Protective Effects of Flavonoid Baicalein against Menadione-Induced Damage in SK-N-MC Cells ()
1. Introduction
The key precept of the oxidative stress theory of aging is that senescence-related loss of function is due to the progressive and irreparable accrual of molecular oxidative damage which is brought about by powerful pro-oxidant species including reactive oxygen species (ROS) [1,2]. ROS include a broad range of partially reduced metabolites of oxygen (e.g. superoxide, hydrogen peroxide and hydroxyl radical) having higher reactivity than molecular oxygen [3]. Their raison d’être remains unclear. Putative explanations for their occurrence range from inadvertent by-products of aerobic metabolism to highly regulated and intricate signaling mechanisms [4]. Free radical or oxidative stress theory of aging was first proclaimed by Denham Harman demonstrating the role of oxidative species in aging process acceleration and cell death [5,6]. This theory can explain many of the senescent changes including accumulation of brown-yellowelectron-dense, autofluorescent bodies in cells called lipofuscin pigments or age pigments [5,7,8]. Correlation of lipofuscin with aging is not only because the amount of lipofuscin elevates with age, but also, more significantly because the rate of lipofuscin accumulation negatively correlates to longevity. High consumption of oxygen via brain makes it susceptible to oxidative damage [9,10]. Reactive oxygen species which are generated by mitochondria through different ways, diffuse into lysosomes which encompass a variety of macromolecules under degradation as well as redox-active low molecular iron which would be released from different sorts of metalloproteins. Based on Fenton reaction, hydroxyl radical can be generated through the reaction of hydrogen peroxide with iron, bringing about the cross-linking of adjacent macromolecules and resultant lipofuscin formation [7,8,10-12]. There is a debate on the function of lipofuscin pigments formed during exposure of cells to oxidative agents. Some researchers believe that lipofuscin formation does not have any serious effects on normal function of cells. On the contrary, some scientists state that although lipofuscin cannot react directly with extralysosomal constituents because of the lysosomal membrane, the high content of iron within lipofuscin granules may promote generation of ROS, sensitizing cells to oxidative injury through lysosomal destabilization. Destabilization of lysosomal membrane results in leaking of hydrolytic enzymes into the cytosol. Hence, oxidative species and redox-active transition metals homeostasis impairments which facilitate further formation of active and hazardous reactive oxygen species might be two main characteristics of age-related neurodegenerative diseases [13].
Human’s aspiration for greater longevity has long been a strong motivation for a lot of studies in the field of aging and age-related disorders. Escalating body of evidence implies that lifestyle factors, and specially the diet, may counteract oxidative damage [2,14]. Dietary flavonoids with blood-brain barrier ability were shown to have potential anti-aging and brain-protective activities [5, 15-18]. Baicalein (5,6,7-trihydroxy-2-phenyl-4H-1-benzopyran-4-one), one of the naturally occurring flavonoids in Scutellaria baicalensis GEORGI known as “Huang qin” in China and “Ogon” in Japan, is prescribed for oxidative stress-related diseases [19]. Numerous studies have shown that baicalein protects neurons from oxidative damage via multiple bio-effects ranging from classic radical scavenging activities to modulation of signaling pathways involved in stress-associated diseases. Moreover, recent studies have denoted that baicalein mitigates formation of hydroxyl radical through its iron-binding (anti-Fenton) and strong chelation properties [20-23].
In this study, we scrutinize the effect of baicalein on menadione (superoxide anion generator)-induced lipofuscin formation in human neuroblastoma SK-N-MC cell line to comprehend the mechanism by which baicalein protect SK-N-MC cells against oxidative damages.
2. Materials and Methods
2.1. Materials
The cell culture medium (RPMI-1640), penicillin-streptomycin and fetal bovine serum (FBS) were purchased from Gibco BRL (Life technology, Paisely, Scotland). The culture plates were purchased from Nunc (Brand products, Denmark). dimethyl sulfoxide (DMSO), FeCl3 and KMnO4 were obtained from Merck (Darmstadt, Germany). Ethidium bromide, acridine orange, Baicalein and Triton X-100 were purchased from Pharmacia LKB Biotechnology (Sweden). MTT [3-(4,5-dimethyl tiazol-2, 5-diphenyl tetrazolium bromide], phenylmethylsulphonyl fluoride (PMSF), leupeptin, pepstatin, aprotinin, monochlorobimane (mBCL), dithionitrobenzoic acid (DTNB)GSH, ascorbic acid, ferrozine and pan-caspase inhibitor (Z-VAD-fmk) were purchased from Sigma Chem. Co. (Germany). 2’,7’-dichlorofluorescein diacetate (DCFHDA) was obtained from Molecular Probe (Eugene, Oregon, USA). Ethylenediaminetetraacetic acid (EDTA) was from Aldrich (Germany). Human SK-NMC neuroblastoma cells were obtained from Pasteur Institute (Tehran, Iran). All antibodies including anti-Bax, anti-Bcl-2, anticleaved caspase-9, anti-tubulin and mouse/rabbit horseradish peroxidase-conjugated second-dary antibodies were purchased from Biosource (Nivelles, Belgium). Chemiluminescence detection system was purchased from Amersham-Pharmacia (Piscataway, NJ, USA).
2.2. Cell Culture
Human neuroblastoma cell line SK-N-MC was cultured in RPMI-1640 medium supplemented with FBS (10%, v/v), streptomycin (100 μg/ml) and penicillin (100 U/ml) and incubated in 5% CO humidified atmosphere at 37˚C. To induce oxidative stress, menadione was freshly prepared from a stock solutions (10 mM), prior to each experiment. Menadione and baicalein were dissolved in a minimum amount of dimethyl Sulfoxide (DMSO) and then diluted with the culture medium to the desired concentration. The concentration of DMSO in the culture medium kept lower than 0.1% and the control cells were treated with the vehicle solution containing the same amount of DMSO.
2.3. Determination of Cell Viability
Cell viability was assessed by the 3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide (MTT) reduction assay. Viable cells with active mitochondria reduce the yellow tetrazolium salt MTT giving dark blue water insoluble formazan crystals. To perform the assay for evaluation of the cytoprotective effects of baicalein and caspase inhibitor (z-VAD-fmk) on menadione-treated SK-N-MC cells, SK-N-MC cells were suspended in medium and seeded at a density of 5 × 104 cells/well in 96 well plates for a day. Cells were pretreated with various concentrations of baicalein (10, 20, 40, 50 µM) and pancaspase inhibitor (50 µM) and then treated with menadione (35 µM) for additional 24 h at 37˚C. MTT was dissolved at a concentration of 5 mg/ml in PBS and stored at 4˚C, protected from light and tightly capped. After incubation, cells were treated with the 10 µl MTT solution for 4 h. Then, the medium was removed and 200 µl DMSO was added to each well. The formazan dye crystals were solubilized in 30 min, and absorbance was measured at 570 nm using an ELISA reader (Exert 96, Asys Hitch, Ec Austria). Results were expressed as the percentage of MTT reduction, assuming that the absorbance of the control cells was 100%.
2.4. Measurement of Intracellular ROS
Oxidation of 2’,7’-dichlorofluorescein diacetate (DCFHDA) to fluorescent DCF is taken as an index of overall oxidative stress in biological system according to LeBel method [24]. Cells were pre-treated with various concentrations of baicalein (10, 20, 40 μM) and 50 µM caspase inhibitor for 3 h followed by menadione treatment (35 µM) for 12 h at 37˚C. Then the cells were incubated with 10 μM DCFH-DA for 1 h followed by washing twice with phosphate buffer saline and suspension in the same buffer. Finally, the fluorescent intensity was monitored using a varian-spectrofluorometer with excitation and emission wavelength of 485 and 530 nm, respectively.
2.5. Determination of Lipid Peroxidation
Malondialdehyde (MDA) levels were measured by the double heating method [25]. The method is based on spectrophotometric measurement of the purple color generated by the reaction of thiobarbituric acid (TBA) with MDA. Briefly, 0.5 ml of cell lysate was mixed with 2.5 ml of trichloroacetic acid (TCA, 10%, w/v) solution followed by boiling in a water bath at 95˚C for 15 min. After cooling to room temperature, the samples were centrifuged at 3000 rpm for 10 min and 2 ml of each sample supernatant was transferred to a test tube containing 1 ml of TBA solution (0.67% w/v). Each tube was then placed in a boiling water bath for 15 min. After cooling to room temperature, the absorbance was measured at 532 nm with respect to the blank solution. The protein concentration was determined by Lowry’s method [26]. The concentration of MDA was calculated based on the absorbance coefficient of the TBA-MDA complex (ε = 1.56 × 105 cm−1·M−1) and it was expressed as nmol/ mg of protein.
2.6. Determination of Protein Carbonyl Formation
The assessment of protein carbonyl content is a widelyused marker for oxidative protein modification. Protein carbonyls (PCOs) were measured using Reznick and Packer method [27]. Briefly, 1 ml of 10 mM DNPH in 2 M HCl was added to the cell lysates. Samples were incubated for 1 hr at room temperature and were vortexed every 15 min. Then, 1 ml of trichloroacetic acid (TCA 10% w/v) was added to each reaction mixture and centrifuged at 3000 rpm for 10 min. The pellets were washed twice with 2 ml of ethanol/ethyl acetate (1:1, v/v) and each dissolved in 1 ml of guanidine hydrochloride (6 M, pH 2.3) and incubated for 10 min at 37˚C whilst mixing. The carbonyl content was calculated based on the molar extinction coefficient of DNPH (ε = 2.2 × 104 cm−1·M−1).
2.7. Fluorescence Microscopy Evaluation of Apoptotic Cells
Acridine orange/ethidium bromide double staining was applied to observe the morphological changes among menadione-treated cell. Using this technique, cells can be distinguished as normal cells (uniformly stained green) and apoptotic cells that are stained orange because of cell membrane destruction and the intercalation of ethidium bromide between the nucleotide bases of DNA. After treatment, cells were washed twice with phosphate buffer saline and adjusted to a cell density of 1 × 104 cells/ml of phosphate solution (1:1 v/v). The nuclear morphology was evaluated by Axoscope 2 plus fluorescence microscope from Zeiss (Germany). The cells with condensed or fragmented nuclei were counted as apoptotic cells. All experiments were repeated three times, and the number of stained cells was counted in 10 randomly selected fields.
2.8. Evaluation of Intracellular Formation of Lipofuscin Pigments
Extraction of intracellular lipofuscin was achieved following lysis of each sample according to a published procedure with slight modification [28]. The cells were seeded in triplicate into 24-well plates for 24 h prior to pretreatments. After pretreatment with different doses of baicalein (10, 20, 40 µM) for 3 h, each cell sample was treated with 35 µM menadione for 24 h. The attached cells in each well were trypsinized with trypsin-EDTA solution followed by cell counting using a hemocytometer. Each plate was then centrifuged, the cell pellet was washed with PBS, and the cell content was lysed with lysis buffer containing 1% Triton X-100, 1 mM EDTA and 1 mM PMSF. Each cell lysate was harvested and its fluorescence intensity was monitored on a varian spectrofluometer, model Cary Eclipse, with an excitation wavelength of 310 nm and emission wavelength of 620 nm [29]. The fluorescence intensities of the samples were then normalized for equal cell numbers.
2.9. Measurement of Intracellular Iron Contents via Ferrozine-Based Colorimetric Assay
The assay was performed directly in 24-well plates. Cells were lysed by addition of 200 µl iron releasing reagent (a freshly mixed solution of equal volumes of 1.4 M HCl and 4.5% (w/v) KMnO4 in H2O2) to each well. The plates were sealed with foil and incubated for 2 h at 60˚C, after which 60 µl of the detection reagent (6.5 mM ferrozine, 6.5 mM EDTA, 2.5 M ammonium acetate and 1 M ascorbic acid dissolved in water) was added. After further incubation for 30 min at room temperature, 280 µl of the mixture was transferred to a well of a 96-well plate and its absorbance recorded at 550 nm and compared to the absorbance of the FeCl3-treated standards under all equal experimental conditions. The determined intracellular iron concentration for each well was normalized against the protein content of replicate wells [30].
2.10. Western Blot Analysis
SK-N-MC cells were seeded at a density of 105 cells/ml in 12-well plates for 24 h. The cells were pretreated with baicalein (40 μM) and caspase inhibitor (50 µM). After 3 h, menadione (35 μM) was added to the cells and incubated at 37˚C for an additional 24 h. Then, the cells were harvested and lysed using lysis buffer containing 1% Triton X-100, 1% SDS, 10 mM Tris (pH 7.4), 100 mM NaCl, 1 mM EGTA, 1 mM EDTA, 20 mM sodium pyrophosphate, 2 mM Na3VO4, 1 mM NaF, 0.5% sodium deoxycholate, 10% glycerol, 1mM phenylmethylsulphonyl fluoride, 10 μg/ml leupeptin, 1 μg/ml pepstatin and 60 μg/ml aprotinin. Protein concentration of each sample was determined using Lowry’ method (Lowry et al., 1951). Equal quantities of protein (40 μg) were subjected to 12.5% SDS-polyacrylamide gel electrophoresis (PAGE) and were transferred to PVDF membranes. The blots were blocked with 5% (w/v) non-fat dry milk in Tris-buffered saline buffer containing 0.1% Tween-20 (TBS/T) for an overnight at 4˚C. The blocked blots were incubated with primary antibodies for 2 hr at room temperature using antibody dilutions as recommended by the manufacturer in Tris-buffered saline pH 7.4 containing 0.1% Tween-20. After 1-hr incubation with anti-rabbit or anti-mouse horseradish peroxidase (HRP)-conjugated secondary antibodies (Biosource), the proteins were detected by an enhanced chemiluminescence detection system (Amersham-Pharmacia, Piscataway, NJ, USA) according to the manufacturer’s instructions. Blots were stripped at 50˚C for 30 min in 100 mM 2-mercaptoethanol, 2% SDS, 62.2 mM Tris-HCl pH 6.7 and reprobed for further investigations. For analysis of the western blotting data, densitometric analysis was performed using Image.J software, and the densities were normalized with respect to β-tubulin as the internal control.
2.11. Statistical Analysis
Data were expressed as percent of values of untreated control cells, and each value represents the mean ± SD (n = 3). The significant differences between the means of the treated and untreated cells were calculated by unpaired Student’ t-test, and p-values < 0.05 were considered significant.
3. Results
3.1. Baicalein and Pan-Caspase Inhibitor (Z-VAD-Fmk) Shield SK-N-MC Cells against Menadione-Induced Cytotoxicity
Menadione is a quinone known to induce an oxidative stress generated primarily by superoxide radicals leading to cell death [31]. We found that menadione at 35 µM caused 55% cell death among SK-N-MC cells (Figure 1). In our previous study, we ascertained that no remarkable changes were seen among the cells in range of 10 - 50 µM of baicalein after 24 h [32]. Thus, cytoprotcetive effects of different doses of baicalein (10, 20, 40, 50 µM) on menadione (35 µM)-induced cytotoxicity in SK-N-C cells were investigated. The detrimental effects of menadione on SK-N-MC cells were considerably blocked by pretreatment with baicalein. The same result was observed for Z-VAD-fmk. As shown in Figure 1, the extent of survival was restored to 67%, 84%, 89% and 71% by pretreatment of cells with different doses of baicalein (10, 20, 40, 50 µM) for 3 h followed by treatment with 35 µM menadione for 24 h. Baicalein at a concentration of 40 µM, provided utmost protection against menadione insult producing a 44% increase in cell survival. Moreover, Z-VAD-fmk (50 µM) increased cell viability to 86% (Figure 1).
3.2. Baicalein but Not Z-VAD-Fmk, Mitigates Menadione-Induced Increase in Intracellular ROS Generation
Increase in ROS generation was measured as one of the indicators of menadione-induced oxidative stress in cells. As shown in Figure 2, generation of intracellular ROS (in term of DCF fluorescent intensity) in SK-N-MC cells increased by almost a factor of 6.2 after 12-h treatment

Figure 1. Cts of menadione, baicalein, and Z-VAD-fmk on viability of SK-N-MC cells. SK-N-MC cells were treated with different concentrations of menadione (20, 35, 50 µM) to find IC50 of menadione for further experiments (35 µM). Then, SK-N-MC cells were pretreated with different concentrations of baicalein (10, 20, 40, 50 μM) and Z-VAD-fmk (50 µM) for 3h and then incubated with menadione (35 μM) for 24 h. Cell viability was examined by MTT assay. Values correspond to means ± SD of three independent experiments. *significantly different from control cells (p < 0.05), #significantly different from menadione-treated cells (p < 0.05).

Figure 2. Effects of baicalein and Z-VAD-fmk on intracellular ROS level in menadione-treated SK-N-MC cells. SKN-MC cells were pretreated with baicalein (10, 20, 40 μM) and Z-VAD-fmk (50 µM) for 3 h and then incubated with menadione for 12 h. ROS levels were monitored using 2', 7' dichlorofluorescein diacetate (DCFH-DA) staining. The fluorescence intensity was monitored on a varian-spectrofluorometer with excitation and emission wavelengths of 485 and 530 nm, respectively. Values correspond to means ± SD of three independent experiments. *significantly different from control cells (p < 0.05), #significantly different from menadione-treated cells (p < 0.05).
with menadione (35 µM) compared to ROS level of the untreated control cells. Pretreatment of the cells with different doses of baicalein (10, 20, 40 μM) attenuated ROS production in SK-N-MC cells by factors of 2.3, 3.6 and 4.3, respectively. However, pretreatment with ZAD-mk (50 µM for 3 h) did not significantly change the ROS level in menadione-treated SK-N-MC cells.
3.3. Baicalein but Not Z-VAD-Fmk, Curbs Menadione-Induced Lipid Peroxidation
Menadione-induced oxidative stress causes oxidation of intracellular biomolecules such as lipids. MDA is produced while lipid peroxidation happens. So, MDA level measurement is used as a marker of menadione-induced oxidative stress. As shown in Figure 3, baicalein repressed lipid peroxidation in SK-N-MC cells. After 12 h of incubation with 35 μM menadione, MDA levels were significantly increased relative to the untreated control cells (0.41 nmol/mg protein in control cells versus 2.33 nmol/mg protein in menadione-treated cells). Pretreatment of cells with different doses of baicalein (10, 20, 40 μM) for 3 h followed by a 12 h treatment with menadione (35 μM) reduced MDA formation to 1.64, 1.01, and 0.66 nmol/mg protein, respectively, indicating that baicalein had quenched lipid peroxidation of the SK-NMC cells. However, pretreatment with 50 µM Z-VADfmk did not significantly alter MDA contents in menadione-treated SK-N-MC cells.

Figure 3. Effects of baicalein and Z-VAD-fmk on intracellular lipid peroxidation and protein carbonyl formation in menadione-treated SK-N-MC cells. SK-N-MC cells were pretreated with baicalein (10, 20, 40 μM) and Z-VAD-fmk (50 µM) for 3 h and then incubated with menadione for 12 h. lipid and protein oxidations were measured by analysis of MDA and PCO. Values correspond to means ± SD of three independent experiments. *significantly different from control cells (p < 0.05), #significantly different from menadionetreated cells (p < 0.05).
3.4. Baicalein but Not Z-VAD-Fmk, Diminishes Menadione-Induced Protein Carbonyl Formation
Protein carbonyl is a marker of protein oxidation in oxidative stress condition. We evaluated the effects of different doses of baicalein (10, 20, 40 μM) and Z-VADfmk (50 µM) on protein carbonyl formation in SK-N-MC cells. After treatment with menadione (35 μM), the amount of protein carbonyl increased to 4.03 nmol/mg protein compared to 0.65 nmol/mg protein of control cells. Pretreatment with baicalein (10, 20, 40 μM) reduced protein carbonyl formation to 2.6, 1.7 and 1.1 nmol/mg protein, respectively (Figure 3). However, pretreatment with 50 µM Z-VAD-fmk did not significantly alter PCO contents in menadione-treated SK-N-MC cells.
3.5. Baicalein and Z-VAD-Fmk Prevent Menadione-Induced Caspase-Dependent Apoptotic Cell Death
To study the protective effect of baicalein on SK-N-MC cells, acridine orange/ethidium bromide double staining technique was used to evaluate the occurrence of apoptosis in cells. As shown in Figure 4, the non-apoptotic control cells were stained green and the apoptotic cells had orange particles in their nuclei due to nuclear DNA fragmentation. The menadione treatment increased the extent of apoptosis relative to untreated control cells and pretreatment with baicalein (40 μM, 3 h) diminished apoptosis compared to menadione-treated cells (Figure 4). We also pretreated SK-N-MC cells with Z-VAD-fmk (50 µM) for 3 h followed by exposure to menadione (35
 (a)
(a) (b)
(b)
Figure 4. Effect of baicalein and Z-VAD-fmk treatments on menadione-induced apoptosis in SK-N-MC cells. (a) SKN-MC cells were treated with baicalein (40 μM) and Z-VAD-fmk (50 µM) for 3 h followed by exposure to menadione (35 μM) for 24 h. cell pretreatment with baicalein and Z-VAD-fmk clearly decreased the number of apoptotic cells relative to cells treated only with menadione. Values correspond to means ± SD of three independent experiments. *significantly different from control cells (p < 0.05), #significantly different from menadione-treated cells (p < 0.05); (b) morphological analysis of SK-N-MC cells by double staining method. White arrow indicates live cells, dashed arrow shows apoptotic cells. Scale bar: 40 µM.
µM) for 24 h. As shown in Figure 4, Z-VAD-fmk reduced the extent of apoptosis relative to menadionetreated cells, confirming the caspase-dependent apoptosis of cells.
3.6. Effect of Baicalein on Menadione-Induced Lipofuscin Formation
Exposure of the cells to 35 µM menadione for 24 h caused 374% increase in the intracellular level of lipofuscin relative to menadione-untreated control cells. Pretreatment of the cells with baicalein (10, 20, 40 µM) diminished the formation of lipofuscin pigments by 155%, 192% and 214% after 24 h of exposure (Figure 5).
3.7. Baicalein Decreases Iron Accumulation in Menadione-Induced SK-N-MC Cells
Iron is important for electron transport in the respiratory chain and for various enzymatic reactions. When present in excess, however, iron can harm biological systems since in redox-active form it catalyzes the generation of highly reactive oxygen species [33]. Since both iron deficiency and overload impaired cellular functions, the quantitation of iron in cells and extracellular fluids is of considerable interest [34,35]. As shown in Figure 6, treatment of SK-N-MC cells with menadione elevated free iron contents compare to basal iron level in the control samples (2.17 nmol/mg proteins compare to 1.1 nmol/mg protein of control). However, pretreatments with different doses of baicalein (10, 20, 40 µM) diminished the iron contents to 1.75, 1.54 and 1.33, respectively.
3.8. Effects of Baicalein and Z-VAD-Fmk on Menadione-Induced Cell Death
Previous studies have shown that menadione-induced

Figure 5. Inhibitory effect of baicalein on the menadionetreated accumulation of intracellular lipofuscin pigments. SK-N-MC cells were exposed to baicalein (10, 20, 40 µM) for 3 h followed by exposure to menadione (35 µM) for 24 h. Then, the extent of lipofuscin in cell lysates were evaluated using a varian spectrofluorometer, model Cary Eclipse, set at an excitation wavelength of 310 nm and an emission wavelength of 620 nm. *significantly different from control cells (p < 0.05), #significantly different from menadionetreated cells (p < 0.05).

Figure 6. Effect of baicalein on intracellular iron contents in menadione-treated SK-N-MC cells. SK-N-MC cells were exposed to baicalein (10, 20, 40 µM) for 3 h followed by exposure to menadione (35 µM) for 24 h. Iron contents were evaluated by colorimetric ferrozine-based assay. *significantly different from control cells (p < 0.05), #significantly different from menadione-treated cells (p < 0.05).
apoptosis is associated with changes in apoptosis-related Bcl-2 family of regulatory proteins. Bax is a pro-apoptotic member of the Bcl-2 family which forms mitochondrial permeability pores for release of cytochrome c to the cytosol via binding to the anti-apoptotic Bcl-2 member. This event in turn will lead to cleavage of procaspase-9 and further activation of procaspase-3 and cell death through apoptosis [36]. Pretreatment of cells with baicalein prior to menadione treatment, reduced Bax/ Bcl2 ratio and pretreatment of cells with baicalein and Z-VAD-fmk decreased cleaved caspase-9 in SK-N-MC cells which showed that baicalein inhibited caspase-dependent apoptosis in this cell line (Figure 7).
4. Discussion
One of the well-accepted theories for explicating the aging process is the free radical theory proposed by Denham Harman [5]. This theory illustrates that there is a causal relationship between oxidative stress and pathogenesis of age-related disorders [6]. Lipofuscin, a histological index of aging, is a highly oxidized cross-link aggregate consisting of oxidized proteins (30% - 58%) and lipids (19% - 51%) clusters accrues mostly in postmitotic cells such as neurons, cardiac myocytes, skeletal muscle fibers and retinal pigments [7]. Since oxidative reactions are compulsory components of normal life processes, the incidence of reactive oxygen species with ensuing lipofuscin formation is an inexorable side effect of life [10]. Many studies have signified that many ROSinduced diseases such as neurodegenerative disorders are associated with high levels of lipofuscin within neuronal cells [37,38]. It has been widely reported that loosely bound iron in the cellular iron pool can react with endogenous hydrogen peroxide to produce the short-lived and highly reactive hydroxyl radicals through the Fenton reaction. These hydroxyl radicals, in turn, can oxidize nucleic acids, proteins or lipids leading to lipofuscin formation [23]. Oxidized proteins within lipofuscin are linked by intermolecular cross-links. Many of these cross-links are caused by non proteineous compounds including oxidized lipids such as Malondialdehyde (MDA) and 4-hydroxy-2-nonenal by means of reactions with lysine amino groups, cysteine sulfhydryl groups and histidine imidazole groups of proteins [39]. Thus, preventing biomolecules peroxidations and maintaining iron homeostasis play major roles in blocking lipofuscin formation.
Menadione (2-methyl-1,4 naphthoquinone) in the cells converts to menadione semiquinone radical via NADPH cytochrome c reductase activity. Then, semiquinone radical is recycled back to menadione through rapid reaction with molecular oxygen. This can result in the formation of superoxide radical which causes oxidative stress [40]. Although superoxide is chemically incapable of
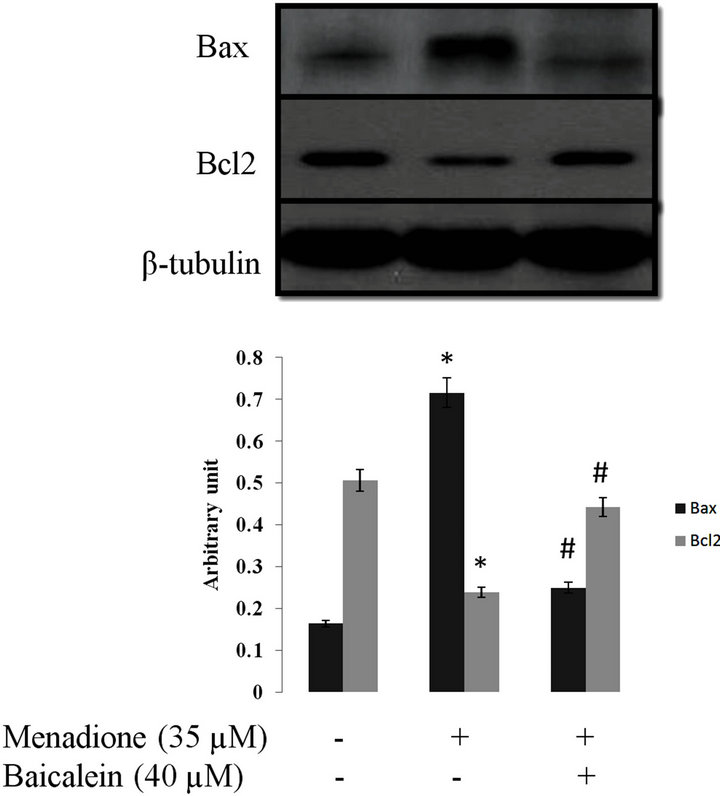 (a)
(a)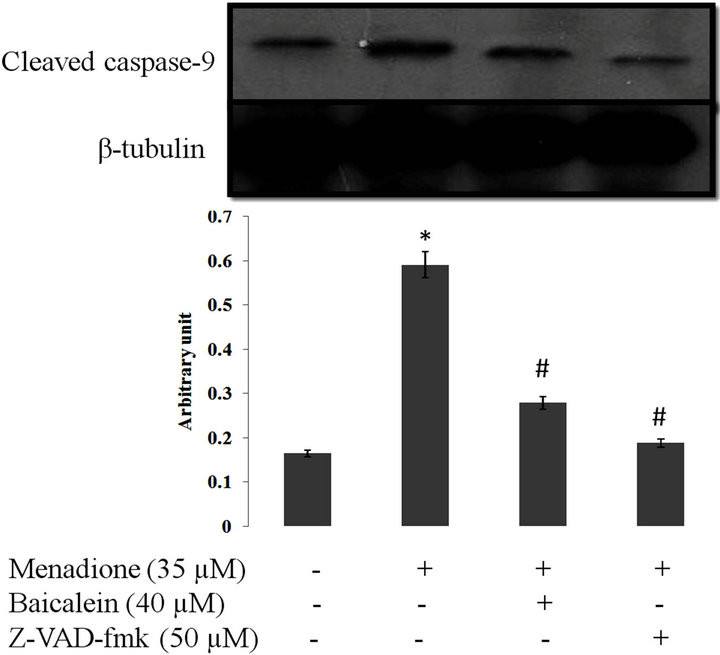 (b)
(b)
Figure 7. Analysis of Bcl-2, Bax and procaspase-9 activation in SK-N-MC cells treated with menadione, baicalein and Z-VAD-fmk. SK-N-MC cells were pretreated with baicalein (40 μM) and Z-VAD-fmk (50 µM) for 3 h and then incubated with menadione (35 µM) for 24 h. (a) bcl-2, Bax and the (b) procaspase-9 expression were estimated by immunoblots using relevant specific antibodies, and intensity of each band was estimated by densitometric analysis. Equal sample loadings were confirmed by tubulin band. Values correspond to means ± SD of three independent experiments. *significantly different from control cells (p < 0.05), #significantly different from menadione-treated cells (p < 0.05).
affecting biomolecules directly, it is assumed to do so indirectly by participating in the production of hydroxyl radicals through Fenton reaction. Superoxide radicals can provide free iron to catalyze peroxidation from two sources: release iron from ferritin and oxidizes the [4Fe - 4S] clusters of enzymes such as dehydratases, precipitating the release of one or more iron atoms [41]. Thus, menadione as a Fenton catalyst, assisted the production of free iron for production of hydroxyl radicals to ignite cross link of oxidized proteins and lipids in order to form lipofuscin.
There is an accumulating evidence denoting that lipofuscin can induce neurotoxicity via its capacity for binding metals such as iron, copper, zinc and calcium which stimulates generation of excessive ROS and decrease proteasomal and lysosomal degradation by inhibition of the proteasomal turnover [7]. Numerous studies have shown that intracellular iron accumulation contributes to the development of several common neurodegenerative diseases such as Alzheimer’s disease (AD) and Parkinson’s disease (PD) [33-35].
In order to restrain the destructive effects of ROS including superoxide radicals in neuronal cells, dietary flavonoids are shown to have potential anti-aging and brain-protective activities. Baicalein (5, 6, 7-trihydroxy- 2-phenyl-4H-1-benzopyran-4-one), a naturally occurring flavonoid, is the major bioactive compounds found in traditional Chinese medicinal herb, Baikal Skullcap (Scutellaria baicalensis GEORGI) [22]. Baicalein produces promising results as a strong antioxidant. Its ability to cross blood brain barrier (BBB), hydrophobicity, presence of hydroxyl groups at C-5 and C-7, a double bond between C-2 and C-3, high trolox equivalent antioxidant capacity (TEAC) and DPPH free radical scavenging activity make baicalein a good ROS scavenger in neurons [20,21]. Presence of hydroxyl groups in baicalein structure results in scavenging of charged species such as superoxide radicals and hydroxyl radicals more efficiently compared to non-charged oxidant species [20]. On the other hand, baicalein can inhibit the production of endogenous hydroxyl radicals produced through the Fenton reaction by forming stable and inert complexes with iron [23]. Iron-binding motifs in some phenolic compounds can clarify the potential ability of them to modulate iron homeostasis in the body. Baicalein contains these motifs and thus expected to chelate iron. Some recent studies have shown that two hydroxyl groups at the 6 and 7 positions on the A ring seems to be the powerful metal binding site [20,23].
In support of what we have explained before, our studies showed that baicalein reduced the harmful effects of menadione by scavenging superoxide radicals which led to increased cell viability and decreased intracellular MDA and PCO. In addition, our results confirmed that baicalein has anti-Fenton properties since it decreased the free iron contents of SK-N-MC cells exposed to menadion treatment. We also observed that baicalein strongly inhibited lipofuscin formation in menadione-treated SKN-MC cells and displays anti-aging features. Morphological analysis and western blot results implied that baicalein prevented apoptotic cell death through inhibition of Bax and procaspase-9 activations and induction of bcl2 expression which averted activation of further caspases and transcription factors, release of cytochrome c and resultant cell death. The results were confirmed by applying pan-caspase inhibitor (Z-VAD-fmk). Moreover, our experiments have shown that Z-VAD-fmk prevented cell death in SK-N-MC cells through inhibition of caspases and did not have any significant antioxidant characteristics.
Overall, flavonoid baicalein can be considered as a strong and auspicious antioxidant which could protect neuronal cells and hence, baicalein is a reliable option for antioxidant therapy in treatment of age-related and neurodegenerative disorders, pending further in vivo and clinical investigations.
5. Acknowledgements
The author appreciates the financial support of this investigation by the Research Council of University of Theran.
Abbreviation
MDA: Malondialdehyde;
MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide;
PBS: Phosphate buffer saline;
PCO: Protein carbonyl;
AD: Alzheimer’s disease;
NOTES