Quantitative Stereological Estimations of Structural Patterns of the Glandular Tree in Benign Hyperplasia of Prostate ()
Received 22 April 2016; accepted 26 June 2016; published 29 June 2016

1. Introduction
Benign prostate hyperplasia (BPH) is the most common benign disease of human prostate [1] [2] . It produces urological symptoms in 54% of men between 60 and 70 years. The BPH detection by microscopic examination at autopsy is greater than the clinic detection of symptoms (about 70% of men in the same age range) [3] . It represents approximately 50% of the medical consultations made by urological disease [4] [5] .
BPH has two phases: pathologic and clinical. The first phase has in turn two stages: microscopic, with the presence of small foci of prostatic hyperplasia, and macroscopic, with evidence of adenomas. Approximately 80% of men over 40 years developed a microscopic BPH, but only half of these evolved to a macroscopic BPH in an average time of five years [6] [7] .
The prostate grows from birth to puberty to reach between 20 and 30 years of age reaching an average weight of 20 g. From here to the 90 years its growth rate decreases. From the fifth decade of life, with the development of histologically identifiable BPH, increased prostate weight occurs reaching an average weight of 33 g [8] .
Prostatic stromal cells play an important role in the pathogenesis of BPH. The first histological changes include microscopic nodules in the stroma surrounding the periurethral glands. The acinar hyperplasia begins around these small nodules and will increase over the years, to produce nodules up to several centimeters in diameter whose histology may show a predominance of epithelial elements, stromal or a mixed pattern. Depending on what elements predominate, BPH can be classified in five varieties: stromal, fibromuscular, muscular, fibroadenomatous and adenofibromiomatous (the most common) [8] . BPH affects mostly the transition zone and the periurethral area of the prostate [9] [10] .
Although the pathogenesis of BPH is unclear, it is known to be multifactorial, being necessary the presence of two factors for prostate growth occurs: the androgen stimulus and age [4] . Different theories have been proposed, based on histological, and hormonal age-related changes, but, currently, not a single explanation is accepted [11] . Androgens possibly act as initiators’ stromal hyperplasia, which in turn induce epithelial hyperplasia [3] [12] . It has been also suggested that neuroendocrine cells are involved in the genesis of BPH, perhaps enacting some mediation between stroma and epithelium.
Currently BPH is associated with unregulated proliferation of connective tissue, smooth muscle and glandular epithelium within the prostatic transition zone, and it has been described as relevant characteristic of BPH―the increase of the total number of cells, and not only an increase in cell size [13] . [14] has shown that early periurethral nodules have a stromal structure, while the early nodules in the transition zone represent the proliferation of the glandular tissue. Glandular nodules rise from the newly formed small ducts, arising as buds on existing ducts; these ducts grow and branch out, creating an entirely new ductal system within the nodule. Histologically, BPH is characterized by progressive hyperplasia of glandular and stromal tissues around the urethra with excessive nodular growth localized to the points where ejaculatory ducts enter into the transitional or periurethral zones of the prostate [15] .
The cytological findings in BPH are generally of little relevance, and rather unspecific, as alterations including basal cell hyperplasia, increased stromal mass (particularly the amount of smooth muscle cells), enhanced extracellular matrix deposition, reduced elastic tissue, more infiltrating lymphocytes around ducts, acinar hypertrophy and more luminal corpora amylacea and calcifications in the form of prostatic calculi [16] .
Although some increase of cell proliferation has been detected in the glandular epithelium of BPH in comparison with normal prostate [17] , the proliferating rate was significantly lower than in prostate cancer [18] . In both normal and BPH glands the proliferative compartment is located in the basal cell layer. Compared to normal and hyperplastic conditions, severe proliferative abnormalities were detected in high-grade prostate intraepithelial neoplasia, as documented by the extension of the proliferative compartment up to the luminal border [19] .
To date, there have been no studies on the quantitative morphology of glandular tree of BPH compared with normal prostate. The only mention of this particular has been noted by [20] , indicating that the histologic hallmark of BPH is the expansile nodule, produced by the budding and branching of newly formed duct-acinar structures. Suggesting that the glandular tree branches and expands as the hyperplastic transformation occurs in the prostate.
To verify if this gland expansion and branching was similar to that occurs in the growth of normal prostate, the present work deals with the estimation of several stereological parameters which can shed some light on the structure and distribution of prostatic glandular tree and its possible changes in BPH.
Briefly, the parameters investigated were:
1) Labeling index for the proliferating cell nuclear antigen (PCNA) to quantify the rate of proliferation of prostate epithelium.
2) Average thickness of glandular epithelium.
3) Fraction of the volume occupied by the epithelium relative to the total prostate volume.
4) Connectivity density of prostate glands, to quantify the branching of prostate glands.
5) The average volume and the volume-weighted mean glandular volume of prostate acini to assess the mean size of the prostate acini and its variability.
All these estimates have been performed in prostates from young men (controls) and in adenomectomy specimens from the adenofibromiomatous variety of BPH.
2. Methods
Twenty prostate specimens were collected at La Princesa Hospital (Madrid, Spain), 10 were from adults, (CTR group), age (mean ± SD): 45 ± 7; range: 30 - 47 years, all these specimens were of healthy subjects, without endocrine or reproductive pathology, deceased in traffic accidents, and eligible as donors for transplant, the age of the subjects of CTR group was in the range indicated to avoid any histological changes of subclinical BPH, relatively frequents in subjects older than 50 years. The other 10 were surgical specimens (adenomectomies) from patients diagnosed of the adenofibromiomatous type of benign prostatic hyperplasia, (BPH group), age (mean ± SD): 75 ± 10, range: 65 - 85 years. All the ethical requirements were accomplished in order to obtain the prostatic tissue either at the moment of the multiorganic extraction for transplant (CTR group) or at the surgery (BPH group). Immediately after extraction, the specimens were fixed during a week in 10% paraformaldehyde in PBS, pH 7.4 (Probus, Barcelona, Spain).
After fixation, the specimens from the two groups were thoroughly sectioned into 2-mm-thick slices, performed by isotropic uniform random sampling (IUR sections) in order to preserve the isotropy of the tissue [21] .
All the specimens were processed for paraffin embedding. The paraffin blocks were exhaustively sectioned. A total of 20 sections were performed on each block: Ten 5-mm-thick sections for immunohistochemistry and routine haematoxylin-eosine, and other ten 15-µm-thick sections for quantitative studies.
Two immunostainings were performed: i) for visualization of proliferating cell nuclear antigen (PCNA) in order to evidence the epithelial proliferating cells [22] , and ii) for detection of the prostatic specific antigen (PSA), to visualize the prostate acini [23] .
At least five randomly selected slides per specimen were immunostained for each antigen in CTR and BPH groups. Deparaffinized and rehydrated tissue sections were treated for 30 min with hydrogen peroxide 0.3% in phosphate-buffered saline (PBS) pH 7.4, to block endogenous peroxidase. To detect PCNA immunoreactivity, sections were incubated with a monoclonal anti-PCNA antibody (Biomeda, Foster City, Ca, USA) diluted at 1:400. To show PSA immunoreactivity, sections were incubated with a monoclonal anti-PSA antibody (Santa Cruz Biotechnology, Santa Cruz, Ca, USA) diluted at 1:50. Pretreatment of sections by heat in citrate buffer pH 6.0 (using a pressure cooker) [24] was performed to enhance immunostaining for both antibodies.
The primary antisera were diluted in PBS pH 7.4 containing 1% bovine serum albumin (BSA) (Sigma, St Louis, USA) plus 0.1% sodium azide (Merck, Darmstad, Germany). The incubation with primary antisera was overnight at 4˚C. The second antibodies employed were a biotin-caproyl-anti-rabbit immunoglobulin (Biomeda, Foster City, CA, USA). The second antibodies were diluted at 1/400 in PBS containing 1% BSA without sodium azide, and incubated for 30 min at room temperature. Thereafter, sections were incubated with a streptavidin-biotin-peroxidase complex (Biomeda). The immunostaining reaction product was developed using 0.1 g diaminobenzidine (DAB) (Sigma) in 200 mL of PBS, plus 40 mL hydrogen peroxide. After immunoreaction nuclear counter-staining with haematoxylin was performed in all the sections immunostained for PCNA, and in some sections immunostained for PSA. No nuclear counter-staining was performed on the 15-µm-thick sections PSA immunostained that were then employed for quantitative purposes. All slides were dehydrated in ethanol, and mounted in a synthetic resin (Depex, Serva, Heidelberg, Germany). The specificity of the immunohistochemical procedures was checked by incubation of sections with nonimmune serum instead of the primary antibody.
Quantitative Methods
i) Estimation of the proliferative index of epithelium
In the two groups, the numerical density (NV) of epithelial cells (cell number per unit of volume of reference space) was estimated for both PCNA immunoreactive (NV PCNA positive) and PCNA negative nuclei (NV PCNA negative), employing the optical disector, an unbiased stereological method [25] , using the Cast-Grid programme (Stereology Software Package, Silkeborg, Denmark). The space of reference employed for estimating the numeric densities was the occupied by the epithelium, then, only was registered the volume associated to the frames with its upper-right corner hitting epithelial tissue. The total NV of epithelial cells was calculated as NV total = NV (PCNA negative + PCNA positive), then, the labeling index to PCNA (LIpcna) for epithelial cells was expressed as the ratio NV PCNA positive/NV total.
ii) Quantitative morphology of prostate acini
Three parameters have been employed to quantitate the acinar pattern: The volume fraction of epithelium (VV ep), the average thickness of epithelial lining (Th ep), and the connectivity density of the acinar tree (ConV gl).
The sampling protocol for the estimate of these parameters is described in detail: For all the specimens in each group of study, three sections immunostained to PSA were randomly sampled. Every section was scanned at a final magnification of ×100, to select an average of 5 microscopical fields per section that were chosen by systematic random sampling [26] . For each selected field, five images evenly distributed along the Z axis of the section were captured, being the distance between each couple of images of 2 µm. The capturing of images was made using a colour digital camera DP 70 (Olympus Corporation of the Americas, PA, USA) with a resolution of 12.5 mega-pixels, attached to an Olympus microscope fitted with a motorised stage controlled by the stereological software Cast-Grid (Stereology Software Package, Silkeborg, Denmark). This program controls the XY displacement of the microscope stage and allows the selection of fields to be studied by random systematic sampling after the input of an appropriate sampling fraction [27] . The result was a series of images from the two groups, sized 512 × 680 pixels. The final magnification (×100) was such that 512 pixels represented 653 μm.
Subsequently, the images were processed using the public domain Java image processing program, Image J (version 1.48), developed at the US National Institutes of Health and available on the Internet at http://imagej.nih.gov/ij/index.html [28] . For each set of images obtained in each sampled field, a stack of a thickness of 10 µm was constructed. Afterwards, for estimation of VV ep, and Th ep, every stack was binarised, and the PSA immunostained cytoplasm was shown as black and the reference space (glandular lumina, stroma, etc.) as white (Figures 1(a)-(d)). Then, for estimation of ConV gl, the binarised stack was subsequently processed by several stages of pixel dilation and hole filling to transform the binarised acini (epithelia plus lumina) in compact contours where the acini were shown as black and the stroma as white (Figure 1(e), Figure 1(f)).
The quantitative parameters studied were estimated onto these stacks of binarised images, employing a plugin that runs in the Image J software [29] , briefly:
a) Epithelial volume fraction (VV ep) is the volume of PSA immunoreactive epithelium per unit of volume of the reference space (acini plus stroma), VV ep is simply the number of foreground (epithelium) voxels divided by the total number of voxels in the image.
b) Average thickness of epithelial lining (Th ep). The plugin employed defines the thickness at a point as the diameter of the greatest sphere that fits within the structure (epithelial lining) and which contains the point [30] . The plugin calculates the average of the epithelial thickness (Th ep) directly from pixel values in the resulting thickness map. It assumes that PSA immunostained epithelium is the foreground of the binarised image, the results are shown in µm.
![]()
Figure 1. In (a) and (b) the images shown are from a specimen of CTR group and a case of BPH group respectively, immunostained to PSA. The same binarised images are depicted in (c) and (d), the PSA immunoreactive cytoplasm is in black, and the space of reference (acinar lumina and stroma) in white. These binarised pictures, processed to obtain the contours of acinar profiles (epithelium plus lumina, in black) are shown in (e) and (f). The scale bars represent 190 µm.
c) Connectivity density of the acinar tree (ConV gland). The number of connected structures in a network can be determined by calculating the Euler characteristic [31] . As other structures, like lung alveoli or cancellous bone [32] [33] , prostate glandular tree is one such network, and its connectivity density (ConV gl), i.e. number of acini connected per unit of volume of reference space, can by calculated by dividing the connectivity estimate by the volume of the stack sampled. The algorithm in the plugin employed uses voxel neighborhoods to calculate the Euler characteristic of the volume sampled in the binarised stack, and adjusts this to give the contribution of this volume to the connectivity of the structure it was chosen from. The amount of connected structures (acini) gives an idea of the complexity of glandular branching.
iii) Estimations of size of the prostate acini
Two methods have been performed in order to measure the mean volume of the acini, first was estimated the average volume of glands that were sampled independently of their size [34] , i.e. the not weighted or numeric distribution of size (VN gl). Then was obtained a volume-weighted mean glandular volume [35] , that provides a weighted distribution of size (VV gl) that gives information about the variability of size population [34] .
The procedures were as follows:
a) The nucleator, an unbiased stereological estimator [36] , was applied to estimate the mean glandular volume (VN gl) for the prostate acini stained with HE from both groups of study. Briefly: The acini eligible for measuring were sampled after Sterio rule [37] using optical disectors at a final magnification of ×40. Their center of mass were determined and employed as the reference point. Through this point, the software generates two isotropically oriented segments hitting four points from the boundary of the gland. The program then employs the following formula to estimate the volume: , where VN is the neuronal volume, and
, where VN is the neuronal volume, and 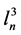 is the mean cubed length of the segments intercepting the boundary of the acini.
is the mean cubed length of the segments intercepting the boundary of the acini.
b) The average volume-weighted mean glandular volume (VV gl) was performed using the point sampled intercept method [34] , that estimates the volume, giving greater chance of volume estimation to structures of greater size.
The program used to evaluate VV gl enables the generation of random test-lines directions that were superimposed onto the microscope images. The gland intercepts can be measured along these test lines. The length of gland intercepts (l0) was processed to obtain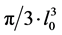 , an unbiased estimate of VV gl independent of acinar shape, which, because of point sampling, emphasizes larger acini rather than smaller ones. In addition, estimates of VV gl combine information about the three-dimensional glandular size with knowledge of variability of glandular size [38] .
, an unbiased estimate of VV gl independent of acinar shape, which, because of point sampling, emphasizes larger acini rather than smaller ones. In addition, estimates of VV gl combine information about the three-dimensional glandular size with knowledge of variability of glandular size [38] .
All the measurements from VN gl and VV gl were carried out in each group of study onto three sections per specimen stained with haematoxylin-eosine and randomly sampled. The fields selected were visualized using an Olympus microscope at a final magnification of ×40. The software employed by measurements was the Cast-Grid.
c) Estimation of the second moment of the VN gl distribution: As estimates of VN gl and VV gl were made on the same population of glands, the coefficient of variation of the acinar volumes in the VN gl distribution [39] is given by
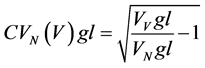
where CVN (V) gl is the coefficient of variation of the acinar volume in the VN gl distribution that is a measure of the variability in size of acini.
iv) Statistical analysis
The parameters measured were expressed as mean ± CI (confidence intervals at 95%). Comparisons between the means from CTR and BPH groups were performed by a Student t test (p < 0.05).
3. Results
3.1. Immunohistochemistry
Immunoreactivity for PCNA has been detected in some nuclei from basal cells of acinar epithelium in both control and BPH specimens. Not relevant differences were observed in intensity and distribution of PCNA immunoreactive nuclei in both groups of study (Figure 2). The cytoplasm of epithelial cells from normal (CTR group) and hyperplastic (BPH group) acini showed intense immunoreaction to PSA (Figure 3). No immunostaining to PSA was detected in other locations of normal and BPH prostates.
3.2. Quantitative Results
The LI pcna did not experiment significant changes when comparing CTR and BPH groups (Figure 4(a)). However, the epithelial thickness (Th ep) and the volume fraction of prostate occupied by epithelium (VV ep) were significantly increased in BPH group in comparison with CTR group (Figure 4(b), Figure 4(c)). Similarly, connectivity density (ConV gl) increases significantly in CTR group compared to the BPH group (Figure 4(d)).
The average volume of the prostate acini (VN gl) does not change significantly in BPH in comparison with CTR groups (Figure 5(a)), whereas the volume-weighted mean glandular volume (VV gl) and the coefficient of variation of the acinar volume -CVN (V) gl- were significantly increased in BPH specimens (Figure 5(b), Figure 5(c)).
4. Discussion
In the present study, it has been noted that in BPH there is no significant increase of cell proliferation measured
![]()
Figure 2. Sections immunostained to PCNA, from a specimen of CTR group (a) and from a case of BPH group (b). Some nuclei of epithelial basal cells are immunoreactive (arrowheads). No immunostaining for PCNA is viewed in the columnar cells of epithelium in both images. The scale bars represent 50 µm.
by PCNA labeling index. This seems in contrast to that described by other authors [18] [19] suggesting an increase in cell proliferation rate in BPH compared to normal prostate. This discrepancy may be due to that the BPH specimens included in the present study are in an advanced stage of hyperplastic growth, where more than a high cell proliferation, increased apoptosis occurs, keeping the size of the epithelial population, since it is known that prostatic hyperplasia shows minor degree of apoptosis that normal prostate, being an initial growth imbalance in favor of cell proliferation that might promote prostatic hyperplasia [17] .
The end point of this shifting towards the cell proliferation is the increase of epithelium that was reflected in the enlargement of the volume fraction of prostate gland occupied by the epithelium as was described in this study. However, other authors [40] , detect an increase of volume fraction of stroma instead of acini, this discrepancy might be due to the type of BPH studied. The augmentation of the epithelial compartment would be preferently found in the adenofibromiomatous type of BPH [8] that was the variety of BPH studied in this work. Also can be detected an increase of stroma-epithelium ratio in symptomatic BPH in comparison with asymptomatic BPH [41] .
The increase of thickness of epithelial lining might be in relation to some hypertrophy at the cellular level that is also present [42] .
![]()
Figure 3. Sections immunostained to PSA: in (a) image from a CTR case, in (b) an image from a BPH case. The cell nuclei were counterstained with haematoxylin. The scale bars represent 100 µm.
![]()
Figure 4. Mean ± CI (confidence intervals at 95%) for: (a) Epithelial labeling index for PCNA (LI pcna), (b) Average thickness of epithelium (Th ep), (c) Volume fraction of PSA immu- noreactive epithelium (VV ep), and (d) Density of connectivity of prostate glands (ConV gl), in control (CTR) and hyperplastic (BPH) groups. The means with significant differences between them are connected by zig-zag lines, indicating the correspondent p values.
![]()
Figure 5. Scatter plots expressing individual values (void circles) plus mean ± CI (confidence intervals at 95%) for: (a) numeric distribution of gland size (VN gl), (b) average volume-weighted mean glandular volume (VV gl), and (c) coefficient of variation of the acinar volume in the VN gl distribution [CVN (V) gl]. The means with significant differences between them are connected by zig-zag lines, indicating the correspondent p values.
The connectivity density from acini in BPH was significantly higher than in control prostate.
This parameter is related with the number of patterns interconnected in a branching structure [31] as occurs for the prostate glands. The increase of ConV gl suggest that in BPH, the glandular tree branches and expands as the hyperplastic transformation occurs in the prostate [20] . It is interesting to note, that the average acinar size estimated by VN does not change in BPH specimens comparing with control cases. Therefore, the increment of epithelial volume fraction correlates more with the increase of the number of acinar buds that with an increasing of mean volume of such elements.
The average volume-weighted mean volume is a simple and efficient method of estimating the size of particles, and has been revealed to be an important prognostic factor in a number of histopathological studies of melanoma [43] , prostate cancer [35] [44] , ovarian cancer [45] and others. All these studies deals with the size of cell nuclei, but to our knowledge the volume-weighted volume has not been employed to evaluate the variability on size of prostate acini.
The VV gl of the acini was significantly increased in BPH cases in comparison with CTR group. This finding is interesting because estimates of VV gl combine information about the three-dimensional glandular size with knowledge of variability of glandular size [38] . In fact, the coefficient of variation of acinar volumes in the VN gl distribution was also significantly greater in BPH than in CTR. Therefore, the size of the acini in BPH is more heterogeneous than the acini controls. That may mean that the growth of acinar buds in hyperplastic prostate is irregular and not fully comparable to what happens in the not hyperplastic gland.
5. Conclusion
This study sheds some new light on the pathogenesis of BPH, from the point of view of the growing pattern of the hyperplastic glandular tree. We can conclude that the epithelial cell proliferation is not the only factor intervening in the development of BPH, at least, in the stage, or for the type of hyperplasia studied in this work. In addition, a more prolonged survival of epithelial population, together with some degree of hypertrophy of acini expressed by the increase of volume fraction and thickness of acinar epithelium, is relevant in order to the growth and expansion of the BPH glandular tree that shows more abundant and heterogeneous acinar sprouts than in normal prostate.
NOTES
![]()
*Corresponding author.