Synthesis of Faujasite from Egyptian Clays: Characterizations and Removal of Heavy Metals ()
1. Introduction
Zeolites are crystalline microporous aluminosilicate consisting of tetrahedral units producing open framework structure, which generates a system of pores and cavities having molecular dimensions [1] . And due to their thigh, exchange capacity and sieve properties have been used by many workers in water purifications from heavy metals, ammonia and water softening. From the early 1940’s on wards, systematic synthesis studies on zeolites were started [2] . Due to the unique properties of zeolites, extensive attempts for zeolite synthesis were begun [3] . Nowadays, the increasing interest in zeolite synthesis from low-cost materials has promoted the development of various studies on their conversion into zeolitic materials, giving rise to an extensive literature [4] . At present, there are 191 unique zeolite frameworks identified [5] , and over 40 naturally occurring zeolite frameworks are known. Many authors used natural clays as start materials for zeolite synthesis. Clay minerals such as kaolinite, illite, montmorillionite, and interstratified illite-smectite have been used as the sources of Al and Si for the synthesis of several types of zeolites [1] [6] -[15] . Synthetic zeolites with faujasite structure have been prepared by designated zeolite X [16] , and by zeolite Y [17] . The differences existing between these substances are compositional and depending on the silica to alumina ratio of their framework. In X zeolites, the ratio is between 2 - 3, while in Y zeolites it is 3 or higher [18] . Zeolites have been studied in terms of the relations among structure, properties and synthesis. The zeolites of class faujasite (NaX, NaY, LTA) are used mainly as ion exchangers, adsorbents or catalysts in chemical, oil refining, gas industries and water softening and purification (Georgiev et al., 2013). Kaolinite is the basic raw material used to obtain zeolite with faujasitic structure and with low content of SiO2 such as NaX, NaY and LTA [6] [19] . The objective of this work is to prove which type of natural clay of the studied samples is better in zeolite synthesis using the method of [6] , as well as evaluation of the products as waste water purification for heavy metals.
2. Materials and Methods
Natural clays were used by many workers as starting materials in zeolite synthesis. In the present study to achieve this purpose, samples of clays with different composition were collected from different locations of Egypt (Figure 1). The samples are Kaolinite from Malha Formtion at El Dehisa mine southwestern Sinai. The Malha Formationm is built up of four distinct white kaolinite horizons interbedded with sandstones (Figure 1). Smectite-rich clay of Abu Tartur, which belong to black shale interbedded with glauconites and phosphorites (Duwi Formation). The third clay sample used here was picked out from the brick-red mudstones and grey siltstones with kaolinite-smectite composition (Madmoud Formation, Said 1981) exposed at Wadi El-Assuitti and Gahdam areas―Asyut, Egypt.
2.1. Zeolite Synthesis
Rios et al. (2009) used two different processes for zeolite synthesis from natural clays: 1) conventional hydrothermal alkaline activation and 2) alkaline fusion prior to hydrothermal reaction. The clay samples were prepared zeolite synthesis were carried out based on the second method of Rios et al. (2009) through the following steps:
![]() (a) (b)
(a) (b)
Figure 1. Location map for the raw material clay samples. A. Koalinite; B. Smectite-rich; C. Kolinite-Smectite clays.
1) Calcined raw materials were mixed with NaOH pellets for 30 min.
2) The resultant mixture was fused at 600˚C for 1 hour.
3) The alkaline fused products were ground in a mortar and then dissolved in distilled water with stirring for 30 min.
4) The reaction gel was aged 24 h to form amorphous precursors.
5) Crystallization was carried out by hydrothermal reactions under static conditions in PTFE vessels of 100 ml at 100˚C for different reaction times (48, 72 and 96 h).
6) The solid is separated by filtration, washed thoroughly several times with distilled water until the filtrate pH was reduced to less than 11.
7) The precipitated solid was dried at 100˚C overnight.
8) The dried samples were weighted and kept in plastic bags for characterization.
2.2. Characterization
X-ray diffraction patterns of raw clay samples and synthesis products were recorded using Ni-filtered Cu-Ko3 radiation X-ray diffractometer between 5˚ and 50˚ 2q with speed rate 06˚/min at Assiut University (Figure 2). Moreover, the clay fraction (<2 mm) of the brick-red mudstones of the Madmoud Formation was separated and treated with ethylene glycol for two hours and heated to 550˚C, then investigated by X-ray diffraction at 2q between 4 and 40. The structure of the synthetic products was studied by methods of XRD, SEM, FTIR-spec- troscopy, and thermal gravimetric analysis (DTA/TGA).
2.2.1. X-Ray Diffraction
As shown in the XRD patterns (Figure 2), kaoline is the predominant mineral phase in the raw material clay sample from Malha Formation (Southwestern Sinai), which can be identified by its distinctive reflections at12.25˚ and 24.84˚ (Figure 2(a)) near to that reported by Zhao et al. (2004). Smectite is the predominant mineral phase in the raw material clay sample from Abu Tartur Formation, and is identified by a series a basal at 15.77 Å and 5.6˚ 2q (Figure 2(b)). The clay sample from the Nile Valley (Madmoud formation?Assiut) composed mainly of kaolinite-smectite and is identified by its distinctive reflections at 4˚, 6.29˚, 11.93˚ and 24.58˚ 2q (Figure 2(c)). In all raw clay samples, quartz was identified at 4.26 Å and 3.35 Å. The later peak of quartz was more intense than the other peaks as shown in Figure 2. An almost complete transformation of the starting clay rich raw materials into faujasite-type zeolite of high purity occurred. However, relictic quartz of the starting materials still remains in the synthetized products. Complete disappearance of the characteristic peaks of clay minerals, accompanied by gradual decrease in peak intensity of quartz, and the appearance of zeolites was recorded after 48 h and showing an increase in the intensity of characteristic peaks between 48 and 96 h (Figure 2). The broad peaks at around 6˚, 10˚, 16˚, 23.5˚, 27˚, 31˚, 32˚, and 37˚ 2q are consistent with NaX-type.
2.2.2. Scanning Electron Microscope (SEM)
To characterize the morphology of the synthetic zeolite SEM images were investigated. Figure 3 demonstrated the occurrence of well crystallized cubic and octahedral morphology of the synthetic materials. Such morphology is characteristic to faujasite-type zeolite [6] [20] -[23] .
2.2.3. Fourier Transform Infrared (FT-IR) Spectroscopy
The characterization of zeolites with transmission Fourier transform infrared spectroscopy (FT-IR) is described (Figure 4). FT-IR spectroscopy is used to probe the structure of zeolites and monitor reactions in zeolite pores. Specifically, structural information can be obtained from the vibrational frequencies of the zeolite lattice observed in the range between 200 and 1500 cm−1 [24] [25] . In general, each zeolite has characteristic infrared pattern however, some common features are observed which, include the asymmetric (950 - 1250 cm−1) and symmetric stretch (660 - 770 cm−1), double ring vibration (500 - 650 cm−1), T-O bending modes (420 - 500 cm−1), and possibly opening modes (400 - 420 cm−1).
The IR-spectral data of the synthesized zeolites and NaX, Y, LTA zeolites are presented in Table 1. The double ring opening vibration 564 cm−1 in the FT-IR spectrum of the synthesized zeolites is near to 566 cm−1, which is characteristics to faujasite [22] . The bands at 444, 463, 467, and 672, 669 are assigned to the internal linkage vibrations of TO4 (T=Si or Al) tetrahedral and to symmetrical stretching respectively. These absorption bands characterizing T-O bending vibrations may shifted to lower frequencies due to decreasing Si/Al ratio in
![]() (a)
(a)![]()
![]() (b) (c)
(b) (c)
Figure 2. XRD patterns of raw materials and synthetic zeolite. (a) From kaolinite; (b) From smectite-rich clay; (c) From kaolinite-smectite clay.
![]()
Figure 3. SEM micrographs of the prepared zeolite: A and B from kaolinite raw material; C and D from smectite-rich clay; E and F from kaolinite-smec- tite clay.
![]()
Table 1. Important IR-bands for Y, LTA, X type zeolites, and the synthesized zeolites from the present study.
the internal linkage due to the different length of the Al-O (1.73 A) and Si-O (1.62 A) bands [25] [26] . The bands at 564, 672, 670 and 653,973,1000 are close to the bands at 560, 668 and 971 cm, which correspond to the 6-membered double-ring vibration , to the symmetric and the asymmetric stretching respectively, zeolite X (Table 1). The bands at 1647 and 1648 cm (Lwis sites) region is assigned to the zeolitic water in the channels of zeolite [27] . The bands at spectra 3446, 3460 and 3482 are attributed to the asymmetric stretching mode of molecular water coordinated to the edges of the zeolite channels [28] [29] .
2.2.4. Thermogravimetric Analysis (DTA/TGA)
Thermogravimetric analyses results of the synthesized zeolites are given in Figure 5. It can be seen from the thermal curves that the materials lost more than 20% of its mass under heating to 300˚C, which is considered enough to deduce that it can absorb significant amounts of water [19] . At temperature between 300˚C and 1000˚C, mass losses ranged from 1% to 3%, which means that the zeolite obtained was structurally stable. Such
![]()
Figure 4. FTIR-Spectra for the prepared zeolites: (a) from kaolinite; (b) from smectite-rich clay; and (c) from kaolinite- smectite clay.
![]() (c)
(c)
Figure 5. DTA/TGA curves of the synthetic zeolites: (a) from kaolinite; (b) from smectite-rich clay and (c) from kaolinite- smectite clay.
property is important for the synthesized zeolite which is used as a sorbent and for catalysis. The synthesized zeolites were found to be structurally stable up to 1000˚C.
3. Heavy Metals Removal
The increasing demands for a healthy environment, particularly with regards to high quality drinking water and removal of pollutants from industrial, agricultural and municipal wastewater, are major reason to search for new materials [4] . Zeolites have been widely used in many applications due their special properties. Adsorption is one of the special properties of zeolites. According to [30] , the amount of metal ion to be adsorbed is strongly affected by nature and concentration of metal ion, pH, and metal solubility, presence of competing and complexing ions. Many authors used the adsorption characteristic of Faujastic zeolites (X, Y and LTA) for heavy metal removal such as Cu+2, Pb+2, Cd+2, and 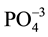 [31] [32] and Cr+3, Ni+2 [6] [33] . In the present study, synthetic solution with 100 ppm concentrations of Cr, Co, Ni and Mn was prepared by dilution of Merck chemical standard solutions (1000 ppm) for the mentioned elements.
[31] [32] and Cr+3, Ni+2 [6] [33] . In the present study, synthetic solution with 100 ppm concentrations of Cr, Co, Ni and Mn was prepared by dilution of Merck chemical standard solutions (1000 ppm) for the mentioned elements.
The treatment process was begun by adding 0.5 gm from synthetic faujasite to 100 ml solution, and left for 1 hour on magnetic mixer using magnetic mixer at room temperature. The absorption of the metal ions Cr3+, Co2+, Ni2+ and Mn2+ were measured before and after treatment using atomic absorption spectrometry. The metal uptake present as a function of metal ions were Cr3+ 100%, Co2+ 90%, Ni2+ 80 and Mn2+ 75% (Cr3+ > Co2+ > Ni2+ > Mn2+).
4. Conclusions
Zeolite was synthesized from Egyptian clays with different mineral compositions as raw material. Using hydrothermal reactions after fusion with NaOH method, the optimum conditions for hydrothermal crystallization of zeolite were found from 72 h to 96 h, at 100˚C, for kaolin and smectite-rich samples, and 48 h for smectite-kao- line sample.
The structure of synthetic product was studied by methods of X-ray diffraction (XRD), scanning electron microscope (SEM), Fourier transform infrared spectroscopy (FT-IR) and thermogravimetric (DTA/TGA) analysis. The synthetic product was proved to have the same structure as zeolite NaX-faujasite. Thermal gravimetric analysis indicates that the synthetic material lost more than 20% of its mass under heating to 300˚C, which is considered enough to deduce that it can absorb significant amounts of water.
The experimental results regarding adsorption characteristic of synthetic faujasite for metal ions Cr3+, Co2+, Ni2+ and Mn2+ removal from aqueous solution showed that the synthetic faujasite exhibits a great potential for adsorption of such metal ions from water.