1. Introduction
Itraconazole (ITZ) is a triazole antifungal used to treat invasive fungal infections. Approved in 1992, ITZ disrupts the conversion of ergosterol to lanosterol via inhibition of the enzyme 14-α-demethylase. In the late 1990s, several case reports of cardiac toxicity associated with ITZ inspired a review of the Adverse Event Reporting System (AERS) database. From 1992-2001, 94 cases of cardiac toxicity were identified from the post-marketing reports [1]. Of these, 58 were classified as possibly associated with ITZ, which led to an FDA-issued black box warning in 2001. The ITZ package labeling now includes a warning against use in patients with a history of heart failure.
The objective of our study was to elucidate the cardiac effect of azole antifungals. This research was planned as an ex vivo assessment of histologic damage to the heart or blood vessels, changes in contractility, changes in rate or rhythm and alteration in pressure in an attempt to identify an etiology for changes in cardiac function.
2. Methods
2.1. Model
Male Harlan Sprague-Dawley rats from 275 - 299 g were housed within the animal facility at the University of Mississippi Medical Center and supplied with standard food and water. All animal care and use procedures were approved by our institution’s animal care and use committee and were conducted in accordance with USDA Regulations, 9 CFR Parts 1, 2, and 3 and the Guide for the Care and Use of Laboratory Animals [2,3]. Using a modified Langendorff procedure similar to that used by Liu et al., rats were anesthetized with aerosolized isofluorane [4]. Thoracotomy was performed during anesthesia and the ascending aorta exposed. After heparinization (~500 IU/kg) through the inferior vena cava, the aorta was cut and cannulated at the descending border of the aortic genu using a 16 gauge stainless needle connected to a perfusion system. Retrograde perfusion of the heart with Krebs-Henseleit solution was started immediately at a constant flow rate of 25 mL/min. During perfusion, the heart was excised and transferred into a water-jacketed chamber (Radnoti; Monrovia, CA), and immediately, a latex balloon (3 × 7 mm, 0.03 ml, Radnoti) was inserted into the left ventricle via the pulmonary artery. Prior to insertion, the balloon was filled with physiological saline and connected to a pressure transducer via polyethylene tubing (PE-90). Changes in left ventricular pressure (LVP), its first derivative (dP/dt) and root mean square (RMS) of LVP over time, heart rate
(HR), and aortic perfusion pressure (PP) were recorded through an A/D convertor (PowerLab/16sp, AD Instruments; Colorado Springs, CO) by an IBM compatible PC with LabChart for Windows v6.1.1.
The Krebs-Henseleit buffer (KHB), pH 7.4, consisted of 118.5 mM NaCl, 4.69 mM KCl, 1.18 mM KH2PO4, 1.16 mM MgSO4, 2.52 mM CaCl2, 25.88 mM NaHCO3, and 5.5 mM glucose that was filtered using a 0.22 µm cellulose acetate bottle-top filter (Corning Incorporated; Corning, NY) before use. During studies, KHB solution was aerated with 95% O2-5% CO2 and warmed to 37˚C using a water-jacketed glass chamber (Radnoti) supplied by a standard water bath pump. Test agents were infused via syringe pump and diluted with buffer solution proximal to the aortic heart catheter using a Y-site port.
In multiple random crossover antifungal dose ranging studies, hearts were perfused with an azole antifungal including; ITZ (Sporanox® for injection; Ortho Biotech, NY), fluconazole (Diflucan®; Pfizer, Inc., NY), voriconazole (Vfend®; Pfizer, Inc., NY) and ketoconazole (Sigma Chemical; St. Louis, MO) at one, four, and ten times therapeutic concentrations in ascending order, for periods of 5 minutes each. Amphotericin B (AmB (Amphotericin B for injection USPTM; X-Gen Pharmaceutical Inc., NY)) was included as a positive antifungal control. Cyclodextrins (beta-sulfobutyl ether (SBECD) and hydroxypropyl beta (HPßCD); Sigma Chemical, St. Louis, MO), and KHB were used as negative controls. Exposures to dose-ranged histamine (Sigma Chemical, St. Louis, MO) or oligomycin (Sigma Chemical, St. Louis, MO) administered to the ex vivo heart for periods of 5 minutes were assessed in attempts to elucidate the mechanism of azole dysfunction. Each experiment was performed in at least triplicate.
Pharmacologic agent exposure associated with nonfatal reductions in myocardial activity (contractility) during a 5 minute exposure was the primary concentration selected and assessed in further studies. Prolonged exposure (>5 minutes) to ITZ was utilized in a noncrossover design. These studies focused on the time required for a myocardial event to occur, specifically focusing on a 50% and 80% decrease in contractility.
2.2. Biomarkers
A group of biochemical biomarkers were selected and screened as etiologic indicators during 5 minute exposures only. All assays were performed using the Roche c 501 or Roche 601 analyzer (Roche Diagnostics, Indianapolis, IN, USA) of the Cobas® 6000 series. The instruments were operated using Roche Diagnostics software version 7277112-04-02 and Roche Cobas® reagents were used to measure alanine aminotransferase, aspartate aminotransferase, creatine kinase, creatine kinase-MB, lactate, N-terminal pro B-type natriuretic peptide, magnesium, lactate dehydrogenase, sodium, potassium, and chloride.
2.3. Electrocardiography (ECG)
Bioelectrical signals were recorded using MLA2540 3 Lead Shielded Bio Amp Cable electrodes connected to a biological FE135 Dual Bio Amp amplifier (AD Instruments). The ECG like recordings were obtained in select 5 minute experiments using electrodes connected to the apex-left ventricle (Positive), right atria (Negative) and aorta (Ground).
2.4. Histochemistry
Post-exposure, hearts were dissected into sections and evaluated macroscopically and microscopically. Histochemistry was performed on 4 µm paraffin-embedded rat cardiac tissue sections prepared from samples after completion of 5 minutes single antifungal experiments. Endogenous peroxidase (3% H2O2/PBS for 30 min] & endogenous biotin [Biotin Blocking System™ (DAKO Corp, Carpinteria, CA)] were used to treat sections. Sections were stained with hematoxylin Gill2 (Sigma, St. Louis, MO) and mounted in Cytoseal 60 (Steohens Scientific, Kalamazoo, MI). Slides were reviewed, by a single blinded, pathologist.
2.5. Transmission Electron Micrograph
Tissue sections were immersion fixed in 2% buffered glutaraldehyde, post-fixed in 1% osmium tetroxide, stained en bloc with 2% uranyl acetate, and embedded in Embed 812 for evaluation by electron microscopy (TEM) on a Zeiss EM 906 transmission electron microscope. Sections post-stained with lead citrate were photographed to obtain longitudinally-sectioned cardio-myofibers from selected experiments.
2.6. Mitochondrial Respiration
Mitochondria were isolated from freshly harvested livers of ~350 - 400 g Harlan Sprague-Dawley rats by standard methods. Mitochondrial respiration was measured polarographically as O2 consumption using a Clark-type electrode in a 1.5 mL stirred vessel maintained at 25˚C in a reaction buffer. Either 2 mM glutamate and malate or 2 mM succinate were used as substrates. State 2 and State 3 respiration were measured in the absence and presence of 0.25 mM ADP, respectively. State 4 was assessed following the addition of 1.2 μM oligomycin, while uncoupled respiration was achieved by the addition of 4 μM FCCP.
2.7. Data Analysis
A one-tailed Student’s t-test was used to compare mean overall decreases in contractility between ITZ and control. Contractility, defined as the strength of contraction of the left ventricle, was calculated using RMS of LVP over time. The primary outcome measure was percent change in contractility, which was calculated by dividing the RMS at the end of drug infusion by the RMS at the beginning of drug infusion or maximal effect times. An alpha (0.05) and beta (0.2) were selected for comparisons. A quadratic best fit equation was determined for the relationship of effect (change in contractility) and ITZ dose (SigmaPlot; Systat Software, Inc., San Jose, CA) along with decrease in mitochondrial oxygen consumption by dose. All other data are represented demonstratively.
3. Results
3.1. Langendorff Model
The Langendorff model appears to be a tremendously stable model for assessments of heart contractility. In negative control studies, KHB was associated with −0.594% ± 4.07% change in contractility at periods of observation up to 6 - 8 hours. Further observation was deemed un-necessary due to the planned length of studies. Amphotericin B (positive control) had >50% reduction in contractility in dose ranging studies at exposures equal to and beyond pharmacologic for humans. Amphotericin B at concentrations of 5 and 10 ug/mL were associated with decreases in contractility of 37.5% ± 28.9% and 97.5% ± 2.1%, respectively. Amphotericin was specifically associated with arrhythmias, which began prior to decreases in contractility. Based on these findings, a 10% change in contractility was arbitrarily defined as an “event” for assessments of change in all further studies.
After 5 minute infusions, ITZ was associated with decreases in contractility. Low concentrations (<2 ug/mL) were associated with no obvious change in contractility, while concentrations greater than 2 ug/mL were associated with decreases in contractility up to 22.2% ± 15.7% (p < 0.05) (Figure 1). No significant changes in heart rate were observed with any dose. The dose response curve modeled to a 2 parameter sigmoidal equation demonstrated a 0.92 correlation coefficient (Figure 1). Data also fit a linear model (Y = 2.46X + 1.1; R2 = 0.89), however, one would anticipate a pharmacologic event to be sigmoidal. At exposures up to ten times the therapeutic concentrations, fluconazole 1 - 80 ug/mL, voriconazole 0.04 - 40 ug/mL and ketoconazole 0.13 - 33 ug/mL had no effect, defined as an impact greater than 10% change in contractility. The later azoles were not carried forward in other Langendorff experiments due to the lack of effect.

Figure 1. Itraconazole dose response curve. Five (5) minutes exposures to ● – itraconazole (Jansen Pharmaceuticals). Correlation associated with a 2 parameter sigmoidal curve was 0.918. Routinely achievable serum concentrations blocked in hatch box. Data shown as percent change from baseline ± standard deviation.
Prolonged infusions of ITZ (0.2 - 2.5 ug/mL) were used to approximate effective therapeutic dose in humans at steady state. This exposure also appears to approximate the effective exposure to induce a 25% reduction in contractility compared to maximal reductions (ED25%). The contractility pressure wave amplitude decreased by 11% ± 0.17% while there was no appreciable change in wave frequency or heart rate. Aortic perfusion pressure increased by 25% ± 6.9% over baseline, reaching maximum pressure at approximately 8 minutes during prolonged infusions. At three specific exposures (0.2, 1.0 and 2.5 μg/mL), 50% decreases in contractility occurred at 129 + 135, 20.1 + 6.6 and 6.5 + 1.1 minutes respectively. An 80% reduction in contractility was achieved at 175 + 140, 31.7 + 10.3, and 8 + 1.1 minutes, respectively (Figure 2). Again, a dose response curve was modeled to a sigmoidal curve (R2 = 0.91). However, in a partial dose-ranging model that represents the therapeutic range, an exponential decay model exhibited a better fit (R2 = 0.996) (Figure 2).
Cyclodextrins were used as negative controls. Betacyclodextrin sulfobutyl ether (SBECD) used to formulate voriconazole did not impact contractility at exposures of 20 to 8000 μg/mL. This SBECD concentration (16:1) would correspond to a voriconazole exposure of 1.25 - 500 μg/mL. However, we identified a decrease in contractility associated with hydroxypropyl beta (HPßCD), the cyclodextran used in a 40:1 ratio to solubilize ITZ. HPßCD tested from 4 to 4800 μg/mL, corresponding to ITZ amounts of 0.1 to 120 ug/mL. Contractility decreased nearly 50% at exposures greater than 2400 μg/mL. A person receiving an azole antifungal like ITZ would
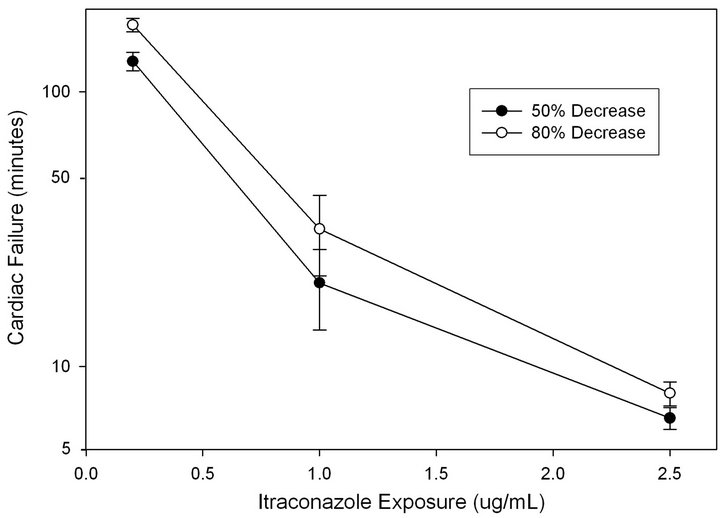
Figure 2. Prolonged infusion exposure to itraconazole (Jansen Pharmaceuticals). Time to achieve a 50% reduction in contractility (●) and 80% reduction in contractility (○) represented. Correlation associated with a 2 parameter sigmoidal curve displayed an R2 > 0.9 and exponential decay at the end of the dose effect (Y = 203e–2.3X) was 0.996. Data shown as time in minutes ± standard deviation.
not be expected to be exposed to these HPßCD concentrations during steady state conditions.
Histamine was tested at concentrations of 0.543 nM to 54,300 nM. Multiple replicates at these concentrations were associated with an average 5.8% ± 21.3% decrease in contractility. Oligomycin was tested at exposure concentrations of 0.5 ug/mL to 125 ug/mL.
At concentrations less than 10 ug/mL, an average decrease in contractility of 8.6% ± 6.5% was seen. At concentrations of 50 ug/mL and higher, an average decrease in contractility of 28.2% ± 28.4% was observed.
3.2. Biomarkers and ECG
Cardiac outflow was measured before, during and after administration with KHB, amphotericin, and each azole. In randomly selected experiments, no obvious, consistent changes in any biomarker were observed. In addition, bioelectrical signals recorded during studies did not help elucidate a mechanism of ITZ cardiotoxicity. ECG changes were only evident after changes in contractile function developed and all were temporally distal to the primary decrease in heart function. Events (slowing heart rate and premature ventricular contractions) all appeared secondary to the failing heat.
3.3. Histochemistry
The experimental hearts were dissected transversely and sent for histopathological examination after ITZ exposure, for comparison to control hearts that had been exposed to only KHB buffer. On gross examination, no pathological differences were observed between the experimental and control hearts. There was no evidence of inflammation, infiltration, ischemia, band necrosis, or hypertrophy at 200×. Although, it appeared with further digital magnification (300% of the 200×), that the experimental hearts had a loss of integrity in the cardiac myocytes (punch hole appearance).
Transmission electron micrograph revealed evidence of changes in mitochondrial structure. Ultrastructural features evident on all images include sarcomeres with thick and thin membranes, Z-discs and mitochondria. Changes in the mitochondria treated with ITZ were suggestive of altered mitochondrial function (Figure 3). To lesser extent than oligomycin, ITZ exposure was associated with mitochondrial swelling and distortion of mitochondrial cristae. Based on this, an assessment of mitochondrial function was conducted.
3.4. Mitochondrial Oxidative Respiration
Isolated liver mitochondria were used to measure respiration driven by glutamate/malate or succinate in the presence of ADP (State 3 respiration). Inhibition of oxygen consumption by intraconazole was observed. Itraconazole inhibited O2 consumption driven by glutamate/malate or succinate or both by approximately 15% at 9 μg/ml and up to 18% at 99 μg/ml (Figure 4). In comparison, oligomycin at 4.1 μg/ml (4.9 μM) inhibited O2 consumption by 57%. Interestingly, the addition of the uncoupler FCCP resulted in a 178% - 236% increase in O2 consumption when added to the mitochondria after ITRA. These results are consistent with two possible mechanisms of inhibition. Itraconazole could partially inhibit respiratory electron transfer and the remaining activity remains sensitive to the membrane potential. Alternatively, the relief of inhibition by uncoupler may

Figure 3. Transmission electron microscope (TEM) images of cardiac tissue after exposure to (A) KHB negative control; (B) Oligomycin positive control; (C) Itraconazole.
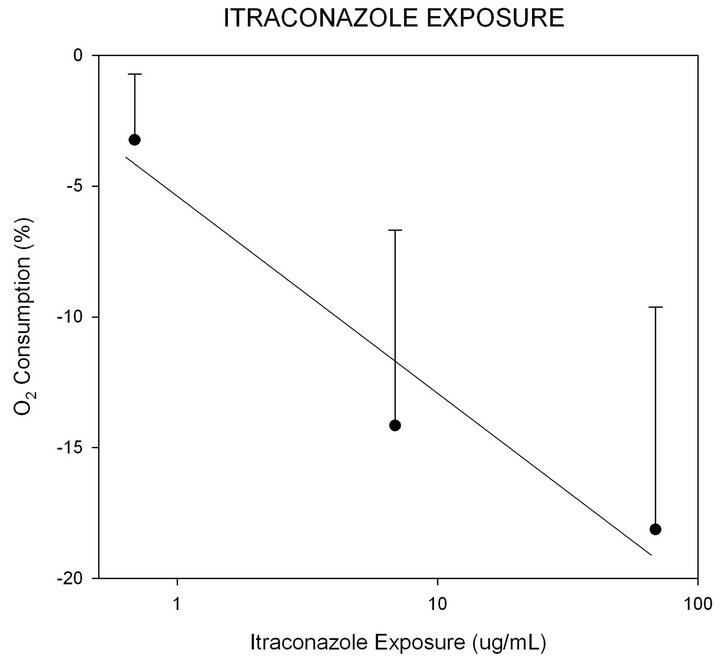
Figure 4. Change in oxygen consumption with increasing ITZ doses represented on a logarithm scale. Data shown as percent change from baseline mitochondrial oxygen use ± standard deviation. Correlation coefficient associated with linear regression (Y = 21.7X + 8.6) was 0.76.
indicate the inhibition of mitochondrial ATP synthase by the antifungal (or possibly inhibition of the ATP-ADP translocator of the inner mitochondrial membrane).
4. Discussion
Cardiac-associated adverse events attributed to antifungals have been recognized and are reported for polyenes and azoles. Amphotericin B is known to cause negative chronotropic effects and abnormal conductance [5], along with dilated cardiomyopathy [6]. Clotrimazole has been reported to inhibit calcium channels and cause vasodilation [7-9], while econazole has been reported to inhibit contractions [10]. However, few agents are labeled as causing cardiomyopathy and only preliminary data on ITZ were available to guide dosing and risk prevention [1].
In the 2001 revised ITZ package labeling, the manufacturer states that dose related negative inotropic effects were observed in anesthetized dog studies, while “transient, asymptomatic decreases in left ventricular ejection fraction were observed” in human volunteers [11,12]. The manufacturer’s packaging material reports a postmarketing surveillance study that identified this reaction most frequently in doses greater than 400 mg/d. This information is not available in the published literature. The FDA advisory was generated associated with 58 reported cases. The report noted 48% of affected patients required hospitalization and suffered a 22% mortality [1]. Since this report, four additional case reports have identified an equal number of possibly related events [13-17]. The four main classifications of cardiomyopathy include dilated, restrictive, hypertrophic, and unclassified [18]. Proposed mechanisms of drug-induced cardiomyopathies include direct toxic effects, modulation of cardiac gene expression, apoptosis, oxidative stress/free radical generation, neurohormonal activation, altered calcium homeostasis, alterations in mitochondrial oxidative function, and, alterations in myocyte protein synthesis/function [19]. Available studies of other antifungal agents demonstrate cardiac effects through vasodilation [9], altered potassium and calcium uptake [8], changes in contractility [10], changes in conductance [7], and arrhythmogenic activity [20]. In an effort to explore these mechanisms of toxicity, we systematically tested the proposed mechanisms discussed.
Alterations in mitochondrial oxidative function appears to be a logical explanation of this observed pathogenesis. Previous investigators have demonstrated that miconzole and ketoconazole inhibit mitochondrial oxidative phosphorylation [21,22]. Ketoconazole was able to inhibit respiratory function at concentrations lower than other azole (miconazole, fluconazole). Specifically, miconazole and ketoconazole were able to uncouple or inhibit glutamate/malate and succinate mediated electron transport. ITZ was not tested in these published studies. However, our observations would support these results. In fact, ITZ 99 μM (~70 μg/mL) was associated with nearly a 20% drop in O2 consumption. Ketoconazole at 500 uM was associated with a 50% decrease in the published studies. Rodriguez & Acosta (1996) report that 32 μM ketoconazole causes 50% inhibition using glutamate/malate while the 500 μM value is their IC50 using succinate. Therefore, they concluded that Complex I was the primary target of ketoconazole inhibition [21]. This could lead someone to hypothesize that ITZ was more potent an inhibitor of this mitochondrial function. However, one needs to reconcile the fact that serum concentrations above 20 μg/mL are usually not achieved in reports of clinically significant heart failure. These data may suggest more than one mechanism exists.
In order to control for damage to the heart that may be caused by the trauma of the procedure, KHB was infused into a sham heart, following the same procedure as test agents. The fact that no effect was seen over several hours implies that any damage seen in the experimental groups was due to a drug effect, not a procedural significant effect. Cyclodextrin is a solubilizing agent used in the parenteral formulation of ITZ; the lack of effect demonstrates that this solubilizing agent is not responsible for the significant decline in cardiac contractility caused by ITZ.
There are reports of cardiac dysfunction in animals post-anesthesia. We used 3.5% isoflurane for all our anesthetic procedures. In C57BL/6 female mice anesthetized with isoflurane at concentrations ranging from 0.5% to 5%, isoflurane caused a reduction of left ventricular function. In man, these agents are used in low doses in combination with other anesthetics. These low doses may be the reason why decreased cardiac output has not been commonly reported in man [23]. Our sham experiments should control for any anesthetic associated effects.
Effects of ITZ > 2 μg/mL on contractility were seen within 5 minutes of exposure, demonstrating that the effect is acute and not due to the metabolite of ITZ, hydroxy-ITZ, that will not form this rapidly. Decreases in contractility were seen within 10 - 30 minutes of administration at lower concentrations of ITZ, suggesting a possible cumulative relationship between degree of exposure and effect on the heart.
Therapeutic concentrations of ITZ in the human body range from 0.28 to 2.9 μg/mL, based on route and administration [11]. The concentrations used in this study, from 0.5 to 10 μg/mL, are achievable in humans by oral administration and parenterally, especially when administered through a central line. The reduction in contractility seen even at therapeutic exposures has implications for the clinical use and monitoring of ITZ when treating invasive fungal infections.
In summary, clinically significant cases of ITZ associated cardiac dysfunction have been reported in the literature. Based on our ex vivo and in vitro studies, we conclude that a causal role for ITZ in the reported cardiac dysfunction is probable. Our data also suggest that ITZ inhibits mitochondrial oxidative phosphorylation in the highly ATP dependent myocardial tissue in a dose dependent manner. Further studies are necessary to resolve the mechanism of inhibition.
NOTES