High Resolution Gas Adsorption Study on Montmorillonite Clays from Côte d’Ivoire: Assessment of Surface Energetic Properties ()
1. Introduction
The strong increase of the population and the uncontrolled industrialization policy in Côte d’Ivoire are the results of a more and more increasing production of solid and liquid wastes [1]. The management of these wastes is generally restricted to a simple collection and a setting of illegal dumping without previous treatment [2].
The discharges, sum total of all types of waste, constitute polluted sites. Indeed, this mode of management of waste presents potential hazards of environmental pollution [3,4]. In order to limit the expansion of the pollution and to guarantee the protection of the environment, the creation of Technical Burying Centers (C.E.T. in French) is considered by using clay materials. Indeed, Côte d’Ivoire has a lot of clayey deposit that has been studied [5-7]. In a lot of processes that make with the clayey material, it is necessary to determine with precision the distribution of the different faces, to study in good conditions the mechanisms that have place and the properties that result. Therefore a better knowledge of surface properties of these clay minerals is necessary.
Currently the approaches based on gas adsorption appear as the most effective in the study of the heterogeneity of materials [8-11].
The acquisition of precise isotherms from the very low pressures, coupled with the modelling of the derivative isotherms today, permits us to reach numerous informations on the superficial energizing properties of the solid. To this effect, the use of the DIS method permits the quantification of the basal and lateral surfaces, and therefore the determination of the total surface of the studied materials [12,13].
The present study is concerned two montmorillonite clays from the deposit of Katiola (Côte d’Ivoire). It is a contribution to the literature on external properties of the argillaceous materials, which are likely to be employed like alternate materials in the technology of environmental protection [14-16].
2. Experimental
The montmorillonite clays (referenced to as K1 and K2) used in this study were collected from Katiola (in the center of Côte d’Ivoire). The mineralogical composition of these clays were reported to be 74.5% of montmorillonite, 7% of kaolinite and 5.4% of illite associated with inter-stratified clay (13.5%) for K1 and 73.4% of montmorillonite 23.5% of kaolinite and 3.1% of illite for K2 [5].
To prevent the effect of exchangeable cations on the gas adsorption, the samples were purified and subsequently saturated with Na+ cations by washing the clay fraction ( < 2 μm) repeatedly with concentred solution of NaCl. Twenty grams portions of the clay were treated with 1 mol·L–1 NaCl solution. The suspension was vigorously shaken at room temperature with magnetic agitation for 1 h. Then, the cation-saturated montmorillonite was separated by centrifugation (30 min at 19,000 rpm) and decantation of the supernatant solutions [17]. This process was repeated three times. The cation-saturated montmorillonite was then washed three or four times with distilled water until the AgNO3 test for chloride became negative [18,19].
< 2 μm) repeatedly with concentred solution of NaCl. Twenty grams portions of the clay were treated with 1 mol·L–1 NaCl solution. The suspension was vigorously shaken at room temperature with magnetic agitation for 1 h. Then, the cation-saturated montmorillonite was separated by centrifugation (30 min at 19,000 rpm) and decantation of the supernatant solutions [17]. This process was repeated three times. The cation-saturated montmorillonite was then washed three or four times with distilled water until the AgNO3 test for chloride became negative [18,19].
The cation-saturated montmorillonite was subsequently dried during three to five days at 40˚C and finally ground in an agate mortar. X-ray diffraction was achieved on the powder gotten with a Brucker’s D8 diffractometer that uses Co-K monochromatic radiation (λ = 1.7889 Å). Adsorption isotherms of nitrogen and argon, studied by a quasi-equilibrium gas adsorption procedure [20,21], were achieved after out gassing at 110˚C during 24 hours. The pressure measurement is achieved with two absolute 0 Pa - 0.1 Pa and 0 Pa - 10 Pa sensors Edwards, with an automatic adsorbometer equipped with a turbo molecular pump, in order to ensure a dynamic vacuum up to 10–8 Pa. The measurements of the saturation pressure of nitrogen and argon at 77 K are done with an independent system equipped with a 0 Pa - 10 Pa sensor Edward.
This method permits one to acquire many experimental data. Then, it is possible to calculate an experimental derivative of the isotherm with respect to logarithm of the relative pressure. Such curves display peaks and valleys that can be modelled with a restricted number of local isotherms by using the Derivative Isotherms Summation (DIS) method [12,13].
3. Result and Discussion
3.1. X-Ray Diffraction
The X-ray diffraction patterns obtained from Na-saturated clays are shown in Figure 1. The results indicate a centred major peak of about 12.2 Å, corresponding to the basal spacing d(001). This position of observed refection (001), translates a basal distance corresponding to the state hydrated means to a layer of water of exchangeable sodium cation. The peak towards 4.521 corresponds to an (hh0) reflexion. The other peaks 3.107 and 2.539 Å are harmonic with the main stripe of the Na-saturated montmorillonite.
The X-ray diffraction patterns of K2 exhibits a peak at 7.3 Å, which is a harmonic to those at 3.542 Å that translates a trace of kaolinite. Purification of K2 sample was therefore not complete.
3.2. Gas Adsorption
3.2.1. Derivative Isotherms of Argon Adsorption
The experimental derivative isotherms of argon adsorption on K1 and K2 are given respectively in Figures 2 and 3. The derivative isotherms were modelled in a satisfactory
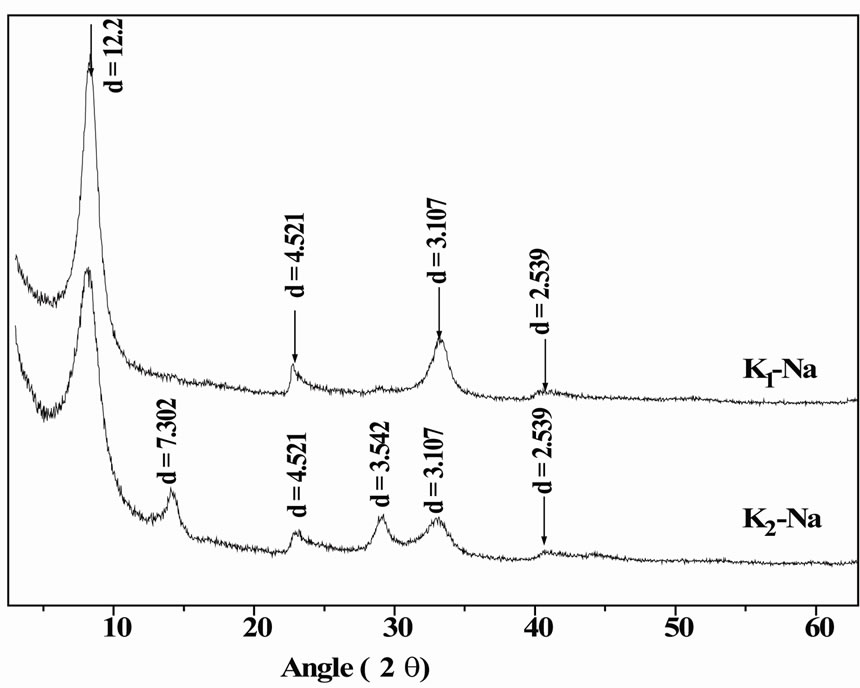
Figure 1. Comparison of X-ray diffraction patterns of cation-saturated clays (K1-Na and K2-Na).

Figure 2. Result of the decomposition by the DIS method of the derivative adsorption isotherm of argon on K1 saturated by sodium. Experimental data (1= dark yellow), calculated line (2 = red) and (A, B, C, D, E) the different domains.

Figure 3. Result of the decomposition by the DIS method of the derivative adsorption isotherm of argon on K2 exchanged by sodium. Experimental data 1; calculated line 2 and (A, B, C, D, E) the different domains.
manner by using five local isotherms, following the BET model.
For the two samples, the obtained derivative isotherms present the same shape (Figures 2 and 3), as it is generally the case for the phyllosilicates. Tables 1 and 2 give the parameters collected by modelling the adsorption isotherms of K1 and K2 respectively. By analogy with data in the literature the local isotherms of stronger energy (lnP/P0 = –7.3 to –12.3) observed can be assigned to the adsorption on the edge surfaces of the clayey particles [12,21,22]. In the same way, the isotherms of weaker energies (toward lnP/P0 = –3 to –4.7), are assigned to the adsorption on the basal surfaces.
3.2.2. Derivative Isotherms of Nitrogen Adsorption
The experimental isotherms of nitrogen adsorption on K1 and K2 were plotted in Figures 4 and 5 respectively. As previously, the derivative isotherms were modelled in a satisfactory manner by using four and five local isotherms following the BET model for K1 and for K2 respectively. Here, the obtained derivative isotherms present small different shapes for K1 and K2 (Figures 4 and 5).

Table 1. Deducted values of modelling the adsorption isotherm of argon at 77 K on K1-Na.

Table 2. Deducted values of modelling the adsorption isotherm of argon at 77 K on K2-Na.
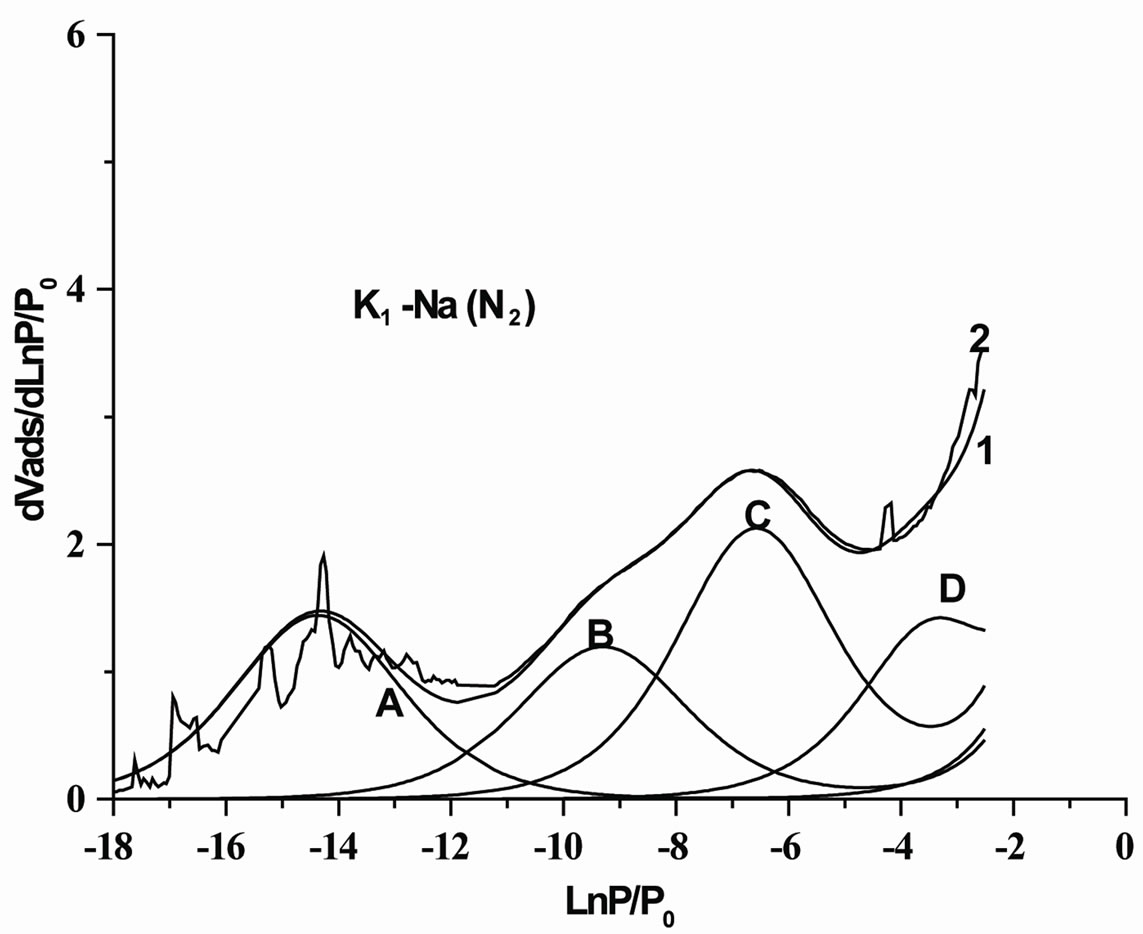
Figure 4. Result of the decomposition by the DIS method of the derivative adsorption isotherm of nitrogen on the K1 saturated by sodium. Experimental data 1; calculated line 2 and (A, B, C, D, E) the different domains.

Figure 5. Result of the decomposition by the DIS method of the derivative adsorption isotherm of nitrogen on the K2 exchanged by sodium. Experimental data 1; calculated line 2 and (A, B, C, D, E) the different domains.
Four and five domains have been classified through decreasing energy for K1 and K2 respectively. The parameters collected by nitrogen adsorption are listed in Tables 3 and 4.
Specifically for nitrogen, one can note that the position of the maximum of the peaks observed is baffled toward the strong energies, or even to the very strong energies (case of the A domain in Table 3).
This energy adsorption differences between argon and nitrogen can be attributable to the difference between the saturation pressures of these two gases, and probably to specific interactions of nitrogen with the solid surface [23].
Here again, in agreement with data found in the literature, the peaks with weak energy (C and D domains for K1 or D and E domains for K2) can be attributable to the adsorption on the basal faces. The other peaks of strong energies are assigned to the adsorption on the lateral surfaces even though; some cautions should be observed, because the affectation of the higher energy peak is not always obvious with nitrogen. It could correspond to particular sites of basal faces (shortcoming of surface), sites of lateral faces, or to micropores [21].
The sites of strong energy present on the surface of the solid studied, are observed with argon equally, but seem more accessible to nitrogen, in relation to the adsorbed volumes (Figures 3 and 4). The sites that have a structural origin are often made of groups SiOH and/or AlOH, which are sensitive to amphoteric dissociation [24]. They can influence the adsorption of micro-pollutants or can be the seat of catalytic reactions, in a non-negligible manner.
The total specific surface of all samples (DIS surfaces), argon and nitrogen, has been calculated while adding the specific surface relative to the set of the domains (Figures 1 to 4). The specific surfaces values deducted from K1 and K2 are recapitulated in Table 5. Those observed in nitrogen are more than the DIS argon surfaces. This difference is explained from the assumption that nitrogen is able to probe “inaccessible” sites to argon [25,26].

Table 3. Deducted values of modelling the adsorption isotherm of nitrogen at 77 K on K1-Na.
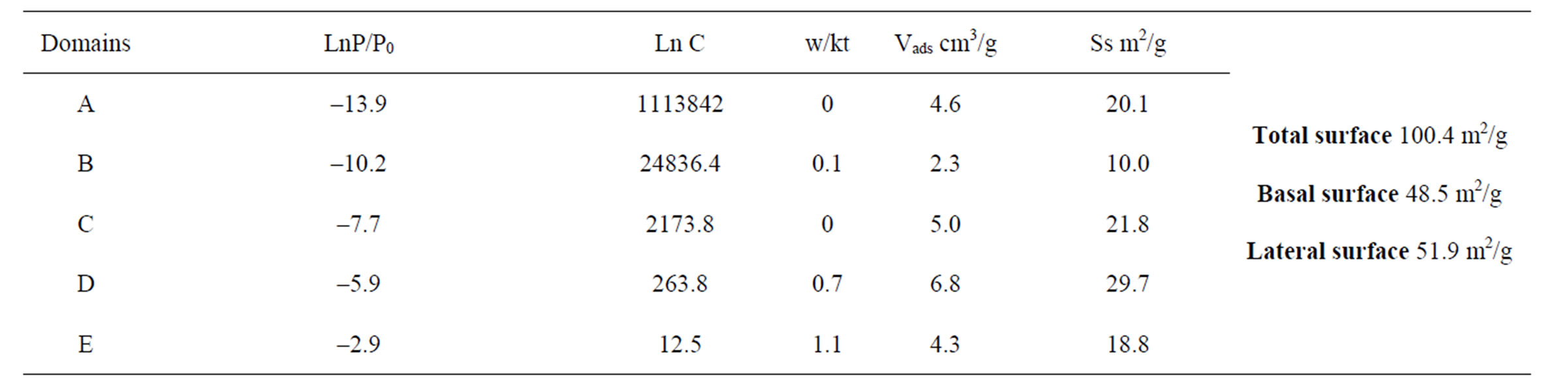
Table 4. Deducted values of modelling the adsorption isotherm of nitrogen at 77 K on K2-Na.

Table 5. Summary of specific surfaces values deducted for K1 and K2 (DIS surface: m2/g).
The size of the nitrogen molecule is appreciably identical to the one of argon. Then, the supplementary sites probed by nitrogen, can be assigned to specific interactions between nitrogen and the superficial polar sites.
This difference supposes the presence of probable polar sites on the surface of the solid, which constitutes important information for the possible uses of these materials. Indeed, according to Legens [27] and Chamerois et al. [28], knowledge of the polar or no polar character of the solid surface permits one to deduce the importance of the exchanges mineral-solution, by electrostatic origin and equally to deduce the retention of organic pollutants capable of being set by hydrogen links, notably non ionic tensioactives, often present in industrial sewages.
Lamellar indication of the samples (IL = basal surface/ total surface) has been calculated from the isotherms of argon adsorption. The determination of the middle shape of the grains is very important for the use of the solid mineral in many domains. Indeed, the mechanisms and the kinetics of sorption, dissolution/precipitation depend on the shape of the particles. In the present case, the values of IL calculated are of 62% for K1 and 72% for K2. These indications suppose for the studied samples, a relative sharpness of the solid grains.
4. Conclusions
At the end of this work, two main conclusions can be drawn.
First, the use of Derivative Isotherms Summation (DIS) method has permitted us to describe and quantify the two bentonite clays (referenced to as K1 and K2) from Katiola in Côte d’Ivoire, total specific surface are for K1: 91.9 m2/g to argon and 100 m2/g and for K2: 85.7 m2/g to argon and 100.4 m2/g to nitrogen.
Second, Beyond the quantitative aspect, the DIS method has permitted us to describe the two samples, a family of high energy site (–14 kT to nitrogen), that shows a strong affinity for the polar adsorbates. In the same way, Lamellar Indication of the samples (IL) calculated are of 62% for K1 and 72% for K2. These indications supposed for the studied samples, a relative sharpness of the solid grains, which shows important property of adsorption in various domains, notably in environment sciences.
5. Acknowledgements
This study was done in the Laboratory Environment and Mineralurgy (LEM) of Nancy. We are grateful to the staff members of the LEM for their invaluable contribution to this work.
We take this opportunity to thank the French department of foreign affairs for financial support to this work.
NOTES