Nephelometric Performance Evaluation of Oxidized Starch in the Treatment of Coal Washery Effluent ()
1. Introduction
Clarity of water is important towards manufacturing and products meant for human consumption. Turbidity in water is caused by suspended and colloidal matters such as clay, silts, finely divided organic and inorganic matter, plankton and other microorganisms [1] .
The efficiency of suspended solid (colloid) separation from wastewater has been achieved by the application of coagulants like alum, ferric chloride, polyelectrolyte, etc. This process highlights a water treatment mechanism that stimulates the aggregation of suspended particles to settlable flocs by the destabilization of the charged colloids thus, neutralizing the forces that keep them apart. The factors that influence coag-flocculation are, among others, temperature, pH, effluent quality, dosage and coagulant type [1] -[3] .
The coag-flocculation process using inorganic coagulants has been well documented, but less attention has been given to the use of organic coagulant of plant origin like starch. However, the high cost, post usage handling and health issues associated with the inorganic coagulants have continued to pose serious challenges. Recently, researchers have shown interest in the use of natural organic derivative like Mucuna seed [4] , Periwinkle shell [5] , etc. for the treatment of waste water. This recent interest could be traced to the environmental friendliness of biocoag-flocculants. Biocoag-flocculants are non-toxic, natural and biodegradable [6] [7] and these made their application very promising.
In present work, cassava starch provides the focus of the study. Cassava starch is a natural, non toxic and biodegradable polymer. Chemically, starch is a hemopolymer made up of basically two molecular species— amylopectin and amylase. While the amylase constitutes the inner part without any phosphorus, the amylopectin makes up the outer skin and contains combined species of phosphorus. Cassava starch is known to contain 17% of amylase and 83% amylopectin [8] . The oxidation of starch using calcium hypochlorite was employed. For effective turbidity removal, it has been found that the ratio of hypochlorite to starch should not be less than 2 and that the quantity of cassava starch per litre solution should not be more than 1 g [1] . Its application for treatment of wastewater can be extended to coal washery effluent.
Coal washery effluent wastewater has high suspended colloidal particle of mainly clay origin. It has an average initial turbidity of 23360.0NTU, pH of 2.68, TSS of 808.76 mg/l, TDS of 449.232, alkalinity of 1.0 mg/l CaCo3 etc. (see Table 1).
This work therefore, demonstrated the effective application of OSC (Oxidized Starch Coagulant) for clarification of coal washery effluent. It further investigated the effect of dosage, pH as well as the blend of OSC. It also studied the process kinetics.
2. Theoretical Background and Principles Development
Turbidity is an expression of the optical property that causes light to be scattered and absorbed rather than transmitted with no change in direction or flux level through the sample [9] .

Table 1. Results of coal wastewater effluent analysis.
Based on the work of Metcalf and Eddy [10] , the relationship between turbidity and total suspended solid is as follows:
TSS (mg/L) = (TSSf) (T)
where TSS = Total suspended solid, mg/LTSSf = factor used to convert turbidity reading to total suspended solids (mg/L).
In the work of Menkiti et al., [11] , it was shown that the coagulation rate constants could be determined by monitoring the changes in the turbidity of the coagulating liquid with time.
The time evolution of the cluster—size distribution for colloidal particles is usually described by the Smoluchowski Equation [12] .
 (1)
(1)
where  is the time dependent number concentration of n-fold cluster, t is the time, and
is the time dependent number concentration of n-fold cluster, t is the time, and 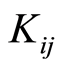 are the elements of the rate kernel which control the rate of the coagulation between an i-fold and j-fold cluster.
are the elements of the rate kernel which control the rate of the coagulation between an i-fold and j-fold cluster.
Note that in the Smoluchowski analysis, the coagulation process is approximated to be entirely controlled by Brownian diffusion and initially having mono dispersed suspension. The implication of this is that the analysis attempts to quantitatively interpret the kinetics of rapid coagulation on the basis of diffusion (Brownian) motion of which is best studied during the early part (say t < 30 min.) of coagulation process [4] [5] [13] -[15] .
According to the work of Menkiti et al. [5] , and Danov et al. [15] , it was shown that the rate of depletion of particle count (TSS or turbidity removal) can be generally be represented as
 (2)
(2)
where K is reaction rate constant, C is the concentration and  is the order of reaction.
is the order of reaction.
The generalized case of Equation (2) can be used to evaluate aggregation constant K and order of the reaction  by taking [Ln] of both sides of the Equation (2).
by taking [Ln] of both sides of the Equation (2).
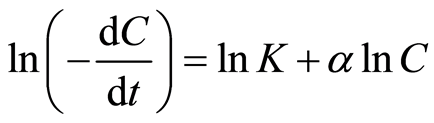 (3)
(3)
A plot of 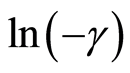 versus LnC gives a slope of
versus LnC gives a slope of  and intercept of LnK from which K is evaluated. Where
and intercept of LnK from which K is evaluated. Where
 .
.
For simple mono-dispersed aggregation model, the rate of coagulation can be found using Equations (1) and (2). The implication is that for Brownian controlled coagulation, Equations (1) and (2) are equal [5] [12] [16] .
The process of aggregation is complex, and for the complicated particles, Equation (1) can be solved exactly to give generalized Equation (4):
 (4)
(4)
where ,
,  (period), Ci = particles distribution concentration, i = 1(singlet), 2(doublet), 3(doublet).
(period), Ci = particles distribution concentration, i = 1(singlet), 2(doublet), 3(doublet).
From generalized Equation (4):
 (5)
(5)
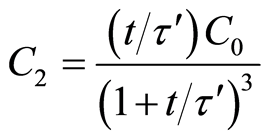 (6)
(6)
 (7)
(7)
3. Materials and Methods
3.1. Collection and Methods of Analyses of Coal Washery Effluent
The coal washery effluent wastewater was collected during rainy season from moribund coal mine site, Enugu, Nigeria. The chemical oxygen demand was determined at the Regional Water Laboratory, Enugu. Other parameters were determined at Enugu State Water Corporation Laboratory. The pH, electrical conductivity and turbidity were determined using Mettler Toledo Delta 320 pH Meter, EI Digital Conductivity Meter (model number 161) and EI Digital Turbidity Meter (model no. 337), respectively. The characterization result obtained and displayed in Table 1 was based on standard methods [9] .
3.2. Preparation of Coagulants Stock Solutions
3.2.1. Preparation of OSC
Cassava starch was obtained from garri processing mill. Garri is a local food in Nigeria produced from cassava. The sludge obtained from milled cassava tubers was allowed to stand for 24 hr. After 24 hr, the solution separated into two layers. The upper layer was decanted, while the caked bottom was dried to become starch. A mixture of 1 g of cassava starch and 2 g of calcium hypochlorite was made into a solution with 100 mL of distilled water in a 1 L beaker. Distilled water of about 55˚C was added to the solution to make it up to 1 L. The solution was stirred for 15 mins using magnetic stirrer. The starch – hypochlorite solution was charged or electrolyzed for two hours to obtain OSC by passing direct current maintained between 0.1 - 0.2 A via an adapter. The voltage measured by digital meter across the electrode stood at 10.5 volts. The solution was constantly stirred using a magnetic stirrer maintained at 10 rpm speed throughout the experiment.
3.2.2. Preparation of Alum Solution
2.5 g of aluminum sulphate was weighed and dissolved in 100 mL of distilled water and made up to a liter to give 2.5 g/L stock solution.
3.3.3. Preparation of Ferric Chloride Solution
2.0 g of FeCl3∙6H2O, was quickly weighed and dissolved in 100 mL of distilled water and made up to a liter to give 2.0 g/L stock solution
3.3. Jar Test
The conventional jar test procedure was employed. 2 min of rapid mixing at 45 rpm followed by 20 min of slow mixing at 15 rpm was adapted. The volume of wastewater used was 500 mL for all the study. After stirring, the solution was carefully poured into 500 mL cylinder and allowed to settle for 30 mins. 20 mL of the clear upper layer of the settling solution was pipetted at 2 cm depth at 5 mins interval. The turbidity of these supernatants were measured and recorded. 50, 100, 150, 200 mg/L and 250 mg/L OSC were used. Furthermore, 10, 20, 30, 40, 50 and 100, 200, 300, 400, 500 mg/L of FeCl3 and alum, respectively were used.
4. Results and Discussion
4.1. Coag-flocculation Performance Plots
Jar test was conducted to determine the coag-flocculation performance of OSC and its blends with aim of obtaining the optimum pH and dosage.
The results of coag-flocculation performance of varying dosages (50 to 250 mg/L) of OSC are presented in Figure 1. Figure 1 indicated that the minimum and the maximum removal of turbidity achieved was 98.2% for 100 mg/L OSC and 99.8% for 250 mg/L OSC, respectively.
Furthermore, it could be observed that turbidity removal increased from 98.2% at 100 mg/L to 99.8% at 250 mg/L optimum dosage. This could be attributed to coag-flocculation mechanism of charge neutralization and inter particle bridging associated with high molecular weight polymer such as starch [17] .
Coag-flocculation performances of OSC blended with FeCl3 and alum are presented in Figures 2-5. Figure 2 indicated that minimum and maximum turbidity removal were obtained for 50 mg/L FeCl3/ 250 mg/L OSC and

Figure 1. Coag-flocculation performance (%) vs. time (min) for varying Oxidized Starch dosage only.
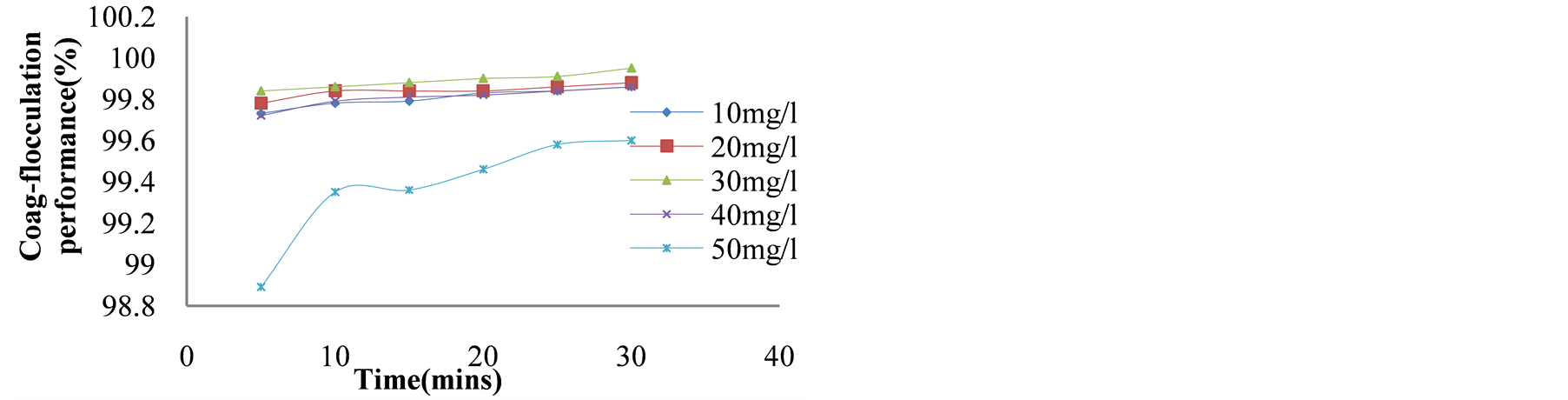
Figure 2. Coag-flocculation performance (%) vs. time (min) for varying FeCl3 dosage at constant OSC dosage of 250 mg/L.

Figure 3. Coag-flocculation performance (%) vs. time (min) for varying OSC dosage at constant FeCl3 dosage of 30 mg/L.

Figure 4. Coag-flocculation performance (%) vs. time (min) for varying Alum at constant OSC dosage of 250 mg/L.
30 mg/L FeCl3/250 mg/L OSC, at 98.9% and 99.95%, respectively. For Figure 3, minimum (98.09%) and maximum (99.9 %) removal were achieved at 50 mg/L OSC/30 mg/L FeCl3, and 250 mg/L OSC /30 mg/L FeCl3,

Figure 5. Coag-flocculation performance (%) vs. time (min) for varying oxidized starch dosage at constant alum dosage of 300 mg/l.
respectively.
Also, in Figure 4, minimum (98.6%) and maximum (99.8%), removal was obtained at 100 mg/l alum/250 mg/L OSC and 300 mg/L alum/250 mg/L OSC respectively. In Figure 5, minimum (97.69%) and maximum (99.84%), removal was achieved at 100 mg/l OSC/300 mg/L alum and 200 mg/L OSC/300 mg/L alum, respectively.
Figure 3 indicates better performance with variation of OSC dose blended with FeCl3 dose when compared with variation of OSC dose only. The turbidity removal of the blend lies between 98.2% and 99.9%. It was however, observed that this blend achieved 99.9% removal in <10 min of coag-flocculation. This could not be unconnected with the sweep floc occasioned by charge neutralization and colloids entrapment due to precipitation of hydrous iron oxide. Inter particle bridging associated with OSC polymer also contributed to this high degree of aggregation. [17] [18]
Figure 5 showed minimum and maximum removal of 97.7 and 99.8% respectively, an indication of lower performance when compared with that of OSC blended with FeCl3. This was due to low pH value and alkalinity of the wastewater. These favour the formation of soluble Al3+ instead of Al(OH)3 which enhances coag-flocculation [17] . However, the relatively high total dissolved solids (TDS) of the wastewater resulting in high ionic strength, was a prelude to the enhanced turbidity removal obtained [19] . This agrees with the understanding that at high ionic strength (shown in Table 1), introducing a coag-flocculant like alum results in the reduction of the electrostatic repulsion between colloid particles, which led to efficient aggregation [6] .
The results of coag-flocculation performance against dose were presented in Figure 6. The results indicate the effect of the coag-flocculation of OSC when blended with FeCl3 and alum. Also, it highlighted the optimum dosage and blend to be 250 mg/LOSC + 30 mg/L FeCl3.
Having obtained the optimum blend of 250 mg/LOSC + 30 mg/L FeCl3, the blend was further subjected to jar test at different pH. The results are shown in Figure 7.
Figure 7 indicated that maximum and minimum removal of 99.9% and 98% for pH value of 4.0 and 10.0, respectively, were recorded.
This clearly highlighted the effect of pH in coag-flocculation, considering the fact that pH value determines the solubility and the type of Fe2+ or Fe (OH)2 formed. These in turn determine the charge of hydrous oxide complex, which affects the coag-flocculation process [1] [17] . The pH value of 4.0 favours the precipitation of Fe(OH)2, which coagulate by combination of sweep floc, charge neutralization and inter particle bridging [17] .
Figure 8 indicated the coag-flocculation performance against pH value at different settling time. It also showed that the optimum pH of the process was 4.
4.2. Coag-flocculation Kinetics
The results of graphical representation of Equation (3) were shown in Figures 9, 10, respectively. Using these Figures, the reaction order α, coagulation rate constant K, and regression constant R2 were determined and recorded in Tables 2, 3, respectively.
The results of varying OSC dose and its blends recorded in Table 2 and Figure 9 indicate first order reaction in all doses and blend. Also, Table 2 indicates variation in the coagulation time and rate constant despite the same order of reaction. The variation of rate constant was observed to be inversely proportional to coagulation
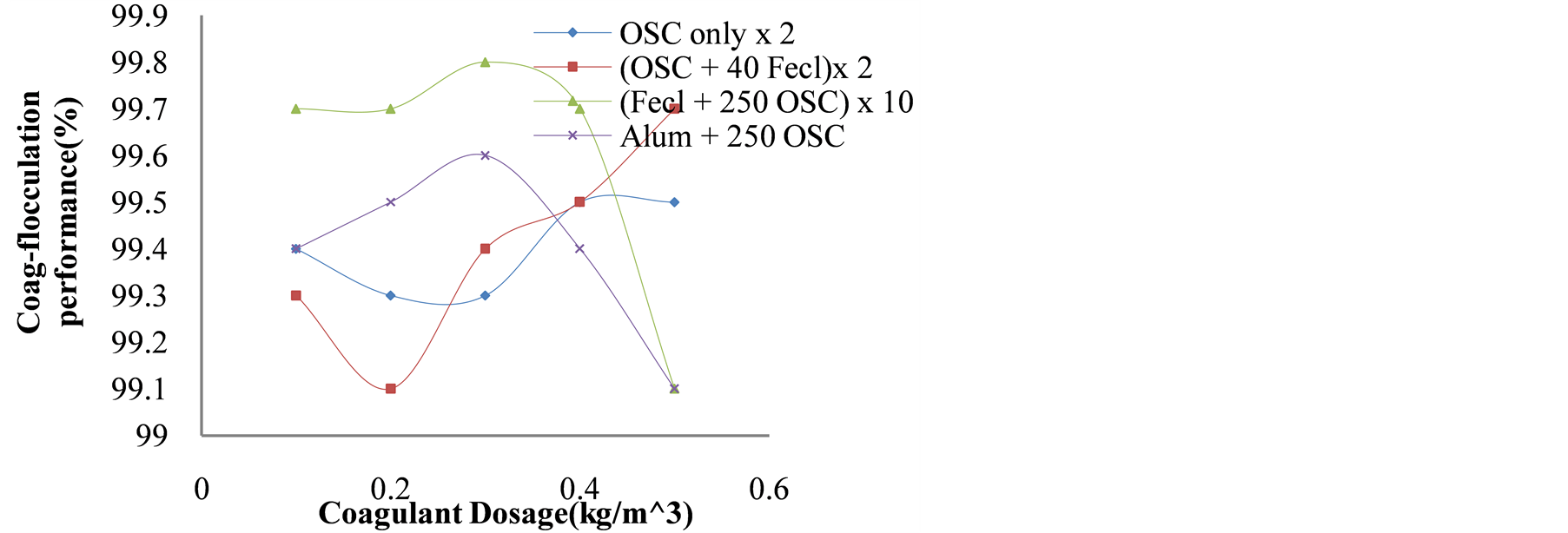
Figure 6. Coag-flocculation performance (%) vs. coagulant dosage (mg/L) for OSC and its blends after 30 minute settling time.

Figure 7. Coag-flocculation performance (%) vs. Time (min) for optimum blend of 30 mg/l FeCl3 + 250 mg/l OSC at different pH values.

Figure 8. Coag-flocculation performance (%) vs. pH for optimum blends of 30 mg/l FeCl3 + 250 mg/l OSC at different time interval. viz: Efficiency at 15, 20, 25 and 30 minutes.
time. This was invariably expected considering the relationship between the two parameters [20] . Table 2 also indicates that the lowest K of 8.933E3 and the highest τ of 235.7 s was obtained with the blend of 250 mg/ LOSC and 40 mg/L FeCl3, while the highest K of 1.345E2 and the lowest τ of 51.6 s was obtained with the blend

Figure 9.  vs.
vs.  for Oxidized Starch and its blends.
for Oxidized Starch and its blends.

Figure 10. 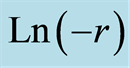 vs.
vs. 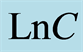 for pH variation at optimum dosage.
for pH variation at optimum dosage.

Table 2. Coa-flocculation parameter for OSC and its blends.

Table 3. Coa-flocculation parameter for optimum blend (30 mg/L FeCl3 + 250 mg/L OSC) at varying pH.
300 mg/L alum and 250 mg/LOSC [20] .
Likewise, the results of coag-flocculation performance of optimum blend at varying pH were recorded in Table 3 and Figure 10, respectively. Coagulation Kinetic studied at pH 4 and 6 proceeded by first order reaction, while that of pH 2, 8 and 10 agreed with second order reaction, respectively. The lowest K of 9.393E5 and τ of 21.3 s was recorded at pH 8, while, the highest K of 8.294E1 and  was recorded at pH 4. These are in agreement with report recorded elsewhere [4] [19] . The values of R2 for all the dosages and pH studied were sufficiently adequate. It revealed the effectiveness of Equations (2) and (3) in describing the coag-flocculation process [20] .
was recorded at pH 4. These are in agreement with report recorded elsewhere [4] [19] . The values of R2 for all the dosages and pH studied were sufficiently adequate. It revealed the effectiveness of Equations (2) and (3) in describing the coag-flocculation process [20] .
4.3. Particle Distribution Plot
Equations (5) - (7) were used to predict the particle aggregation with time for varying OSC and its blends with alum and FeCl3. Also, the particle aggregation with time for varying pH using optimum blend was studied. The particle distribution C1, C2 and C3 represent primary, secondary and tertiary particles, respectively. Figures 11-14 indicate similar trend in all the curves and represent the expected particle distribution in a typical low coagflocculation process [4] [19] [21] .
Sharp primary particle aggregation occurred in <5 minutes of the process. This suggests instant but not continued charge neutralization and destabilization mechanism with low bridging.
Further look at the Figures 11-14 indicate low doublets and triplets. This is evident considering the fact that

Figure 11. Particle distributions for varying OSC dosage from 50 mg/l to 250 mg/l for  = 86.0 s.
= 86.0 s.

Figure 12. Particle distributions for varying FeCl3 from 10 mg/l to 50 mg/l at constant OSC of 250 mg/l for  = 77.6 s.
= 77.6 s.

Figure 13. Particle distributions for varying alum from 100 mg/l to 500 mg/l at constant OSC of 200 mg/l for  = 51.6.0 s.
= 51.6.0 s.

Figure 14. Particle distributions for optimum blend (30 mg/l FeCl3 + 250 mg/l OSC) at pH 4 for  = 23.9.0 s.
= 23.9.0 s.
doublets and triplets are formed by the charge neutralization and destabilization of singlet, though instant, but not massive.
5. Conclusion
From the present study, it can be concluded that OSC has the potential of biocoag-flocculant that could be applied in large scale water treatment, especially acidic water. This is supported by high turbidity removal efficiency range of 96 ≤ E ≤ 99.9. The optimum condition was observed with the blend of 250 mg/L OSC and 30 mg/L FeCl3 at pH 4.
NOTES
*Corresponding author.