Design and Multicriteria Optimization of a Two-Stage Reactive Extrusion Process for the Synthesis of Thermoplastic Polyurethanes ()
1. Introduction
Polyurethanes are known to be amongst the most versatile materials in the world [1,2]. They can be tailored by catalyzed polyaddition reactions between various polyols and polyisocyanates [3-9] which, due to their functionality, can lead to the formation of linear, branched or crosslinked macromolecules containing a significant number of urethane groups (-HN-COO-), regardless of the nature of the rest of the molecules [10].
This diversity has given ground to a wide variety of applications ranging from rigid and flexible foams to structural and coating elastomers, adhesives, TPUs appropriate for medical devices, leather-like materials, sealants and auxiliary agents.
TPUs, which constitute the subject of this study, are linear polymers in which the principal chain structure is composed by two sections R1 and R2 connected via polar urethane groups (~R1-NH-COO-R2~). In this context, R1 denotes an aliphatic, aromatic or alicyclic radical derived from the isocyanate monomer while R2 denotes a more complex group derived from the polyol component (polyether or polyester). TPUs are typically obtained via a stepwise, polyaddition reaction between diisocyanates and bi-functional polyols with hydroxyl terminal groups, according to the following general scheme:
 (1)
(1)
It must also be noted that TPU chains do not contain only urethane structures. Depending on the specifications of feeds and on the method adopted for the polyaddition process, one can also find urea groups, biuret groups, allophanate groups, carbodiimide groups, aromatic hydrocarbon rings, isocyanurate or oxazolidone structures, and even ionic groups in some cases.
As a result of their physical and mechanical properties, associated to their good stability, low free surface energy, physiological inertness to living organisms and resistance to biodegradation, specialty TPUs are widely used as biomedical materials [11,12] capable of preventing inflammation of tissues, destruction by body fluids and deposition of blood components [13-16].
In most industrial cases, they are manufactured using traditional discontinuous processes which often give rise, from batch to batch, to products with inconstant quality.
The aim of the present work will be to develop and optimize a continuous process able to produce linear, solvent free and pure TPUs with controlled structure and specific end-use properties making them appropriate for on-line processing medical probes, catheters, cardiac valves and vascular prosthetic and endoprosthetic devices. In such case, reactive extrusion presents significant advantages as it can be used under well controlled operating conditions (feed rate, temperature, mixing rate, etc.).
This technique has already been widely exploited for the synthesis [17-22], grafting [23], hydrolysis [24] and depolymerization of TPUs [25], as well as for the production of oligomers [26]. However, in most cases, all the reactants were introduced into the extrusion machine in a single stage.
In the present study, we propose to elaborate TPUs in a two-stage process:
• First, a prepolymer will be synthesized in a batch reactor through catalyzed reactions between a macrodiol and a diisocyanate;
• The resulting prepolymer and a chain extender will then be introduced, under controlled operating conditions, into a twin-screw extruder to elaborate TPU chains.
It is expected that this procedure will contribute to the production of homogeneous TPUs with constant quality.
This work will start with a preliminary study devoted to determine the operating parameters for which the polymerization time and the average residence time of the reactants in the extruder must be of the same order of magnitude.
Then, an experimental strategy will be developed to study and model the effect of the extrusion parameters on the properties and the purity of the resulting TPUs.
The final objective will be to find a set of optimal operating conditions for the production, with the lowest energy consumption, of TPUs with targeted properties and high purity.
Accordingly, this optimization will be confronted with a multiobjective decision problem for which a unique solution that yields optimal values for all the objective criteria rarely exists. This is a common scenario in many industrial production optimizations which dictates the implementation of a decision-maker, in order to choose the best tradeoffs among all defined and conflicting objectives.
The methodology that will be used in this work has already been successfully applied to different polymerization processes [27-32]. It will be briefly described in the next section, while its specific application to this process will be discussed later.
2. Multicriteria Optimization Methodology
2.1. General Considerations
To develop industrial processes, companies need to obtain the desired products quality associated to the highest productivity and to the lowest cost investments. To reach these objectives, a multicriteria optimization of the process is necessary. In engineering processes, multiple objectives have usually been combined, often through linear [33] or empirical [34] combination, to form a scalar objective function. Another classical method consists in optimizing one criterion and setting up constraints on the others [35]. These techniques depend on the first user’s choice, so preferences can bias the result.
Methods incorporating a domination criterion are often more interesting because they are more general, more accurate and without any a priori knowledge. The aim is to find a non dominated zone in which a decision maker will be able to choose the best solution. This region, called Pareto’s zone, is the set of all non dominated points. It can be obtained using Pareto’s domination concept which is defined in such manner that a solution (compromise or vector), x1, dominates another solution, x2, if it is better or equal for all criteria and strictly better for at least one criterion [36]. The study does not allow to find immediately the preferred solution but to exclude all conditions which are not interesting. Two terms are used: Pareto’s domain and Pareto’s front which are related to the input variables and output criteria respectively.
Pareto’s domain can be approximated by a large number of possible solutions using an evolutionary algorithm [27,36-42]. It constitutes important information for the industrialist who will then have to classify these solutions.
Multiobjective optimization is commonly realized in a three-step procedure:
• Modelling of the process to encapsulate the underlying phenomena that relate all input and output process variables;
• Reduction of the decision space to include only the non-dominated solutions, providing an approximation of Pareto’s domain, using evolutionary algorithms;
• Ranking of all the solutions contained in Pareto’s domain using preferences from a human expert in order to choose the best compromise.
The most difficult step is usually the third one as it relates to a human-centred process that exhibits, by nature, a higher level of fuzziness. As a consequence, it is necessary to develop systematic procedures to unequivocally capture the preferences of the expert on the basis of his knowledge of the process.
In this respect, all the output criteria could be viewed as particular objective functions that need to be optimized. However, some of them are conflicting, making the optimization task significantly more difficult.
2.2. Methods for Ranking the Non-Dominated Solutions
A number of different methods have been proposed in the literature for performing the ranking procedure. In the present work, two methods will be used and compared, namely the “Net Flow” and the “Rough Set” methods. The main difference between these methods relies on the way the preferences of the experts are captured and used to determine the optimal zone of operation. It must be noted that it is not the objective of this study to detail these well-established methods, but rather to test, for this specific problem, whether convergent results can be obtained using them.
2.2.1. Net Flow Method (NFM)
NFM is a hybrid of two outranking methods (ELECTRE and PROMETHEE). It uses the knowledge of the objective criteria values and compares all non-dominated solutions, in a pairwise manner, to show a possible outranking before a total synthesis of the alternatives.
The user has to express several parameters to define his preferences.
First he must introduce a weight, wk, for each criterion k which serves as index of its relative importance. In the algorithm, these coefficients are normalized:
 (2)
(2)
The decision maker has also to define indifference, qk, preference, pk, and veto, vk, thresholds for each criterion. The indifference threshold is defined so that two alternatives cannot be differentiated below it. The preference threshold is defined to show the real preference of one alternative against another while the veto threshold is defined like a constraint if an alternative is too bad in one criterion. In this respect, an alternative is penalized if one of its criteria is over the veto threshold compared to another alternative, even if it is considered as a good alternative for the other criteria. Accordingly, these three thresholds are defined, for each criterion, in such a way that:
 (3)
(3)
The criteria difference for each pair of alternatives, [i, j], and for each criterion, k, is:
 (4)
(4)
for ,
,  ,
,  and
and , and where fk(i) is the value of the kth criterion of alternative i, and hk is an optimization indicator (hk = 1 if fk is to be maximized and hk = −1 if fk is to be minimized).
, and where fk(i) is the value of the kth criterion of alternative i, and hk is an optimization indicator (hk = 1 if fk is to be maximized and hk = −1 if fk is to be minimized).
A global concordance index, C[i, j], is then calculated as with ELECTRE III for each pair of alternatives (for i = 1, ∙∙∙, m and j = 1, ∙∙∙, m) [43].
The preference of one alternative over another is named “concordant” up to the indifference threshold and the local concordance index is then equal to 1. On the contrary, this preference is named “not concordant” up to the preference threshold and the local concordance index is then equal to 0. Between these two thresholds, a linear approach is used again to define the local concordance index:
 (5)
(5)
where
 (6)
(6)
A discordance index Dk[i, j] is also calculated for each criterion, k, as with ELECTRE III, to take into account a bad criterion value which allows to relegate the concerned point in the total ranking (for i = 1, ∙∙∙, m, j = 1, ∙∙∙, m and k = 1, ∙∙∙, n):
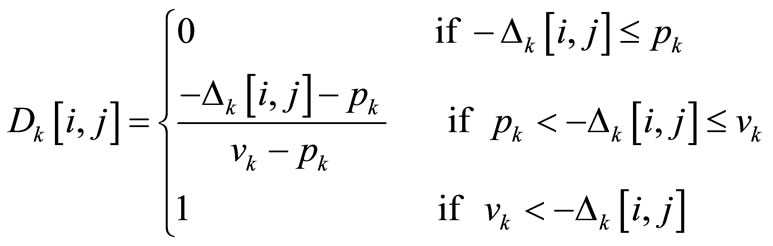 (7)
(7)
The preference of an alternative versus another is called “discordant” up to the veto threshold and the discordance index is then equal to 1. On the contrary, it is named “not discordant” up to the preference threshold and the discordance index is then equal to 0. Between these two thresholds, a linear approach is used to define the discordance index.
Using the concordance and the discordance indexes, outranking degrees, σ[i, j], can be generated for every pair of alternatives. These outranking degrees are obtained using the following formula (for i = 1, ∙∙∙, m, j = 1, ∙∙∙, m) [44]:
 (8)
(8)
i outranks j all the more than the outranking degree is close to 1 .
.
The resulting outranking relation sets may be formulated in the form of an outranking matrix. From these outranking degrees, two preorders are established, as with PROMETHEE, by the outgoing flow  and the incoming flow
and the incoming flow  (for i = 1, ∙∙∙, m) [45].
(for i = 1, ∙∙∙, m) [45].
 (9)
(9)
Finally, the total ranking of the alternatives, i, is determined from the net flow with possible ex æquo (for i = 1, ∙∙∙, m):
 (10)
(10)
The alternative, with the highest net flow, is considered as the “best” solution, while the one with the lowest net flow is considered as the “worst” solution, also known as “nadir”.
2.2.2. Rough Sets Method (RSM)
RSM is based on the Rough Set theory originally introduced by Pawlack [46-48] for ranking a large number of feasible solutions from Pareto’s zone. It has been used in several applications [49-54].
The classification of the compromises constitutes a “measurement” of the decision maker’s preference. A human expert classifies a small number of significant alternatives of Pareto’s Domain, in a preference descending order. Decision rules are then extracted in the form of a binary code (bits), thus creating a form of expert system. This decision-maker profile of preferences is subsequently applied to all the compromises. This allows different preference zones to be established.
The Rough Set theory uses a tabular representation of the preferential information expressed by the decision maker. The lines of this table correspond to objects whereas its columns correspond to attributes.
In the present study, the resulting table will be similar to the one suggested by Greco et al. [55]. More explanations will be given in Section 4.2.4.
In the original theory of Rough Sets, the rough approximations were performed using an indiscernibility relation on the pairwise comparison table. These approximations might result in inconsistencies between the generated rules, as discussed by Greco et al. [51]. To overcome this imprecision drawback, a different set of inconsistency rules [56] (Zaras (2001)) will be used in the present work. Finally, the finite set of solutions, representing the entire Pareto’s front, will be ranked by order of preference.
To achieve this classification, the Rough Sets Method uses the following steps:
• A small number of solutions (between 5 and 10) from different regions of Pareto’s zone are selected and presented to the expert who must classify them in a preference descending order (preferences measurement);
• Pairwise comparison of the solutions enables the extraction and validation of the “preference/no preference” rules, according to the RSM;
• These rules are subsequently applied to all the compromises, using the following principle: when two compared solutions are in accordance with a preference rule, then (+1) is added to the score of the first solution and (−1) is added to the score of the second solution;
• Finally, the resulting solutions are ranked in a scoredecreasing order.
3. Materials and Methods
3.1. Chemical Products and Reactions
Polyurethanes were synthesized using the products given in Table 1.
The first stage of the process was carried out by reaction between HMDI and PTMG to obtain the prepolymer (DI), made up of a flexible segment (PTMG) bearing one isocyanate function at each end. The corresponding reaction scheme is:
 (11)
(11)
With R: -(C6H10-CH2-C6H10)-.
In the second stage, the two isocyanate functions of DI reacted with the chain extender (EX) to give a blockpolyurethane composed of flexible segments alternating with the rigid ones. The chain extension took place according to the following reaction scheme:
 (12)
(12)
The prepolymerization was carried out in the presence of a catalyst, (dibutyltin dilaurate: DBTDL), which remained present and active during the chains extension stage as well. DBTDL concentration was 1 w% compared to the initial reactional mixture of PTMG and HMDI. Stoechiometry between HMDI, PTMG and EX was 2:1:2.
PTMG was used above its melting temperature (33˚C - 36˚C). Three PTMG were used with respective number average molar masses: MPTMG = 1000, 1400 and 2000 g/mol.
3.2. Reactors and Their Environment
Two batch reactors were used to develop the preliminary

Table 1. Main physical characteristics of the reagents.
studies:
Reactor 1 (i.e., the prepolymerization reactor) was a 200 mL jacketed glass batch reactor equipped with a stirrer, a reflux condenser, a cryostat, a sampling device and a nitrogen-supply system. The stirrer used was composed of a pitch blade turbine. Its rotation speed was kept constant at 500 rpm.
Reactor 2 was a Haake Rheocord-Rheomix internal mixer 540 p, equipped with a device which makes it possible to follow the time evolution of the torque and the temperature of the molten product.
It was composed of a basic device (Rheocord RC300 p) and a measuring cell (Rheomix R540 p). The whole system was controlled by a computer interface between the basic apparatus and the mixer, which was composed of a chamber with two rotating propeller-shaped rotors. The volume of this chamber was 50 mL, limited by a stop valve driven by a piston. The back wall, the intermediate room and the frontal plate were heated electrically and were equipped with independent control loops. A thermocouple, placed on the frontal plate of the mixer, measured the matter temperature. Cooling was achieved via channels crossed by compressed air and placed in the intermediate zone.
A Pilot unit for the chains extension was designed. It was composed of:
A prepolymerization batch reactor and its environment which includes 1) a tank for the storage of HMDI, equipped with a drainage sluice and a centrifugal pump, 2) a double-walled tank for the storage of PTMG, connected to a thermostat, an agitator, a drainage sluice and a gear pump, 3) a 15 liters capacity reactor, equipped with an agitator, feeding devices for HMDI, PTMG and the catalyst, a drainage sluice, a gear pump and temperature sensors, 4) an automat allowing to control the temperature and the feed rates of the reagents. Another tank was also available to store the solvent (Tetrahydrofuran) which was used for the cleaning of the two tanks and the reactor.
A corotative twin-screw-extruder (CLEXTRAL, BC21), equipped with interpenetrated screws and connected to the prepolymerization reactor via a gear pump and a flow-meter.
The extruder was also connected to a tank containing the chain extender via an HPLC pump.
The screws profile was determined so as to obtain a good mixture of the prepolymer and the chain extender and to maximize the residence time in the extruder, this in order to obtain the highest conversion. This profile included four elements of transport to quickly transport the prepolymer and the chain extender to the first zone of mixture, a regular alternation of elements of transport, elements with inversed steps and elements of mixing of variable steps and lengths. These last two types of elements allowed the formation of a pressure zone. The product accumulated in the threads of the upstream transport screws, thus increasing the rate of filling of the extruder and consequently the residence time of the reacting mixture.
The temperature control of the extruder barrel was achieved by means of electrical resistances and water circulation in the jacket of each constitutive element of the barrel, thus making it possible to establish the desired temperature profile along the barrel.
3.3. Analytical Methods
3.3.1. Number and Weight Average Molar Masses
The number and weight average molar masses were determined by size exclusion chromatography (SEC), using two detectors: a multi-angle laser light scattering (MALLS) apparatus (Dawn DSP-F) and a differential refractometer (Waters 410, Millipore).
Elutions were performed at 35˚C with tetrahydrofuran (THF) containing di-tertiary-butyl-2.6 methyl-4 phenol as stabilizer. The flow-rate was 1 mL∙min−1. The concentration of the polymer solutions and the corresponding injected volume were 1 g/L and 25 µL respectively. Prior to chromatography, THF and polymer solutions were passed through a Nylon filter of 0.45 µm porosity.
The SEC assembly consisted of a degasser, a Waters 510 Millipore pump, a U6K Millipore injector, a precolumn, two chromatographic columns assembled in series and filled with linear ultrastyragel and an electric oven to control the temperature of the columns. Data from the two detectors were acquired and exploited by means of the software Astra from Wyatt Technology which allowed the determination of the molar mass distribution and the number and weight average molar masses of the samples.
3.3.2. Residual Yield of Prepolymer (TDI)
TDI is given by:
 (13)
(13)
With:
 : TPUs number average molar mass;
: TPUs number average molar mass;
MDI = 2 (MHMDI) + MPTMG + 2(MEX) = 705 + MPTMG where: MPTMG is 1000, 1400 or 2000 g/mol.
3.3.3. Glass Transition Temperature and Young’s Moduli
The glass transition temperature, Tg, as well as the temperature evolution of Young’s modulus, were obtained by dynamic mechanical thermal analysis, using the tensile testing machine DMTA V 902-50010 of Rheometric Scientific. This apparatus was coupled with a computer equipped with Orchestrator software which provided instantaneously the values of the shear storage modulus and loss modulus. Moreover, the apparatus was used in the “autostrain” mode, i.e. the imposed sinusoidal deformation was automatically adapted so that the measured stress remained higher than the sensitivity of the sensors. The initial distance between the jaws of the tensile testing machine was fixed at 6 mm.
Measurements were carried out on rectangular samples of 20 mm length, 13 mm of width and 2 mm of thickness which were obtained by using a manual hydraulic press. The operating conditions were the following:
• Test in three points.
• Frequency: 10 Hz.
• Constant deformation (0.1%).
• Temperature range: between −100˚C and +80˚C.
• Temperature rise speed: 3˚C∙min−1.
From the acquired data, the ratio of Young’s moduli, determined at Tg − 20˚C and Tg + 20˚C was subsequently calculated.
4. Results and Discussion
4.1. Preliminary Study
A preliminary study was carried out, in batch reactors and with moderate quantities of reagents, in order to define the operating conditions (temperature, catalyst concentration and mixing speed) which could be used to develop the continuous extrusion process. The first part of this study was focused on the kinetic modeling of the prepolymer synthesis while, the second part, dealt with the evaluation of the reaction time of the chains extension and the feasibility of the overall process.
4.1.1. Kinetic Model of the Prepolymer Synthesis
Initially, a set of experiments were carried out under the following operating conditions:
• Temperature: 100˚C.
• PTMG: 90.875 g.
• HMDI: 34.125 g.
• Catalyst: 3 different concentrations: 0.001 w%, 0.01 w% and 0.1 w%.
Samples were taken out at various reaction times and the weight concentration of unreacted isocyanate (NCO functional groups) was determined by a back-titration [57] (Otey et al., 1964).
The corresponding experimental data were subsequently used for the construction and the validation of a mathematical model of the prepolymerization kinetics which, in addition to the primary reaction given in scheme (11), considered also:
• One of the most important secondary reactions between urethane and isocyanate functional groups, which takes place after a certain conversion and results to the formation of allophanates, according to the following simplified mechanism: 
 (14)
(14)
• The influence of the catalyst concentration on the reaction kinetics.
The rate of consumption of isocyanate functional groups was expressed by the following equation:
 (15)
(15)
where k1 and k2 are the rate constants of the primary and secondary reactions respectively. The technique of the minimum squared deviation between experimental and theoretical values was used to determine the values of these rate constants.
The results showed that:
• k1 depends on the catalyst concentration according to the following relation:
 (16)
(16)
and,
• k2 is constant and equal to 9.45 × 10−3 (kg/mol/s).
Figure 1 depicts the time evolution of NCO-groups conversion for the 3 catalyst concentrations and demonstrates:
• The good agreement between simulated and experimental data;
• That at 100˚C, the maximum conversion is obtained after around 180, 900 and 1100 seconds with 0.1, 0.01 and 0.001 w% catalyst concentration respectively.
4.1.2. Study of the Overall Process Using Haake Internal Mixer
The overall polymerization was carried out according to the following procedure:
In a first step, PTMG and HMDI were introduced in the mixing chamber and heated at 100˚C. The catalyst was then introduced (at a concentration of 0.1%). After 10 minutes of reaction, which exceeds the maximum time required to achieve the prepolymer synthesis, the temperature was raised to 160˚C and the chain extender was subsequently added.
The mixing speed of the first stage varied at each run from 5 to 200 rpm while that of the second stage was fixed at 160 rpm. The mixing torque was continuously recorded. During the reaction, the molar mass of the resulting TPU increased thus increasing the viscosity of the mixture as well. Hence, the mixing torque served as an indirect measurement of the progress of the reaction.
Figure 2 illustrates this interesting aspect showing clearly three distinct domains. In the first one, the value