Quantitative determination of low molecular weight antioxidants and their effects on different antioxidants in chicken blood plasma ()
1. INTRODUCTION
The oxygen is a highly reactive molecule ready to damage living organisms [1,2]. In order to safely and effectively consume oxygen, organisms raise a complex network of enzymes, metabolites and molecules with antioxidant activities which prevent oxidative damage of cellular components. Active components terminate oxidation processes in the net by removing or quenching radical intermediates, and are capable of slowing or preventing the oxidation of other molecules [3].
Biological antioxidants may be classified into several defense groups; enzymes (catalase, peroxidase and superoxide dismutase), low molecular weight compounds (LMWA) with antioxidant activities based on a chain of chemical reactions, for instance glutathione, vitamin C, vitamin E, α-lipoic acid (ALA), coenzyme Q10 (CoQ10), urea etc., and the group of widespread natural plant pigments which protect biological samples mostly against 1O2—mediated damages by physical quenching. Such substances are carotenoids, for instance ß-carotene, lycopene, lutein and zeaxanthin. The interactions between different antioxidants are a complex processes, with various metabolites and enzyme systems having synergistic and interdependent effects on one another. The amount of protection provided by any antioxidant therefore depends on its standard potentials E0 (V), concentration, reactivity towards the particular reactive oxygen species, and the status of the antioxidants with which it may interacts (Table 1).
Until recently, scientists believed that each antioxidant worked separately, independently of the others. The results of research work performed at the Packer Lab at the University of California, Berkeley showed that there is a dynamic connection among certain key antioxidants. These special antioxidants work together and represent the dynamic defence of the organism. The expression antioxidant network was first used by Packer in his book The Antioxidant Miracle [4], where he stated that antioxidants do not act alone but are linked together into a net. The interaction of antioxidants had already been established before Packer, yet he was the first who outlined a concept of the mode of action of the antioxidant network including five molecules, CoQ10, ascorbic acid, tocopherol, glutathione and lipoic acid. According to his opinion no antioxidant acts alone, but is included in the net or supporting it, Figure 1.
Coenzyme Q10 or ubiquinone [5-7] is a lipid soluble substance consisting of quinone head and isoprenoid side chain. Apart from being a cofactor in the respiratory chain CoQ10 serves as an important antioxidant in mitochondria and lipid membranes [8,9]. In an organism the requirement for CoQ10 is usually fulfilled by biosynthesis, but in many cases administered substance to the diet is an important supplementary source [10]. CoQ10 is a lipophilic substance, and is usually converted into a more soluble formulation before it is added to various forms of food and fodder products. Our contribution in this field was a new originally prepared water soluble inclusion complex of CoQ10 with β-cyclodextrin [11,12], which


Table 1. Some antioxidants and their standard potentials E0 (V), which participate in formation of antioxidant network [38,39].

Figure 1. Chain structure of synergistic effect of five LMWA in chicken plasma, and standard potentials of some plant antioxidants, for instance carotenoids and catechins.
showed advantageous applicability, better enteric absorption, and bioavailability [13].
Alfa lipoic acid (ALA) is a yellow solid organosulfur compound [14]. Structurally it exists as a R+lipoic acid, S-lipoic acid and as a racemic mixture R/S-lipoic acid. RLA exists in nature and is essential for life and aerobic metabolism. All forms can be reduced to dihydrolipoic acid with tissue specific or stereo selective reactions. ALA was recognized to have antioxidant potential in 1959 but it took more than 40 years to gain significant public attention and application in maintaining or restoring human health. ALA is presented in almost all foods, like animal kidney, heart, liver, spinach, and broccoli, however in very low concentration and as lipoyllisine. ALA is the rate-limiting factor for the production of acetyl coenzyme A. It is the generator of intracellular glutathione, which is the most efficient intracellular antioxidant and is a very efficient free radical scavenger, which removes many inflammatory products that interfere with normal organ regeneration. In addition ALA regenerates several antioxidants taking the active part in antioxidant network to scavenge the free radicals, like vitamin C, vitamin E, CoQ10 [14-16]. All ALA supplements are chemically synthesized because the naturally occurring lipoic acid is always covalently bounded and is not directly available from dietary sources.
Our research work concerning industrial poultry farming pointed out that chickens and hens may be very suitable subjects for scientific estimation of the protective effect of LMWA in an antioxidant network. Previous studies were done mostly on rats and mice, where CoQ9 is the predominant homolog. In our experiments we selected chickens and hens, which major homolog is CoQ10 similar to human and animals with longer life time.
In the first part of our work we studied the accumulation of administered CoQ10 in particular parts of chicken body and the quality of prepared chicken products from the fortified meat and organs. The highest increase of CoQ10 concentration was observed in chicken breast cells after 40 days of feeding with CoQ10. Fractionation of chicken breast cells showed, that exogenous CoQ10 was mainly incorporated into cell membranes and not in mitochondria. This confirmed our hypothesis that CoQ10 added in chicken tissues acts mostly as an antioxidant [17]. From these results we concluded that CoQ10 in young chickens has an important role as an antioxidant in chicken plasma and in chicken tissues [17,18].
In the second part of our work-analytical results presented in this paper, we evaluated the effects of CoQ10 and ALA on a minimization of the oxidative stress during the raise. The positive effects of exogenous CoQ10 and ALA have been described in many papers [7, 15,19-22], but only a few studies have assessed the effect on non-laboratory animals such as chickens and hens on farms [23-26].
Together with the Perutnina Ptuj d.d., Faculty of agriculture and life science from University of Maribor, and Biotechnical faculty from University of Ljubljana, we prepared a research project in which the health and plasma antioxidant status of broiler chickens during the controlled experimental and industrial raise was studied. The effects of supplemented CoQ10, and ALA and the combination of both substances on the reduction of oxidative stress and regeneration of other antioxidants in the animal body were assessed. The benefits of applied additives were estimated through the measured concentrations of selected LMWA (CoQ10, ALA, α-tocopherol), enzyme antioxidant activities (superoxide dismutase (SOD), glutathione reductase (GPx) and total antioxidant capacity (TAC)), cholesterol and glucose. Concentration levels were measured with the intention to find synergistic effects and possibly confirm the presence of “an antioxidant network” in plasma.
2. MATERIALS AND METHODS
2.1. Experimental Design
Animals were divided into four treatment groups: three test groups ( , GALA,
, GALA, ) with 50 animals (25 male, 25 female) and a control group (GC) with 200 animals (100 male, and 100 female). Each group was placed in separate stable. Tests were done in optimal breeding and healthy conditions. Animals in all groups were supplied with the same basic food. The flow chart of experimental procedure is presented in Figure 2.
) with 50 animals (25 male, 25 female) and a control group (GC) with 200 animals (100 male, and 100 female). Each group was placed in separate stable. Tests were done in optimal breeding and healthy conditions. Animals in all groups were supplied with the same basic food. The flow chart of experimental procedure is presented in Figure 2.
Chicken fodders BRO-S and BRO-F1 were prepared according to the standard Ross 308 fodder specifications. In test group (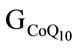 ) the standard fodder was fortified with 40 mg CoQ10 per kg, in test group (GALA) it was fortified with 400 mg of ALA per kg, and in test group (
) the standard fodder was fortified with 40 mg CoQ10 per kg, in test group (GALA) it was fortified with 400 mg of ALA per kg, and in test group ( ) the fodder with 40 mg CoQ10 and 400 mg ALA per kg of BRO-F1 was prepared. The required amount of CoQ10 was provided as the water soluble additive originally synthesized in our laboratory by in-capsulation of CoQ10 into corn dextrin. The additive was commercially named AmilQ. The applied food grade ALA and raw CoQ10 were purchased from Linyi Tianliheng Trade Co (China).
) the fodder with 40 mg CoQ10 and 400 mg ALA per kg of BRO-F1 was prepared. The required amount of CoQ10 was provided as the water soluble additive originally synthesized in our laboratory by in-capsulation of CoQ10 into corn dextrin. The additive was commercially named AmilQ. The applied food grade ALA and raw CoQ10 were purchased from Linyi Tianliheng Trade Co (China).
Test groups started to receive the supplemented diet on day 14 and were fed for a period of 26 days. Concentrations of supplemented CoQ10 and ALA were calculated from feed instruction tables so that each chicken received an average of approximately 5 mg of CoQ10 and (or) 50 mg of ALA per kg of animal weight
per day. During the 41-day pilot raise, all animals were treated under identical environmental and growing conditions. Health, appearance and behavior of each animal were recorded. The control group was raised and controlled under the same conditions but without fortified fodder.
During the raise blood samples were taken three times (on day 16, 28, and 40). In each group 7 male and 7 female animals were randomly selected and up to 2 ml of blood were taken from vene cutaneae ulnaris. One part of the blood sample (0.5 mL) was stored for biochemical analysis (SOD, GPx) and the rest of the blood (up to 1.5 mL) was used for plasma sample prepared for chemical analysis and TAS. Plasma end blood samples were stored in cool storage of −80˚C until the start of analyses.
In order to reduce the number of analysis plasma samples were pooled. Three plasma samples in each group (male and female separately) were randomly pooled into a “big” sample. Four pooled samples were prepared in the control group and in each of the test groups. Pooled samples were analyzed in two replicates. Biochemical parameters were measured with multi-channel analyzers and in this case collected blood samples were analyzed individually, for each animal, and the mean values were reported.
2.2. Materials and Methods
2.2.1. Chemicals
Methanol, ethanol, 2-propanol, 1,4-dioxane, acetonitrile, hexane, perchloric acid and acetic acid (LC grade) were supplied by Merck (Darmstadt, Germany). Carbohydrate standards (D-glucose, D-lactulose, D-mannitol), CoQ10, and α-tocopherol standards were purchased from Sigma Aldrich (Steinheim, Germany). ß-Cyclodextrin (food grade) was supplied by Xi’an Hong Chang Pharmaceuticals Co. (China), CoQ10 and ALA (pharmaceuticals grade) was obtained from Linyi Tianliheng Trade Co (China) and Lutein (food grade powder) from Shaanxi Sciphar Biotechnology Co., China.
Deionized (DI) water with a resistivity of 18.2 MΩ cm or greater was obtained from an Aqua Solutions (Jasper, USA) water purification system.
2.2.2. Experimental Procedures
Quantitative analyses of selected substances were done with different chromatographic, spectroscopic and electrometric methods. Concentrations of CoQ10 and lipoic acid were measured quantitatively using HPLC-APCIMS methods, α-tocopherol was measured with HPLC and fluorescence detector, lutein and zeaxanthin with HPLC and UV detector, sugars were evaluated with ion chromatography (IC) combined with pulsed amperometric detection (PAD) and ESI-MS/MS detection. Cholesterol was measured with HPLC-MS and quantitative HPTLC. All methods were originally developed and validated in our laboratory.
2.2.2.1. Quantitative Determination of Glucose
Glucose levels were quantified with Dionex ICS3000 liquid chromatograph (Sunnyvale, CA) equipped with EG eluent generator and Electrochemical Detector. Separation was performed with a strong anion-exchange stationary phase Dionex CarboPac PA10 analytical column (2 mm × 250 mm). Eluted carbohydrates were simultaneously quantified with Sciex QTRAP LC/MS/ MS system from Applied Biosystems/MDS (Sciex Concord, ON, Canada). Determination of glucose was carried out in Q1 MS scanning mode in the mass range from 150 to 200 m/z. A glucose quasi-molecular ion was formed with the added lithium ion [180+7]+ in positive ESI-MS ionization mode.
Plasma concentration was additionally confirmed with PAD, using standard four step potential waveform and originally developed two step potential waveform for quantitative determination of glucose in plasma samples [27].
2.2.2.2. Quantitative Determination of CoQ10
Before the analysis plasma samples stored at −80˚C were thawed in water bath at temperature 35˚C - 39˚C. Exactly 100 μL of plasma sample was transferred into the 4 mL plastic vials and 100 μL of para-benzoquinone (0.4 mg/ml in 1-propanol) was added. Mixture was vortexed for 30 seconds and then set aside for the next 15 minutes. In this time para-benzoquinone oxidized the reduced form of CoQ10. Additionaly, 400 µL of 1-propanol was added and solution was vortexed for 2 minutes followed by centrifugation for 4 min at 10.000 g on a tabletop centrifuge (Centrifuge Kendro Lab. Products, Heraeus, Biofuge, Stratos) to remove proteins. The 100 μL aliquot of supernatant was diluted into a 5-mL volumetric flask and analyzed with a HPLC-APCI-MS method [28].
Quantitative analyses of CoQ10 were done with Surveyor HPLC system (Thermo Electron corporation, San Jose, CA, USA), equipped with Hypersil Gold column 1.9 µm (50 mm × 2.1 mm) with 3 µm (Thermo Scientific (Runcorn, GB). Separated substances were detected with Finnigan-LCQ mass detector (Finnigan MAT, San Jose, CA, USA). The mobile phases used for gradient separation were: (A) a mixture of ethanol, methanol, 1,4-dioxan and acetic acid (65:30:5:0.1, v/v/v/v); and (B) 100% acetonitrile. The initial conditions were 12% A and 88% B. Mobile phase A was changed linearly and after 3 minutes reached 95% A. For next 3 min it was held at 95%. After that the HPLC system reverted immediately to the initial conditions for 2 min. At the flow rate of 0.5 mL/min and column temperature of 35˚C the retention time of CoQ10 was 4.7 ± 0.5 min. Identification and quantification of CoQ10 by MS in positive APCI ionization mode were done. Ionization discharge voltage was 6.0 kV, discharge current 5.0 μA, and source temperature 450˚C. Capillary voltage was 3.0 V, tube lens offset was −5.0 V, capillary temperature was 250˚C, sheath gas pressure was 0.2 MPa, auxiliary gas flow was 1.7 L/min, and injection time 5.0 ms. Chromatograms were obtained in SIM mode. Molecular mass (M+H)+ m/z 863.4 ± 0.5 was used for quantitative determination of CoQ10. It was possible to quantify cholesterol (M+H)+ m/z 386 ± 1 in the same run as CoQ10. Data were processed with Xcalibur 1.3. Software pack.
2.2.2.3. Quantitative Determination of α-Tocopherol
Plasma samples stored at −80˚C were thawed in water bath at temperature 35˚C - 39˚C. Exactly 200 μL of plasma sample was transferred into the 4 mL plastic vial and 200 μL of ethanol was added and solution was vortexed for 0.5 minute. In the next step 400 µL of hexane was added and vial was vortexed for another 0.5 minute followed by centrifugation for 2 min at 10.000 g on a tabletop centrifuge (Centrifuge Kendro Lab. Products, Heraeus, Biofuge, Stratos). The 20 μL of supernatant was injected into a HPLC (Thermo Electron Corporation, San Jose, CA, USA). Separation was done with Phenomex LiChrosorb column 5 µm (125 mm × 4 mm), the mobile phases was 1% 2-propanol in hexane and the flow rate was 1.0 mL/min. Identification and quantification of tocopherols were done with Hitachi F-1000 fluorescence detector (Hitachi Ltd, Tokyo, Japan). The excitation wavelength was 295 nm and the cut-off wavelength was 380 nm. The linear calibration curve was prepared in the range between 0.2 µg/mL do 20 µg/mL [29].
2.2.2.4. Quantitative Determination of Lipoic Acid
Plasma samples were thawed in water bath at temperature 35˚C - 39˚C. Exactly 300 μL of plasma sample was transferred into the 4 mL plastic vial and 600 μL of acetonitrile added and solution was vortexed for 0.5 minute. The vial was transferred into a cooled tabletop centrifuge (Centrifuge Kendro Lab. Products, Heraeus, Biofuge, Stratos) at 4˚C and centrifuged for 10 minutes at 10.000 g. Supernatant was analyzed with HPLC system combined with the Sciex QTRAP LC/MS/MS system equipped with TurboIonSprayTM source. Separation was done with Hypersil Gold column 1.9 µm (50 mm × 2.1 mm) equipped with adequate 3 µm precolumn Thermo Scientific (Runcorn, GB). The mobile phases were 1% 2-propanol in hexane and the flow rate was 1.0 mL/min. The method followed the procedure from the literature [30].
2.2.2.5. Quantitative Determination of Lutein and Zeaxanthin
Chicken plasma samples (200 μL) were pipetted into centrifuge tubes and 400 μL of hexane: 1-propanol, (1:1, v/v) was added. The content was vortexed for 1 minute. The vial was transferred into a tabletop centrifuge (Centrifuge Kendro Lab. Products, Heraeus, Biofuge, Stratos) and centrifuged for 10 minutes at 10.000 g. The upper layer was transferred into a vial and analyzed with HPLC with UV detector. Calibration curve with six standard solutions from 0.05 to 50 μg/mL of all-translutein was prepared.
Separation was done at room temperature 25˚C on Luna 3 μ C18(2) 100A column, 100 × 3.0 mm, with 3 μm particle size (Torrance, California, USA), equipped with a Phenomenex C18 guard column (Torrance, California, USA). Flow rate was 0.5 ml/min and injection volume was 20 µl. The mobile phases used for gradient separation were: methanol (solvent A), 1-propanol (solvent B) and water (solvent C). Gradient: t = 0 min, 85% A, 15% C; t = 1 min, 85% A, 15% C; t = 5 min, 40% A, 60% B; t = 10 min, 40% A, 60% B; t = 11 min, 5% A, 15% C; t = 16 min, 85% A, 15% C. Co-eluted ubiquinone was analyzed in UV range at 280 nm and lutein and zeaxanthine, were eluted together and measured in VIS range at 450 nm. ChromQuest 4.2 was used for the evaluation of the data.
2.2.2.6. GPx, SOD and Total Antioxidant Capacity
The methodology of Paglia and Valentine (1967) was used for measurements of GPx [31]. Measurements were done with colorimetric method (340 nm) with biochemical analyzer RX Daytona (RANDOX, Crumlin, UK) following the instructions of the kit manufacturer. The decrease in absorbance at 340 nm was directly proportional to the GPx concentration. The GPx activity was given in units related to gram of hemoglobin (U/g Hgb) measured with Technicon H*1 (SIEMENS, Munchen, D) automated hematology analyzer.
The activity of Superoxide dismutase (SOD) was measured with sensitive assay kit RANSOD (RANDOX, Crumlin, UK) according to the method of McCord and Fridovich (1969), containing the necessary reagents for convenient measurement of activity by colorimetric method at 510 nm, using the Randox RX Daytona biochemical analyzer [32]. The activity of SOD was given in units related to the amount (gram) of hemoglobin (U/gHgb) measured with Technicon H*1 automated hematology analyzer.
TAC (Total Antioxidant Capacity) was measured at 600 nm using reagent kit for TAS (Total Antioxidant Status, RANDOX, Crumlin, GB) on the automatic Randox RX Daytona biochemical analyzer. The method is based on the ability of antioxidant molecules to quench the long-lived ABTS•+, a blue-green chromophore with characteristic absorption at 734 nm, compared with that of Trolox, (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid) a water-soluble vitamin E analog. The amount of antioxidants was measured with the drop of absorbance at 600 nm (after 3 minutes). Results were expressed as mmol of Trolox per L of plasma.
2.2.3. Statistical Analysis
From data pre-arranged with Microsoft Excel 2007, we calculated basic statistical parameters (mean and standard deviation) using SAS/STAT (SAS Software, 1999).
3. RESULTS AND DISCUSSION
During the raise plasma samples were collected three times (on day 16, 28, 40) from four different groups (GC,  , GALA,
, GALA,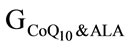 ) and several parameters were monitored and recorded. In the following figures and text we shall describe observed differences in concentrations of measured substances and variations in enzyme activeties.
) and several parameters were monitored and recorded. In the following figures and text we shall describe observed differences in concentrations of measured substances and variations in enzyme activeties.
3.1. Quantitative Determination of Glucose Plasma Levels
Harms produced by a protracted oxidative stress are somewhat connected with the carbohydrates, and that was the reason to select also the concentration of glucose as a parameter in our study. Several different analytical techniques from TLC to IC-PAD and IC-MS were used for quantitative determination [33,34]. Collected results (Table 2) show that normal concentrations of glucose in chicken plasma increased during the 41 day raise.
In the control group the final concentration, measured on day 40 was 20% higher than the starting concentration measured on day 16. In test groups, which administered fortified fodder the increase in glucose level in plasma was smaller than in the control group. In the case of administered CoQ10 the final concentration was 7% higher, and in the case of fodder with combination ALA and CoQ10 the final concentration was only 2% higher than on day 16. The rewarding result was obtained with pure ALA, where the final concentration was even 6% lower than on day 16. Our findings are comparable to previously published data [14] and show that exogenous CoQ10 and ALA as the food additives may reduce the plasma level of glucose. Our result confirms a beneficial effect of ALA, described in papers taking an interest in the diabetes.
3.2. Quantitative Determination of Total CoQ10
CoQ10 exists in nature and in the body in two forms: the oxidized form (ubiquinone) and reduced form (ubiquinol). Reverse chromatography with electrochemical detector was mostly used for quantitative determination [35]. The reduced form of CoQ10 in plasma can be stable at least 48 hour storage at −80˚C [36], and therefore we decided to measure the total concentration of CoQ10 in their oxidized form. As can be seen, the concentration of CoQ10 in plasma increased noticeably in all groups during the raise (Table 3).
In the control group the concentration of total CoQ10 in plasma increased for nearly 100% from starting concentration of 0.5 mg/L to nearly 1.0 mg/L at the end. In the test group feed with ALA the concentration of CoQ10 increased from 0.5 mg/L to 1.3 mg/L and the final plasma concentration was 0.3 mg/L higher than in the control group. Similar results were obtained in the GCoQ10&ALA supplemented group. Concentration of CoQ10 increased nearly 3 times, to final concentration 1.5 mg/L. The highest concentration was recorded in the test group using the fodder fortified with CoQ10. The final concentration was 360% higher than starting one, from standard value about 0.5 mg/L to nearly 1.8 mg/L.
Very high increase of total CoQ10 plasma concentration in test group supplemented with ALA was not expected, and it is a certain surprise. In our opinion the increased concentration is a result of in-line protection of CoQ10 due to the electrochemical potential of ALA. It is assumed, that ALA stabilizes the endogenous CoQ10, which is carried with appoproteins into the blood. Additional research will be necessary to confirm this thesis.
The final concentration in the group foddered with CoQ10 and ALA is unexpectedly lower than in the GCoQ10 group. Our explanation is that in the case of higher concentration of both substances the synergistic effect of LWMA in the antioxidant network takes place and CoQ10 is eliminated from the blood through the degradation of CoQ10 in liver. Liver functions are kept on a high working level and liver is not forced to produce new CoQ10, because the exogenous coenzyme keeps enough high concentration.
3.3. Quantitative Determination of α-Lipoic Acid
Lipoic acid can be found in the body as free and protein bounded form [15]. Concentrations of free ALA in plasma and animal tissues are very low. In chicken plasma concentration of ALA is in general, up to hundred times lover then adequate concentrations of CoQ10 and was in the µg/L range (Table 3). The reason for low levels of free ALA is very quick formation of protein-bound lipoic acid in plasma [14]. The bioavailability studies showed that concentration of ALA in plasma fall in relatively short time (40 minutes) after

Table 3. Influence of added CoQ10, lipoic acid and combination of CoQ10 and lipoic acid in chicken feed on CoQ10 concentration (mg/L), lipoic acid concentration (×10−1 mg/L), α-tocopherol concentration (mg/L) and lutein and zeaxanthin (mg/L) in plasma in groups (GC,  , GALA,
, GALA, ) during chicken raising; (n (GC) = 100, n (GCoQ10, GALA,
) during chicken raising; (n (GC) = 100, n (GCoQ10, GALA, ) = 50,
) = 50,  = mean, sd = standard deviation.
= mean, sd = standard deviation.
administration. In these circumstances the correct collection time of plasma samples may be crucial. Also in our analysis we observed some high unrealistic variations in the measured concentrations.
As we expected, measured concentrations of ALA in the control group and also in the test groups supplemented with CoQ10 were slightly lower at the end of the raise. It is known that concentrations of ALA are higher in young than in grown animals, and humans. When the fodder fortified with ALA was used the relative overall concentration increased significantly, however absolute concentration was still below mg/L. Also in this case the combination of CoQ10 and ALA was not very successful. Measured concentrations represent the free ALA, which has not yet been bound to a protein. Relatively lower concentration of ALA in the test group  may be explained with interacting effects of both substances. ALA is a strong antioxidant with a possibility to protect CoQ10 due to its electrochemical potential. The same trick is used in commercially available reduced form of CoQ10. 30 mg of CoQ10 (M = 865) and “only” 10 mg of ALA (M = 206) are packed into the same capsule; however, the high molar concentration of ALA is needed to support the stability of reduced form of CoQ10 in the capsules.
may be explained with interacting effects of both substances. ALA is a strong antioxidant with a possibility to protect CoQ10 due to its electrochemical potential. The same trick is used in commercially available reduced form of CoQ10. 30 mg of CoQ10 (M = 865) and “only” 10 mg of ALA (M = 206) are packed into the same capsule; however, the high molar concentration of ALA is needed to support the stability of reduced form of CoQ10 in the capsules.
3.4. Quantitative Determination of α-Tocopherol
Vitamin E deficiency is associated with several diseases therefore the Vitamin E is normaly added in chicken feed. Only the α-tocopherol was quantified due to its highest concentration in plasma.
Concentration of α-tocopherol in chicken plasma increased during the raise and was nearly 50% higher at the end than at the start of our experiment (Table 3 and Figure 3).
The same increase of 50% was recorded in test group fed with ALA and CoQ10. In the test group foddered with a food supplemented with the CoQ10 the final concentration was 60% higher and in the test group using fodder with ALA the final concentration was nearly 100% higher than the start concentration, Table 3. Our opinion is that increased concentration of α-tocopherol in the test group fed with ALA is the result of its actions in antioxidant network. The very high reduction power and its mobility in lipophilic and hydrophilic medium can directly reduce α-tocopherol and increase its concentration. The same direct access of administered antioxidant can be seen in the case of CoQ10 too. On the contrary, the results showed that the combination of two strong antioxidants; CoQ10, and ALA with different electrochemical potentials and different solubility is not so successful. The reduction of α-tocopherol is done according to the control functions of normal antioxidant network.

Figure 3. Relative changes of E vitamin concentrations in test groups fed with different fodder additives. Applied labels (—○—) control group, (—◊—) CoQ10, (—■—) ALA, (—Δ—) CoQ10 & ALA.
3.5. Quantitative Determination of Lutein and Zeaxanthin
The most prevalent carotenoids in the plasma of broilers are lutein and its structure isomer zeaxanthin. In our experiment we measured them in order to determine if supplemented CoQ10 and ALA with strong antioxidant properties had some impact on the plasma concentrations of carotenoids. Corn is an important natural ingredient in the chicken’s fodder and the total contents of carotenoids may overcome 10 mg/kg. In some circumstances they are the main carrier of antioxidant activities, but they are not a true member of an antioxidant network in an animal biological system [37]. The main difference is the way how they function. LMWA are reduced and oxidized reversibly to inhibit oxidizing species and are not destroyed in this process. While carotenoids function as singlet oxygen quencher. In this process the energy of the excited oxygen is transferred to the carotenoid molecules and is dissipated through surrounding solvent. In the physical quenching molecules are not destroyed and are used several times. When carotenoids reacts with free radicals in the presence of oxygen and are not strongly protected by some other antioxidants, for instance vitamin E, their mission, which is normally not destructive, is transformed into suicidal peroxidation. The higher the concentration of oxygen or carotenoid the more devastating is the effect of auto-oxidation. The mechanisms of action of carotenoids regarding their antioxidant properties are much less understood than are the properties of “other” LWMA.
Due to expected increased antioxidant activity we decided to monitor the concentrations of lutein and zeaxanthin in plasma samples collected during the breeding period. In Table 3 the influence of added CoQ10, lipoic acid and combination of CoQ10 and lipoic acid in chicken feed on selected antioxidants in plasma of tested groups of chickens is presented. Obtained results are still informative, however if we want to know the real contribution of physical antioxidants and suicidal molecules in integral antioxidant system we have to carry out more experiments with different combinations of interesting molecules.
3.6. Biochemical Analysis
TAC is a biochemical parameter suitable for evaluating the overall antioxidant status of serum or plasma. According to our expectations the value of total antioxidant capacity in chicken plasma decrease during the raise, see Table 4.
The biggest decrease was recorded in control group, in which the final value was 20% lower than at the start. Smaller decrease was observed in the test group administered CoQ10. Even better result was obtained in the group foddered with a combination of ALA and CoQ10. A very good result was achieved in the case of the fodder fortified with lipoic acid. Our results show that fodder additives can significantly influence on TAC values in plasma. Furthermore, ALA exhibits the very high antioxidant potential in protection of animals exposed to oxidative stress.

Table 4. Influence of added CoQ10, lipoic acid and combination CoQ10 and lipoic acid in chicken feed on TAC (mmol/L), SOD content (U/mL) and GPx content (U/mL) in plasma in GC,  , GALA,
, GALA,  groups during chicken raising; (n (GC) = 100, n (
groups during chicken raising; (n (GC) = 100, n ( , GALA,
, GALA, ) = 50,
) = 50,  = mean, sd = standard deviation).
= mean, sd = standard deviation).
The GPx values increased during the experiment in all groups, but much higher increase was obtained in test groups using some fodder additive, the increase of control group was 20%. Six times higher increase of 120% was recorded in ALA test group. Lower than expected increase was noticed in  and
and  groups. Measured values were about 60% higher than in control group (Table 4). Increased GPx activities may be connected with improved function of entire antioxidant network. Very high influence of ALA can be explained with high reactivity, movability and solubility of ALA in lipophilic and hydrophilic media and the lowest electrochemical potential of −0.320 mV. All these characteristics support the direct antioxidant access of ALA to all tissues in organism, especially in plasma.
groups. Measured values were about 60% higher than in control group (Table 4). Increased GPx activities may be connected with improved function of entire antioxidant network. Very high influence of ALA can be explained with high reactivity, movability and solubility of ALA in lipophilic and hydrophilic media and the lowest electrochemical potential of −0.320 mV. All these characteristics support the direct antioxidant access of ALA to all tissues in organism, especially in plasma.
The measurements of SOD activities in chicken plasma show a constant increase from the start. On day 40 the activity is 18% greater than on day 16. In the case of fortified fodder the activities of SOD were reduced. The lowest value was obtained in the group administered with ALA, a little higher activity was obtained in the case of fodder supplemented with ALA and CoQ10 and the smallest reduction in activity of SOD was measured in the group with CoQ10, Table 4. According to our opinion the possible explanation may be that the increase of SOD in control group was the result of stronger oxidative stress during the raise, and the reduction of SOD activities could be a result of the preventive effect of LWMA (CoQ10 and ALA) in the antioxidant network.
In our experimental work a large amount of data were collected. In order to simplify the interpretation of obtained data we prepared a simplified presentation scheme. Pre-experimental values (starting values) measured at the start on the day 16 were taken as hundred percent. All measurements collected on the days 28 and 40 were normalized to the starting values of 100% on the day 16. In the final step values of the relative changes were added. Obtained sums represent the integral changes which appeared at each measured parameter, during the 40 day chicken production raise and 25 days of administration of fortified fodder. In presented results it is necessary to consider that relative percents may often show an excessive estimation, but predicted trends are correct. It is shown that the basic concentrations of ALA decreased, which is seen in control group and also in CoQ10 group. A very high increase was recorded when fodder with ALA and combination of ALA and CoQ10 were used. According to our expectation CoQ10 additive cannot exert influence on the level of ALA. In the case of simultaneous application of CoQ10 and ALA the synergistic effect is seen, but is not so pronounced as in the case of addition of pure ALA (Figure 4).

Figure 4. Relative changes of concentration of three net antioxidants (ALA, CoQ10, Vitamin E), cholesterol and glucose and changes of activities of some enzymes (SOD, GPX) and total antioxidant capacity (TAC) in chicken plasma during the experiment are shown. Three test groups were fed with food fortified with ALA, CoQ10 and combination of both substances. All measurements were normalized to the starting values.
While observing the changes in total CoQ10 concentrations it is seen that during the raise period basic level of total CoQ10 increased. Administration of CoQ10 additive provoked a very high increase of blood level of CoQ10. A significant increase was recorded in ALA test group. When both additives were used simultaneously the total increase in CoQ10 concentration was higher than in ALA group and lower than in CoQ10 group. Non-additive increase may be a result of synergistic effect of both substances in antioxidant network.
During the raise the concentration of α-tocopherol in chicken blood plasma increased, which is seen in the control group. Fodder additives increased the integral level of α-tocopherol. Once again, it is seen that the combination of two additives with antioxidant properties did not produce additive result and the final effect was the result of a synergistic action. It is possible to notice a very high protective effect of ALA.
The level of cholesterol decreased comparably in all four groups during the experiment. This means that selected fodder additives had no effect on cholesterol plasma level in our experiment.
A very meaningful is a small increase of glucose concentration in test groups consuming CoQ10 and combination of CoQ10 and ALA. Final concentration is much smaller than the concentration in the control group. In the case of pure ALA the overall decrease was recorded. This is in good correlation with a data from literature in which a positive effect of ALA on reduction of glucose in blood is observed. Lower blood level of glucose is the result of deactivation of substances blocking the insulin penetration in cells. This action can have a very positive effect in diabetes type 2.
Changes in enzyme activities recorded in determinations of SOD, GPx in TAC show the positive effect of applied additives with antioxidant activities on antioxidant network. Outstanding results were obtained with the use of ALA. These results show the strong influence of ALA with active tiol groups on the antioxidant defense.
Some preliminary determination of plasma concentrations of lutein and zeaxanthine in chicken plasma were also made. Both carotenoids are important components of chicken fodder. Collected results show that the sum of both carotenoids decrease during the raise in all four groups. When fodder additives CoQ10 and ALA were used the decrease was smaller than in the control group (Table 3).
At the beginning of our experimental raise we did not plan to measure the plasma level of carotenoids, because we tried to collect as much as possible the information about chemical antioxidant. Later we found out that also the measurement of carotenoids may be a convenient and useful indication of the body's overall antioxidant status. The reason for this is that carotenoids are delivered to tissues by LDL circulating in the blood and their concentrations are correlated with the amounts of the other fat-soluble antioxidants in the body such as vitamin E or CoQ10. Therefore, increased levels of carotenoids reflect overall levels of antioxidant defense and diminished oxidative stress. In addition, non invasive methods have been developed based on an optical method known as Resonance Raman Spectroscopy [40]. This method has been used for many years in research laboratories for carotenoid investigations in biological systems using optical signals, called raman signals. These signals identify the unique molecular structure of carotenoids, allowing their measurement without interference by other molecular substances.
In the next step we intend to develop an in-line control system which will monitor the intensity of oxidative stress. For this reason we need to know the correlation between concentration of carotenoids and concentrations of LWMA. If we succeed, this will be an enormously important development, since the presence of scanners for use in the field studies brings the possibility of in-line assessing antioxidant and nutritional status of animals during the raise.
4. CONCLUSIONS
In our research work we assume that participating antioxidants form a network which carry out regulation, inducibility, interactivity and balance between the members. To test such hypothesis it was necessary to develope suitable methodologies for assessing the amount of antioxidants and their activity in tissues and biological fluids. Obtained results may give us more rational recommendations for dietary intake, and help to explain the effects of specific antioxidants. Today, still many questions accompany the beneficial or harmful effects of LMWA supplementation.
Experiments on animal were carried out in accordance with the animal protection law, which is valid in the Republic of Slovenia and is controlled by the Ethic Commitee of the Ministry of Agriculture, Forestry and Food of the Republic of Slovenia.
5. ACKNOWLEDGEMENTS
This work was supported by the Slovenian Research Agency (Research Project L1-2174) and the Perutnina Ptuj, d.d. The authors wish to thank Dr. Roman Glaser, Brigita Vindiš-Zelenko and Boštjan Donša from Perutnina Ptuj, d.d.