Boron Nitride Nanostructured: Synthesis, Characterization and Potential Use in Cosmetics ()
1. Introduction
The cosmetic industry currently has researched materials that offer advantages and which are attractive to customers. Ceramic materials are widely used in cosmetics and are manufactured by ceramic powder processing, sol-gel technology and precipitation solutions. The boron nitride is a ceramic material with a chemical formula (BN). Rhombohedral (r-BN), wurtzite (w-BN), cubic (c-BN) and hexagonal (h-BN) are the crystal structures of the BN caused by the arrangements of nitrogen and boron atoms [1,2]. The hexagonal structure allows the molecules to be arranged in layers so they slide one over the other and their use becomes interesting in cosmetic formulation to confer better lubricity mixtures.
Regarding the new researches in parameters used for the h-BN nanostructure synthesis aim to improve productivity, quality and control the size of synthesized particles. Recently, many studies have reported the preparation of nanostructures of the boron nitride with special morphologies, such as nanotubes [3,4], nanocapsules [5], nanocages [6] porous structures [7], hollow spheres [8], and nanofibers [9].
Traditionally, h-BN is prepared by the classical hightemperature synthesis routes, including a reaction of B2O3, boric acid, or borax with carbon and nitrogen/ammonia and urea at a temperature around 2000˚C [10]. On the other hand, for special BN nanostructures synthesis, laser ablation, chemical vapor-phase, carbothermal reduction of B2O3 and B4C and other methods have been developed [11]. Most of these methods cannot meet the needs of high yields, and therefore, the understanding on its synthesis is still a challenging subject. Moreover, BN nanotube obtained from different synthetic methods has different physical properties. Quality, quantity, and type of nanotubes synthesized depend on the synthetic method used.
In the cosmetics field, the particle size is important because smaller diameters may enable an increase in the surface area and the possibility of encapsulating cosmetics actives. The h-BN may offer advantages for use in cosmetics, particularly in sunscreens, since it allows manipulation of the structure during different phases of formation and, consequently, control of the size and the specific surface area; nevertheless, it has an excellent performance in the dispersion, it is non-toxic, transparent, and chemically inert.
Solar radiation is known to damage skin health and the cosmetic industry has invested in the research and manufacture of effective sunscreens. The range of the solar spectrum that normally reaches the surface of the Earth and has a special interest for photomedicine is that of ultraviolet B (UVB) and the Infrared (IR) [12]. The effect of ultraviolet rays is harmful to the health of the skin and can result in an increased cancer incidence, aging and other undesirable skin changes. Ultraviolet B is responsible for sunburn, erythema and inflammation. The effects of infrared rays are less described in the scientific literature, but these rays are known to increase the damages on the skin induced by ultraviolet rays [13], premature aging [14,15] and infrared radiation may have a photocarcinogenic potential [16].
Sunscreens have been researched for a long time and currently they have been developed using the nanoscience and nanotechnology knowledge, as titanium dioxide, an important cosmetic used as sunscreen against ultraviolet radiations. However, for the protection against infrared radiation little has been researched. In the market, there are several inorganic actives such as titanium dioxide (TiO2) and zinc oxide (ZnO), which are able to block effects of ultraviolet radiation. These materials currently have smaller sizes and have advantages over the conventional product, as the phenomenon of radiation spread and therefore a white color under the skin [17].
With this regard, there is a great need to create formulations of sunscreens which can offer protection against ultraviolet rays as well as infrared rays. The boron nitride nanostructure is a potential material for this purpose as it is possible to manipulate its size, an important factor to use in sunscreens, thus reducing shine from oily skin as it has a high thermal conductivity. Indeed, it has absorption in the infrared ray region and special formulation containing titanium dioxide particles with nanostructured boron nitride would be a perfect combination to be applied as a cosmetic for protection against solar radiations in a broad spectrum.
2. Experiments
2.1. Synthesis
BN nanostructured samples were prepared using 12.36 g of boric acid (H3BO3, Merck) dissolved in 400 ml of distilled water at 100˚C. After dissolution 12.60 g of melamine (C3H6N6, Merck) was slowly added to the medium under agitation. We observed the formation of a precipitate early in the reaction. The material was allowed to stand for 48 hours at room temperature. Then it was filtered and dried at 37˚C. The product obtained is B4N3O2H, which is the precursor to BN. This precursor material was heated in tubular furnace without gas flow at 500˚C for 3 hours constantly and followed by nitrogen gas flow, for 1 hour at 800˚C (5˚C/min) at a constant temperature. This precursor was placed in an aluminum boat and heated at 1600˚C (10˚C/min) for 2 hours with nitrogen gas flow.
2.2. Characterization
The prepared samples were characterized by Fouriertransform infrared spectroscopy (FTIR), X-ray diffraction (XRD), Brunauer-Emmett Teller (BET), scanning electron microscopy (SEM), energy dispersive spectroscopy (EDS), transmission electron microscopy (TEM) and Spectroscopy (UV/VIS). FTIR measurements were carried out in a Galaxy-Matson 3020 spectrophotometer ranging from 4000 to 400 cm−1 at room temperature using KBr pellets. XRD patterns were obtained using a Rigaku Geigerflex diffractometer with a Cu-Ka tube. The specific surface area was measured by Quantachrome NOVA 1200 adsorption analyzer. SEM characterization was performed in a scanning electron microscope (JEOL, JSM, 840) operating at 15 kV. EDS was performed by (JEOL JXA-8900R).TEM characterization was performed in a transmission electron microscope (JEOL, JEM, 2100F) with an acceleration potential of 200 kV. UV/VIS characterization was performed in a Shimadzu model (2401) spectrometry.
3. Results and Discussion
The synthesis begins with the formation of the melamine diborate precursor [18]. It was prepared from boric acid and melanin reaction as below. They can be described in Equation (1) (see below).
Various temperatures and heating rates were tested after preparing the precursor and which showed the best results when the sample was subjected to 1600˚C at a heating rate of 10˚C/min for 2 hours with N2. For this synthesis, an initial 2-hour purge was carried out in order to minimize the presence of contaminants. This was crucial to the proposed result.
Figure 1 shows the FTIR spectra of samples treated at 1600˚C. The sample has characteristic peaks in the hexagonal boron nitride formation. The spectra showed an asymmetric bond at 1380 cm−1 which corresponds to B-N stretch and the bond at 775 cm−1 assigned to the connection which may be attributed to B-N-B. The band at 3400 cm−1 can be attributed to the moisture absorbed. The spectrum has to be free of impurities, which are related to other crystalline forms of boron nitride. It is noteworthy

that this technique generates important data to characterize the h-BN phase, since it is possible to distinguish sp2 bonds of hexagonal h-BN phase samples and sp3 of cubic c-BN phase. According to Hao and colleagues [19] sp2- type bonds (h-BN) are thermodynamically stable under the synthesis conditions of this work, whereas for the formation of sp3 bonds typical of c-BN, there is a kinetics barrier to formation. Other authors have presented similar results in which the formation of h-BN occurs at around 1380˚C [20,21]. Peaks in 1096 and 1166 cm−1 are due to the formation of c-BN phase and they were not found in the sample treated at 1600˚C.
Figure 2 shows the X-Ray diffraction pattern of the sample treated at 1600˚C. The peaks at 2q = 26.32˚, 2q = 41.60˚ and 2q = 55.00˚ (JPCD, No. 9 - 12) may be attributed to hexagonal boron nitride formation. The peak at 26˚ corresponds to the (002) diffraction peak of h-BN [19] and it is very intense and well defined. This sample is more crystalline compared to other temperatures tested. The turbostratic structure (imperfect structure with no three-tridimensional order) has been produced at a low temperature. This structure is transformed in an ordered hexagonal from by heat treatment at a temperature above 1500˚C [22].
 (a)
(a) (b)
(b)
Figure 1. (a) Infrared spectrum of the sample treated at 1600˚C, and (b) Expanded Infrared spectrum treated at 1600˚C.
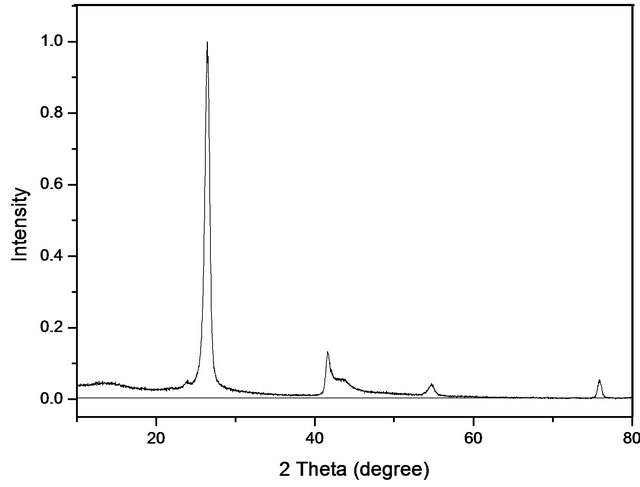
Figure 2. XRD pattern of the sample treated at 1600˚C.
From the diffraction data, we measured the particle size according to the Scherrer equation:
 (2)
(2)
where Dhkl is the particle size perpendicular to the normal line of (h k l) plane, k is a constant (it is about 0.9), bhkl is the full width at half height of the (h k l) diffraction peak, qhkl is the Bragg angle of (h k l) peak and l is wavelength of X-ray. The average size of the particle found was 15 nm. This value is important for the objective of the study since it shows that the material is in the nanoscale.
Figure 3 shows nitrogen isotherm and pore distribution of the sample. According to the literature, boron nitride may have a surface area between (5 m2/g - 820 m2/g) but these values depend on factors related to the synthesis conditions and polymeric precursors used [23]. The value found (90 m2/g) may be considered interesting since the increasing temperature leads to a decrease in the surface area. This material also has the potential to be used as a matrix for embedding drugs due to area surface. The isotherm is similar to type IV (mesoporous material) with micropores contribution and display type H3 hysteresis loop according to the IUPAC classification, which is often observed with aggregates of plate-like particles that give rise to slit-shaped pores. The adsorption of the gas first starts with micropores and then continues with mesopores. The adsorption and desorption mechanism occurs at the same time in micropores. While adsorption occurs by the condensation towards the walls of pores, desorption occurs by evaporation from the walls of pores. In mesopores, adsorption and desorption follow different ways. The filling of the mesopores occurs by the condensation on the wall of pores with formation of layers. On the other hand, desorption occurs by moving away from pore opening with the evaporation. Hence, in mesoporous region where the P/Po > 0.4, type H3 hystreresis was observed. This experiment revealed that the final material possess a narrow pore size distribution with an average pore size of about 3.6 nm, characteristic of mesoporous materials. The pore size distribution (Figure 3(a)) for the produced nanostructured boron nitride was plotted using desorption data of BJH (Barrett-JoynerHalenda) method at 0.35 < P/Po < 0.99 corresponding to mesoporous site. The material presents a total pore volume of 0.26 cc/g calculated using the BJH method and with a pore size of 3.6 nm in diameter. Table 1 shows the main results obtained from the N2 adsorption.
Figure 4 shows the micrograph of the sample treated at 1600˚C. The formation of an agglomerate of fine grain of hexagonal boron nitride with diameter around 0.5 µm is observed. This image is important to demonstrate the application of the material studied in the field of cosme-