Symptomatic improvement in an acute, non-traumatic spine pain model with a combination of uridine triphosphate, cytidine monophosphate, and hydroxocobalamin ()
1. INTRODUCTION
In a previously published clinical trial by Mibielli et al., on spinal acute non-traumatic pain [1], carried out in a double-blind comparative manner, the authors randomized a sample of patients with non-traumatic lower back, hip, and neck pain into two groups: 1) group DN (nND = 40)—a combination of diclofenac-cholestyramine (equivalent to 70 mg of diclofenac) once daily + two capsules with uridine triphosphate (UTP) 1.5 mg, cytidine monophosphate (CMP) 2.5 mg, and hydroxocobalamin 1000 mcg, thrice daily, and 2) group NB (nNB = 41)—two capsules with UTP 1.5 mg, CMP 2.5 mg, and hydroxocobalamin 1000 mcg, thrice daily. Their primary objective was to evaluate the superiority of group DN over group NB, regarding pain visual analog scale (VAS), patient functionality questionnaire (PFQ), and overall efficacy evaluation by the investigating physician. Group DN showed statistically significant better results in all three evaluated endpoints.
The original study reported on the results of a doubleblind, randomized comparative trial comparing the effects of treatment with the combination of diclofenac + CMP + UTP + vitamin B12 versus the combination of only CMP + UTP + vitamin B12. Efficacy measures included a 100 mm visual analog pain scale, in which each subject placed a vertical mark along a 100 mm line from 0 mm on the left side, corresponding to “no pain” to 100 mm on the right side, corresponding to “most severe pain”. The scores were recorded as the distance from the left side of the line (0 mm) to the subject’s mark. The Patient Functionality Questionnaire was also used as an efficacy measure, in which one point was awarded to each “Yes” answer to the following questions:
1. Due to the pain in my (lower back/hips/neck):
2. I do not sleep well 3. I have to lie down more often 4. It is difficult for me to get up from my bed or a chair 5. I can stand only for a short while 6. I can walk up stairs only slowly 7. It is difficult for me to wash or dry off my whole body 8. It is difficult for me to put on my clothes 9. I can only walk short distances 10. I try to avoid picking things up from the floor 11. I have to change my posture more often 12. I cannot carry heavy things 13. I have to ask other people for assistance The authors noticed an unexpected favorable performance of group NB, regarding mean pain VAS endpoint, after publishing their paper. Seemingly, mean VAS score decreased at visit V2 (5th treatment day) in relation to pretreatment levels, and from V3 (10th treatment day) in relation to V2 and to pretreatment levels, in a self-paired fashion. This finding sounded interesting, given that the combination of UTP, CMP and hydroxocobalamin was pharmacologicaly classified as peripheral neuroregenerative, and no analgesic properties were attributed to it in medical literature. A new investigational process was then derived, in order to assess the clinical efficacy responses obtained among the subjects of group NB.
2. METHODS
We performed an analysis of previously unpublished data from investigators’ files pertaining to the pain VAS and PFQ scores of the patient group treated with the combination of UTP, CMP, and hydroxocobalamin (Group NB). The data was tabulated and statistical evaluation was performed using GraphPad Prism software version 5.04. The VAS score results from Pretreatment, V2 (after 5 days of treatment), and V3 (after 10 days of treatment) were compared using the one-way repeated measures analysis of variance (ANOVA). PFQ scores from Pretreatment, V2, and V3 were analyzed using the chi-square test.
We also performed an updated literature review based on a search of PubMed and SciElo using the search terms “uridine + cytidine + hydroxocobalamin + analgesia”, “uridine + analgesia”, “cytidine + analgesia”, “uridine + cytidine + analgesia” “nucleotides + analgesia”, “hydroxocobalamin + analgesia”, and “vitamin B12 + analgesia”.
3. RESULTS
Briefly, the demographic data of the NB group is summarized in Table 1, reported as n or mean ± standard
deviation (SD).
Mean VAS scores at pretreatment in the NB patient group were 59.44 mm (±12.43). At Visit 2, mean VAS scores decreased to 42.41 mm (±17.66), a reduction of 17.03 mm in mean total score in relation to pretreatment. At Visit 3, mean VAS scores were 30.35 mm (±17.51), representing a further mean reduction of 12.06 mm in relation to Visit 2, and a 28.29 mm reduction in relation to pretreatment values. The difference in mean VAS scores from pretreatment to V3 was statistically significant (p < 0.0001) (Figure 1).
At Pretreatment, PFQ score median was 7 points, while at Visit 2, score median decreased to 6 points and at Visit 3, score median was 4 points, indicating an improvement in functionality during the treatment period. The improvement in PFQ scores throughout the study was found to be statistically significant (χ2 = 60.74; DF = 24; p < 0.0001) (Figure 2).
4. DISCUSSION
Pyrimidinic nucleotides UTP and CMP, plus hydroxocobalamin, are available as a peripheral neuroregenerating combination. Pharmacological properties attri-

Figure 1. Self-paired evolutionary performance of mean VAS scores in NB group (pretreatment, V2, and V3).

Figure 2. Distribution of Group NB PFQ Scores ranging from 0 (best possible score) to 12 (worst possible score) at Pretreatment, Visit 2, and Visit 3.
buted to these nucleotides are: 1) stimulation of nerve cell protein synthesis, 2) stimulation of nerve cell membrane synthesis, 3) stimulation of myelin sheath synthesis, and 4) neurite sprouting through P2Y receptors stimulation [2-12]. Hydroxocobalamin pharmacodynamics in peripheral nerve insults consists in: 1) stimulation of myelin basic protein (MBP) synthesis, 2) modulation of Prion Particlecellular (PrPc) levels in Schwann cells, 3) oxidative stress protection to Schwann cells through stimulation of conversion of homocystheine to methionine, and 4) stimulation to methionine synthesis in nerve cells [13-20].
We were not able to find any evidence of direct analgesic properties of UTP, CMP and hydroxocobalamin combination in medical literature. Nevertheless, the pathophysiology of this complex symptom is partially based on the structural compromise of nerve cells, glia and connective tissue of peripheral nerve. If one assumes that the stimulus for tissue regeneration could lead to an anticipated reversion of this process, then neuropathic pain improvement parallel to peripheral nerve restoration becomes a plausible proposition. One can find a correspondence for this assumption in a study published by Goldberg et al. [21]. Actually, the present study partially reproduces the results from this former trial. In this comparative, double blinded study, the authors randomized a sample of patients with spinal and hip structural bone changes complicated with neural compression and neuropathic pain, into two groups: 1) group A-two capsules with UTP 1.5 mg, CMP 2.5 mg, and hydroxocobalamin 1000 mcg, thrice daily, and 2) group Btwo capsules of hydroxocobalamin 1000 mcg, thrice daily. Results from group A mean VAS scores and SD progression from pretreatment, V2, and V3, are detailed in a selfpaired box-plot graph (Figure 3). The difference between V3 (15) and pretreatment (58) VAS means was statistically significant (p < 0.0001).
In a clinical assessment of patients presenting with alcoholic polyneuropathy, the combination of UTP, CMP and hydroxocobalamin was found to reduce pain, paresthesia, altered motor coordination and altered vibration perception among 120 subjects treated with a 6-day intramuscular injection of: cytidine monophosphate (CMP) 5.0 mg; uridine triphosphate (UTP) 3.0 mg; hydroxocobalamin 2.0 mg; and lidocaine 20 mg (for local pain relief), followed by a 30-day treatment period of thrice daily oral ingestion of: cytidine monophosphate (CMP) 2.5 mg; uridine triphosphate (UTP) 1.5 mg; hydroxocobalamin 1.0 mg [22].
Regarding analgesic capacity itself, pharmacological properties of two pyrimidinic nucleotides were experimentaly demonstrated by Okada et al.
These investigators developed normal and neuropathic murine pain models, in order to measure analgesic effects
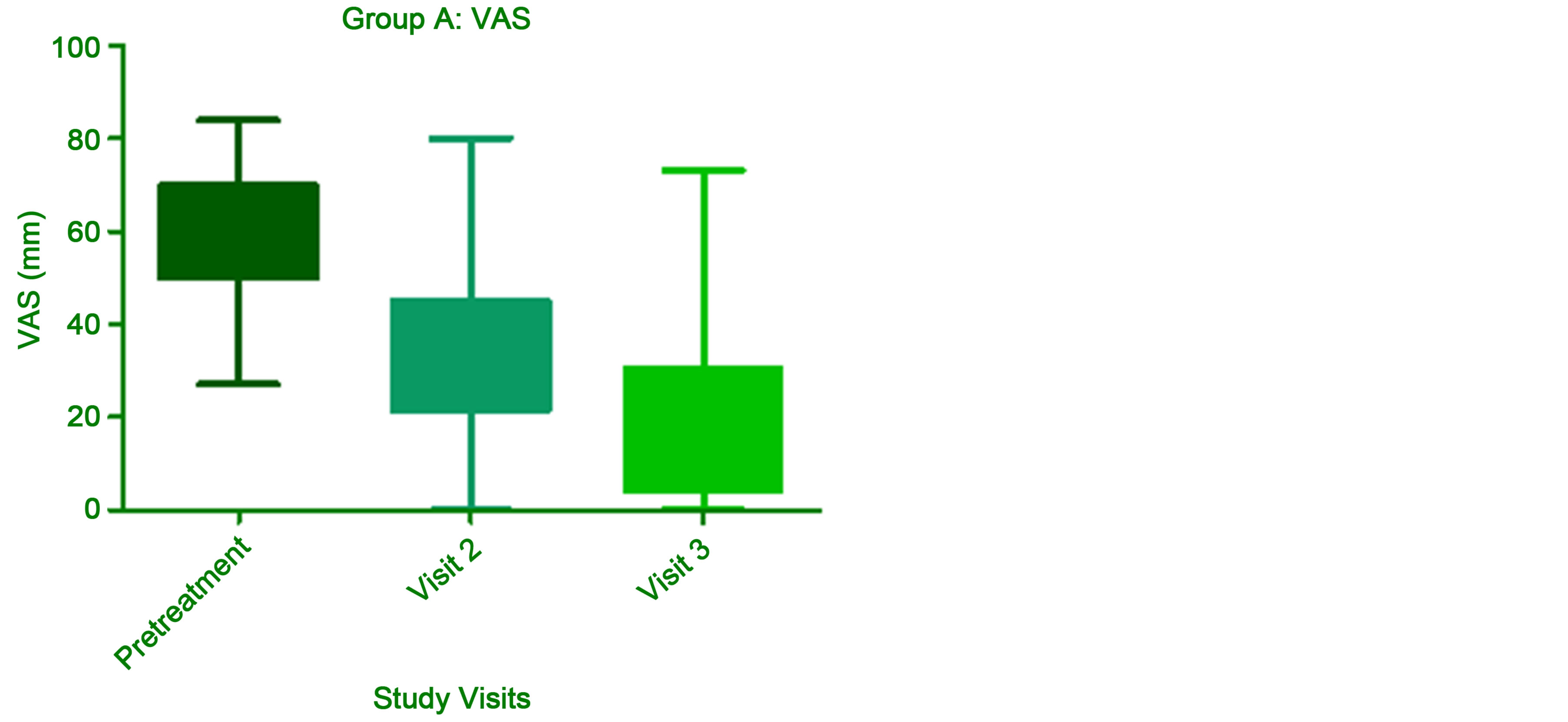
Figure 3. Self-paired evolutionary performance of mean VAS scores in group A (pretreatment, V2, and V3) (graph not present in the original paper; built on data from published tabled results).
associated to intrathecal administration of UTP and UDP, against a vehicle. These investigators found statistically significant differences favoring UTP and UDP regarding mechanical nociception, thermal nociceptive latency, and allodynia endpoints. The authors concluded that the activation of UTP-sensitive P2Y2 and/or P2Y4 receptors produces inhibitory effects on spinal pain transmission [23].
The influence of vitamin B12 in pain relief has been extensively investigated in both the clinical and preclinical settings. In 2004, Granados-Soto et al. reported that administration of vitamin B12 in rats, resulted in a dose-dependent reduction of tactile allodynia induced by spinal nerve ligation [24]. Similar results were reported in a 2008 study at doses of 6 mg/kg [25]. In a study examining the effects of high doses of vitamin B12 on nerve regeneration rates in a rat model of acrylamide neuropathy, methylcobalamin doses of 500 micrograms/kg resulted in significantly faster recovery rate in compound muscle action potentials after tibial nerve stimulation compared to a saline-treated control group [26].
Vitamin B12 has shown clinical efficacy in the treatment of diabetic neuropathy. The effects of high-dose intravenous administration of the vitamin B12 analog methylcobalamin (500 µg 3 times per week) over a 6- month period was evaluated in chronic hemodialysis patients presenting diabetic neuropathy. The authors reported reduction in pain and paresthesia along with significant improvement of ulnar motor and median nerve conduction velocities [27]. Methylcobalamin was also shown to be effective in the treatment of diabetic neuropathy at oral doses of 500 mg; treatment resulted in improvement in pain as well as somatosensory and autonomic symptoms [28,29]. Mauro et al. (2000) assessed the efficacy of intramuscular vitamin B12 in the treatment of low back pain in patients presenting mechanical or irritative lumbago, reporting that after a 2-week treatment period consisting of once-daily 1000 mg vitamin B12 injections, treated subjects experienced a significant decrease in pain and disability that was statistically superior to the placebo-treated control group [30].
The results of the above-mentioned studies indicate the following mechanisms of action for vitamin B12 activity in pain relief: promotion of nerve regeneration and/ or remyelination by accumulation of exogenous B12 [30]; because vitamin B12 may act as a methyl donor in DNA metabolism, high concentrations upregulate gene transcription, increasing protein synthesis for nerve regeneration [31]. Additionally, Takeshige et al. (1971) suggested a selective blockade of sensory nerve conduction as a mechanism of action for vitamin B12 in painful conditions [32].
5. CONCLUSION
Combination of UTP, CMP and hydroxocobalamin seems to present analgesic effects in peripheral neuropathic pain secondary to spine structural disorders, in mediumterm use. These properties can be attributed to a complex pharmacodynamics involving indirect (nerve cell protein synthesis, myelin sheath synthesis, MPB synthesis, etc), as well as direct (P2Y receptors stimulation) mechanisms. Further studies are needed to corroborate these findings, as well as to better establish the temporal relationship between the combination administration and significant levels of pain improvement.