionine production was examined. A 100 ml Erlenmeyer flask containing 20 ml of the basal medium, maltose 20.0 g, nitrogen source 10.0 g, pH 7.2 was inoculated with 1ml of a 24 h culture of the seed inoculum. Fermentation was carried out as previously described and methionine accumulation determined from the broth culture. The nitrogen source with the highest methionine yield was used for further studies.
Effect of varying concentrations (10, 20, 40, 60, 80 g/l) of ammonium sulphate was also studied.
2.2.4. Effect of Growth Stimulators
The influence of 0.1% (w/v) yeast extract, peptone, casein and their mixtures on methionine yield by B. thuringiensis EC1 was examined. Methionine was determined as previously described.
2.2.5. Influence of Vitamins
Fermentation process was conducted to determine the influence of varying concentrations (0.10, 1.0, 5.0, 10.0 µg/ml) of riboflavin, pyridoxine, nicotinic acid and thiamine HCl on methionine production by B. thuringiensis EC1.
2.2.6. Influence of Bivalent Metals
Fermentation process was carried out to investigate the influence of varying concentrations (0.10, 1.0, 5.0, 10.0 µg/ml) of CaCl2, ZnCl2, BaCl2 and MgO on methionine accumulation in the broth culture of B. thuringiensis EC1.
2.2.7. Time Course Experiment for Growth, pH, Sugar Utilization and Methionine Production
Time course experiment for growth, pH, sugar utilization and methionine production by B. thuringiensis EC1 was studied. The fermentation medium consists of basal medium, maltose, 80 g; (NH4)2SO4, 10 g; yeast extract, 0.05% (w/v); peptone, 0.05% (w/v); riboflavin, 1.0 µg/ml; MgO, 0.10 µg/ml; H2O 1 L; pH 7.2. Fermentation process was carried out as previously described and growth determined turbidimetrically with a spectrophotometer at 660 nm. Methionine concentration in the broth culture was determined as previously described and sugar utilization estimated following the method described by Miller [13] .
3. Results and Discussions
The methionine producer, Bacillus thuringiensis EC1, isolated from fermented food product was found to accumulate 1.89 mg/ml methionine in submerged medium. The isolation of Bacillus organisms as methionine producers agrees with the works of other investigators [14] . However, Bacillus species are not yet known to be very active producers as species of Corynebacterium, Brevibacterium and Arthrobacter [8] [15] .
There is need for adequate aeration in microbial production of metabolites [16] . The results obtained with regard to oxygen tension (Figure 1) showed that as the liquid volume increased above 20 ml, methionine production decreased. Therefore, 20% is the optimum medium/fermenter volume ratio for methionine accumulation in submerged culture of B. thuringiensis EC1. This finding is supported by the work of Pham et al. [4] .
The size of the inoculum in a fermentation process influences the metabolite production [10] [17] . As presented in Figure 1. inoculum size of 5% enhanced methionine production by B. thuringiensis EC1. High inoculum size results in too much biomass and depletes product formation [18] and also reduces dissolved oxygen, and increases competition towards nutrient [19] . Very low inoculum size may lead to insufficient biomass and reduced metabolite formation [17] . Therefore, at a suitable inoculum size, the nutrient and oxygen levels support sufficient growth of organism and therefore enhance metabolite production [17] .
The effect of carbon sources on methionine production indicates that maltose is the carbon of choice. At 8%
Figure 1. Effect of medium/fermenter volume ratio and inoculum size on methionine production.
level, it stimulated a high methionine yield of 2.49 mg/ml (Figure 2). There is significant positive correlation (r = 0.896, P = 0.039) between maltose concentration and methionine concentration, thus as maltose concentration increases, methionine production also increases. This observation agrees with the work of Banik and Majumdar [20] , who also reported maltose as the best carbon source for methionine production. Camila et al. [21] , also observed an improved extracellular protease production by Bacillus spp. when maltose was the carbon source in the culture medium.
The effect of nitrogen source on methionine production shows that ammonium sulphate gave the highest methionine yield. Statistical analysis of the result shows that there is significant negative correlation (r = −0.965, P = 0.008) between ammonium sulphate concentration and methionine concentration, thus as ammonium sulphate concentration increases, methionine production decreases and vice versa. This according to Pham et al. [4] may be due to the sulphur content in the ammonium sulphate. Since methionine is a sulphur-containing amino acid, it is expected that more synthesis could occur as the elemental sulphur is supplied to the medium. The use of ammonium sulphate in methionine production have been reported by Kase and Nakayama [8] , Anike and Okafor [22] . Pham et al., [4] reported that methionine production is a function of nitrogen concentration. As shown in Figure 2, methionine production decreased with increase in concentration of ammonium sulphate. This decrease as suggested by Pham et al. [4] , may be attributed to the osmotic pressure exerted by high nitrogen concentration, which have adverse effect on the growth and methionine accumulation by B. thuringiensis EC1. Maximum methionine yield was observed at 1% ammonium sulphate (Figure 2) concentration.
All growth promoting substances and their mixture enhanced methionine yield by B. thuringiensis EC1 (Figure 3). The stimulatory effects of these growth promoters or their mixtures have been reported by many researchers. Carlos and Meirre [23] obtained a maximum thermostable amylase production by Bacillus spp. with a mixture of yeast extract and peptone. Moringa et al. [24] , noted an increase in methionine production by Pseudomonas spp. FMS18 with the addition of yeast extract into the culture medium. Phadatare et al. [25] , reported an enhanced protease production in Conidiobolus coronatus in the presence of yeast extract, peptone and tryptone. Ekwealor and Obeta [26] , observed an improved lysine production with the addition of casein in the culture broth of Bacillus megaterium.
Vitamins effects on methionine accumulation by B. thuringiensis EC1 (Figure 4), showed that riboflavin and thiamine HCl at 1.0 µg/ml enhanced methionine production. There is significant difference (F = 4.522 (ANOVA), P = 0.024) in the different concentrations of vitamins in relation to methionine production. The increase in methionine yield observed in this study is in line with the findings of the other workers. Tani et al. [27] , reported a high methionine yield when 200 µg/l thiamine was added to the fermentation medium in methylotrophic yeast, Candida biodinii. Sen and Chatterjee [28] , noted that thiamine HCl stimulated lysine accumula-
Figure 2. Effect of maltose and ammonium sulphate on methionine production.
Figure 3. Influence of growth-promoting factors on methionine accumulation.
tion in Micrococcus varians 2fa. Ekwealor and Obeta [29] , reported improved lysine yields in Bacillus megaterium SP 86 and Bacillus megaterium SP 76 with the addition of 10 µg/ml and 100 µg/ml of folic acid and riboflavin respectively.
Metal ions are known to play vital roles in fermentation. Most of them serve as cofactors for various enzymes or probably act as activators of enzymes involved in the synthetic steps of metabolites [3] [30] . The influence of bivalent metals on methionine production by B. thuringiensis EC1 (Figure 5), showed that Zn2+, Ba2+ and Mg2+ increased methionine yield. Zn2+ at all concentrations stimulated methionine production. This result is supported by the works of Ekwealor and Obeta [29] , who reported an improved lysine production in a broth culture of Bacillus megaterium supplemented with Zn2+. Sigel [31] , also reported the role of zinc ion in the synthesis of industrially and medically significant microbial secondary metabolites. Mg2+ at 0.1 µg/ml increased methionine ac-
Figure 4. Influence of vitamin on methionine production.
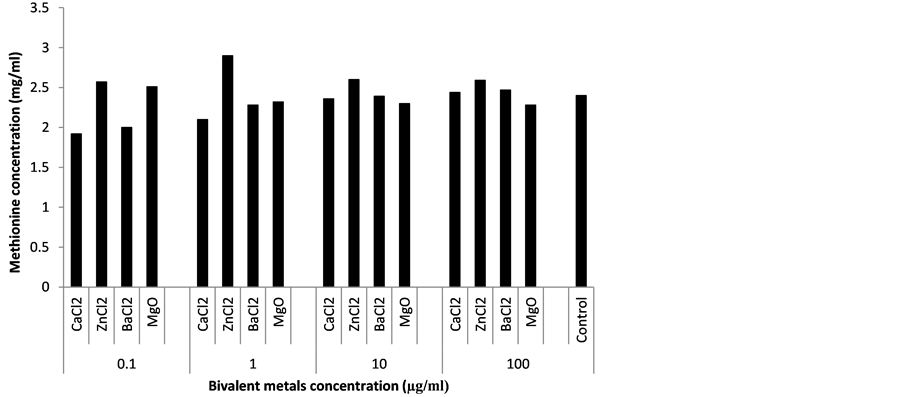
Figure 5. Influence of bivalent metals on methionine production.
cumulation by B. thuringiensis EC1 (Figure 5). The stimulatory effect of Mg2+ observed in this study is supported by the findings of El-Sayed [32] , who reported an increase in activity of L-methioninase by Aspergillus flavipes when magnesium ion was added to the fermentation medium. Similarly, Saxena and Singh [33] , observed an enhanced amylase production by magnesium ion in a solid-state fermentation of agro-industrial wastes using Bacillus spp.
Ba2+ at 10 µg/ml improved methionine accumulation by B. thuringiensis EC1 (Figure 5), indicating the possibility of this metallic ion playing a significant role in methionine production.
Time course experiment for growth, pH, sugar utilization and methionine accumulation by B. thuringiensis (Figure 6) showed a maximum methionine yield of 3.18 mg∙ml after 120 h, at a pH of 6.9 and residual sugar, 39%. The relationship between sugar consumption and methionine production (Figure 6) is similar to that observed by Murgov and Zaitseva [34] in their study on lysine production by Brevibacterium flavum 178. Ekwealor and Obeta [26] obtained a similar relationship in their work on lysine accumulation by B. megaterium.
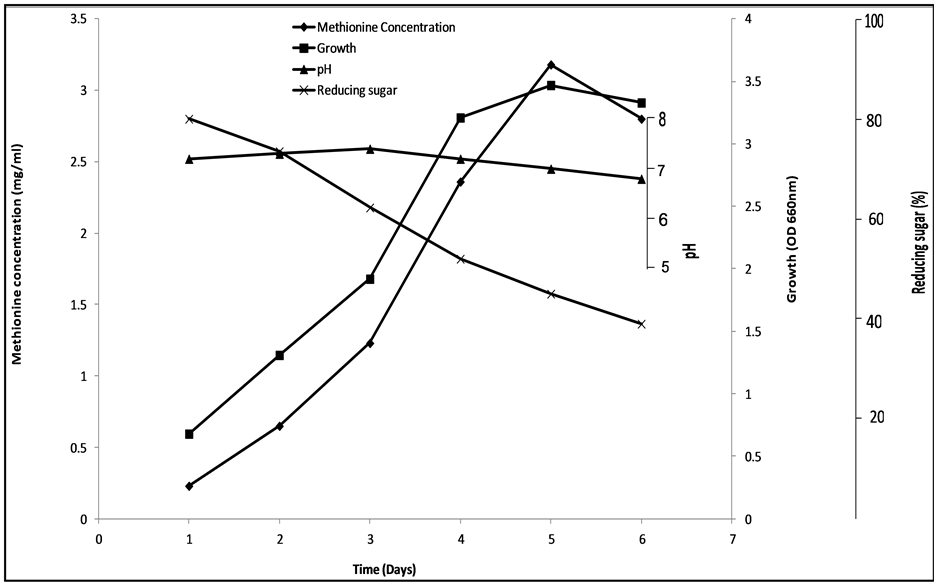
Figure 6. Time course experiment for growth, pH, sugar utilization and methionine accumulation.
4. Conclusion
A 20% medium to fermenter volume ratio containing maltose, ammonium sulphate, yeast extract, peptone, riboflavin and magnesium oxide was observed to increase methionine accumulation by B. thuringensis EC1. Therefore, this study has shown that methionine yields by B. thuringensis EC1 in submerged medium can be stimulated by improving the cultural conditions. It is also possible that strain improvement will likely bring about further increase in methionine accumulation.
Acknowledgements
We are very grateful to Tertiary Education Trust (TET) Fund, Nnamdi Azikiwe University, Awka, Nigeria, for funding this research and to Macrogen Incop., Republic of Korea for their timely help in the identification of the bacterial isolate used for this study.
NOTES
*Corresponding author.