Antioxidant and Cytotoxicity Potential of Six Synthesized Chalcones ()
1. Introduction
Chalcones are widely present in nature as important intermediates in the flavonoid biosynthetic pathway in various ferns and plant species [1] . The basic structure of chalcones contains a C6-C3-C6 backbone including two aromatic rings linked by an α, β-unsaturated carbonyl group [1] .
Numerous studies have described chalcones as compounds that have various pharmacological activities such as anticancer [2] [3] , anti-inflammatories [4] [5] , including antioxidants [6] [7] .
Prompted by all these considerations, in an effort to design a unique template endowed both with radical scavenger properties and cytotoxicity activities, we replaced a phenyl ring of chalcone with heterocyclic rings namely imidazopyridine and benzimidazole. The purpose of the herein study was to evaluate the antioxidant and cytotoxic activity of six synthesized chalcones in the perspective to propose them as forward models on which we can rely for the development of new molecules effective against cancers.
2. Materials and Methods
2.1. Chemicals
Sodium chloride (Pharmivoire, Ivory Coast); Dimethyl sulfoxide (DMSO, Sharlau, Germany); ABTS and Trolox (Sigma-Aldrich, France).
2.2. Samples under Study
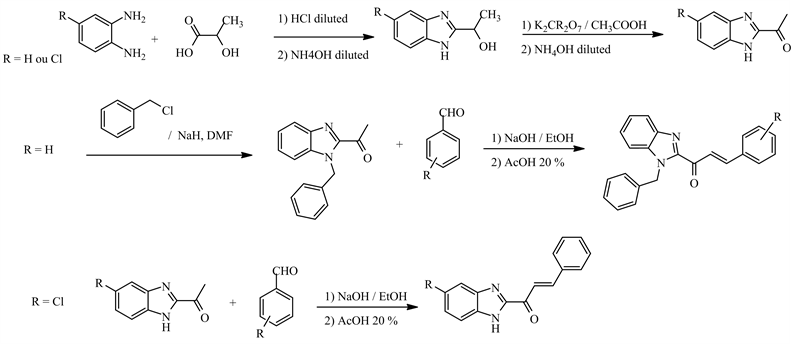
The six compounds tested, i.e. benzimidazolyl-chalcones (V1, V2, WAC1), and imidazopyridinyl-chalcones (V1, V2, V3), including molecular model (compound R), were synthesized and provided as pure powder by the Department of Organic and Therapeutic Chemistry of Training and Research Unit of Pharmaceutical and Biological Sciences (Côte d'Ivoire). From the molecular model (1,3-diphenylpropenone or compound R: Figure 1), chemical modifications were made on the aryl groups (ring A and B) to obtain the compounds under study.
The ring A replaced by benzimidazole or imidazopyridine rings, different substituted in ring B with electro donor element, allowed the synthesis of benzimidazolyl-chalcone derivatives (U1, U2, WAC1) and imidazopyridinyl-chalcones (V1, V2, V3). All of the chalcone hybrids were characterized by nuclear magnetic resonance (300 MHz for 1H and 75 MHz for 13C) on a Brucker Avance 300. Mass spectra were recorded on a JEOL JMS DX300 spectrometer on mode ESI, and details on their synthesis and structural information are located in the supporting information [8] [9] .
![]()
Figure 1. Molecular model (1,3-diphenylepropenone).
These products were in powder of different colors, insoluble in water and the usual organic solvents, except for dimethyl sulfoxide (DMSO).
2.3. Antioxidant Ability Test
We selected the decolourisation assay of ABTS•+ cationic radical for antioxidant potential evaluation, because it is suitable for the screening of hydrophilic as well as lipophilic molecules [10] [11] .
2.3.1. ABTS Test
The test was performed according to Choong et al. [12] . ABTS [2,2’-azino-bis (3-ethylbenzothiazolin-6-sulfonic acid)] solution produces the ABTS•+ radical cation with a green blue colour in solution, for which the absorbance is measured at 730 nm. Molecules that have an antioxidant ability, by donating an electron, reduce the ABTS•+ cation and turn the green blue reaction medium to a completely discoloured solution. The amount of antioxidant in the sample test is therefore inversely proportional to the formation of the ABTS.
2.3.2. Obtention of Radical Cation ABTS•+
The ABTS•+ radical cation is obtained by mixing volume-by-volume (1:1 v/v) of a stock solution ABTS 7.0 mM with 2.6 mM potassium persulfate solution, followed by incubation in the dark for 12 to 16 hours. The absorbance measured by spectrophotometry was adjusted between 1.0 and 1.5 at 734 nm, adding 60 mL of phosphate buffer solution (pH 7.4) to 1 mL of solution containing the radical ABTS•+.
2.3.3. Procedure of the ABTS Method
The tests were carried out according to the following steps:
- Performing a sample concentration range (1 - 0.5 - 0.25 - 0.125 - 0.625), by successive dilutions to half (1/2) in DMSO;
- Adding in a test tube, 100 μL of the sample +2500 μL of ABTS reagent for each concentration, then incubate in the dark for 30 minutes;
- Preparing the negative control (white): Mix of 100 μL of DMSO + 2500 μL of ABTS reagent then incubation in the dark for 30 minutes;
- Preparing the positive control (Reference): standard of an antioxidant (Trolox) which absorbance was measured under the same conditions as the samples: mix 100 μL of Trolox + 2500 μL of ABTS reagent, then incubate in darkness for 30 minutes;
- Reading the absorbance at 734 nm (UV-VIS spectrophotometer);
- Performing three (3) times reading for each compound.
The results, expressed as percentage of reducing the ABTS•+ radical cation, were calculated at the dose of 1 mg/mL for all the samples tested, including Trolox (standard substance), according to the following formula:
DO1: average Optical Density of neutral control;
DO2: average Optical Density of tested compound.
2.4. Cytotoxic Capacity
It was carried out at Chatenay Malabry biology laboratory (Paris-France). The evaluation focused on a healthy umbilical endothelial cell line (HUVEC) and two (2) malignant lines of human origin, namely a liver cancer line (HEP-G2), and a lung cancer line (A549).
2.4.1. Cell Culture and Cytotoxicity Tests
HUVEC, HEP-G2, A549 cells were cultured in specific media supplemented with 10% Foetal Calf Serum (FCS) and 1% Penicillin Streptomycin (PS). The autotoxicity test with MTT was carried out according to the method described by Price et al. [13] .
2.4.2. Obtention of Samples
- A fresh solution at 10 mM mixed with DMSO was prepared for each tested sample;
- Positive control: Doxorubicin (hydrochloride, 5 μM);
- Negative control: DMSO at 0.1%, and untreated control (endothelial cells alone, i.e. 100% viability).
2.4.3. Procedure of the Cytotoxicity Tests
The tests were carried out according to the following steps:
- Day 1: inoculating 3 wells with 5000 cells + 100 μL of each sample then incubating them for 24 hours;
- Day 2: treating cultures with additional 100 μL of the sample;
- Day 3: carrying out the MTT autotoxicity test after 48 hours of incubation and analysing in cytometry;
- Measuring the optical density (OD) 3 times for each sample and the controls.
The results, expressed as percentage of inhibition cells growing, were calculated at using the same formula of antioxidant percentage inhibition.
2.5. Statistical Method
Data were analyzed using Graph Pad Prism® 7.0 software. The Dunnett’s test compared mean values ± SD (standard deviation) by analysis of variance (ANOVA) at risk α = 0.05.
The codification for statistically significant difference was: *: 0.01 < p ≤ 0.05; **: 0.001 ≤ p ≤ 0.01; ***: p < 0.001.
3. Results
3.1. Antioxidant Potential (Figure 2)
![]()
Figure 2. Compounds antioxidant potency. Dunnett’s test at risk α = 0.05%. Values measured 3 times and expressed on average ± SD (standard deviation). ***: p ≤ 0.001: Significant difference compared to R: U1 (p = 0.0001), U2 (p = 0.0001), WAC1 (p = 0.0001), V1 (p = 0.0001), V2 (p = 0.0001), V3 (p = 0.0001).
3.2. Cytotoxicity Activity on Human Healthy Line (HUVEC) (Figure 3)
![]()
Figure 3. Cytotoxic effect of compounds against healthy human cells HUVEC.
3.3. Cytotoxicity Activity on Human Hepatic Malignant Line (HEP-G2) (Figure 4)
![]()
Figure 4. Cytotoxic effect of compounds against hepatic malignant cells HEP-G2.
3.4. Cytotoxicity Activity on Human Pulmonary Malignant Line (A549) (Figure 5)
![]()
Figure 5. Cytotoxic effect of compounds against pulmonary malignant cells A549.
4. Discussion
Antioxidant screening showed that in benzimidazolyl-chalcones series, the introduction of a phenyl group on pyrrolic-N (U1), or a hydroxyl group substitution in position 2 (U2) of N-benzylbenzimidazolyl-chalcones, allowed these compounds scavenging the ABTS•+ with a potent respectively 11 and 14 times greater than that of the molecular model.
Our results are compatible with those reported by Murti et al. [14] , who specified in their study that the antioxidant activity of benzimidazolyl-chalcones was around 39% to 70%. Also, our results suggest that the presence of electron-donating substituents type hydroxyl on benzylbenzimidazolyl-chalcone (U2), is favorable for the enhancement of antioxidant potency, as revealed by the work of Prakash et al. [15] ; Ghawalkar et al. [16] ; and Lembege et al. [17] .
In imidazopyridinyl-chalcones series, the introduction of hydroxyl group at position 2 or 3 (V1, V2); or dimethylamine [-N(CH3)2] at position 4 (V3), made possible to obtain compounds with high antioxidant potential respectively at 91.62%, 99.84% and 97.45%, similar to that of the positive control (Trolox: 99.12%) and 15 to 16 times greater than that of the molecular model.
The cytotoxicity study revealed that compound V2 inhibited HEP-2 malignant cells growth with a cytotoxic effect similar to Doxorubicin, whereas compound WAC1 was more cytotoxic against A549 malignant cells with a greater effect compared to Doxorubicin. In contrast, V1 and V3 molecules were not cytotoxic on healthy HUVEC cells, in comparison with DMSO control.
The mechanism action suggested for V2, whose cytotoxic effect was similar to that of doxorubicin on HEP-G2, is that it interacts as DNA intercalating agents leading to cell death [18] .
The same mechanism action could also be attributed to WAC1, but the fact that its effect was superior to that doxorubicin on A549 cells, would allow us, to expand its action as an agent that can interact on the signalling pathways of cell transduction. Indeed, WAC1, would behave as an agonist of tumor necrosis factor α (TNFα) which selectively binds to TNFR1 receptors, activates the transduction of signals mediated by the apoptotic pathways TRADD and FADD, resulting in the stimulation of caspases which cleaves multiple proteins responsible for cell death [19] [20] .
Our results are consistent with a recent work led by Kamal et al. [21] , who showed that derivatives of chalcones vectorized by various heterocyclic rings like imidazopyridine, imidazopyrimidine or benzimidazole, exhibited significant antiproliferative activity against the A549 human lung malignant line.
5. Conclusions
The imidazopyridinyl-chalcones and benzimidazolyl-chalcones could be good profiles of antioxidant activity. Depending on the position of the phenolic group in phenyl ring, the radical scavenging activity of the imidazopyridinyl-chalcones decreased in the following order: 3-OH > 4-N(CH3)2 > 2-OH. In the benzimidazolyl-chalcones series, the presence of a 2-OH group on the phenyl group was favourable to antioxidant activities when another phenyl group was inserted on pyrrolic-N. However, insertion of a 5-chloro group in benzimidazole didn’t enhance the activity of corresponding chalcone (WAC 1).
In addition, the best cytotoxic activity was provided by benzimidazolyl-chalcone bearing a halogen at the 5-position of the heterocycle (WAC1). The reported structure-activity observations can strongly contribute to designing and synthesizing chalcones endowed of ABTS radical scavenging or cytotoxic activities.
Acknowledgements
We express our sincere gratitude to Chatenay Malabry biology laboratory (Paris-France) for the study of in vitro cytotoxicity on HUVEC, HEP-2 and A549 human cell lines and to CEISAM Laboratory of the University of Nantes (for chemical reagents, MS and NMR spectroscopy).
Corresponding Author’s Institution
Laboratoire de Pharmacologie, UFR Sciences, Pharmaceutiques, Université FHB, 01 BP V34, Abidjan, Côte d’Ivoire.
Supporting Information
STRUCTURAL INFORMATION OF COMPOUNDS
SYNTHESIS INFORMATION OF THESE COMPOUNDS
Synthesis of Imidazopyridinyl-chalcone [8]
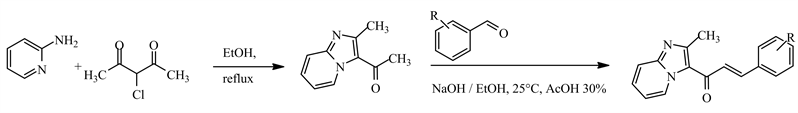
Synthesis of benzimidazolyl-chalcone [9]