1. Introduction
The kingdom of Bahrain is an aggregate of a group of scattered islands forming an archipelago lying almost in the middle of the Arabian Gulf. The desert environment predominates with high average temperature of 35˚C (ranging from 14˚C to 41˚C), and scanty average rainfall of 74 mm/year (varying from 39 - 128 mm/yr). The climate is semiarid to arid, with several hundred plant species [1] . The plants have long history of evolution and adaptation in dry and hot deserts. They are important sources of good gene pool in food chain, extreme environmental adaptability and herbal medicine. In Bahrain, 81 indigenous plant species are reported to be used in traditional herbal medicine [2] . The importance of medicinal plants both in drug research and biodiversity conservation is well recognized. Medicinal plants are the good sources of secondary metabolites which are used in pharmaceuticals and cosmetic industry [3] [4] . Biodiversity in many areas of the world is under threat and Bahrain is not an exception. Environmental factors are responsible for genetic diversity in a natural habitat. Therefore, the impact of various physiographic factors coupled with diverse climatic variations may generate the diversity in vegetation of particular region. Awareness and great concern about global biodiversity loss have developed in the last two to three decades. Global concern about the loss of valuable genetic resources has stimulated many new programs for the conservation and management of biodiversity [5] . In situ and ex situ conservation has applied according to the need of preservation process [6] . Al-Areen Wildlife Reserve in Bahrain Island has established in 1976 to promote scientific research, ecotourism, public awareness and conservation of biodiversity [7] .
The application of tissue culture as a conservation tool of the threatened plants has gained huge thrust in the last two decades as one of the processes of ex situ conservation strategies [8] [9] . Heliotropium kotschyi is locally known as “Ramram” in Bahrain, an important medicinal plant of Boraginaceae family. It is perennial bush but woody at base and can grow up to 20 - 50 cm in height, develop small white colored flowers (3 - 4 mm) during November to January. It is used as anti snake venom either by drinking the water extract of leaves or by putting a leaf paste on the bite [10] [11] . It is also reported as a endangered plant in Bahrain [8] . No reports of in vitro plant regeneration from tissues of medicinal plants are available in Bahrain. Therefore, the present study was undertaken to determine the effect of different growth regulators on shoot formation and multiplication of multiple shoots from axillary bud and shoot tips of Heliotropium kotschyi. Considering the great value of this multipurpose desert plant, development of micropropagation protocol is an important alternative for ex situ conservation strategy of this endangered plant. Moreover, the present study showed efficient in vitro plant regeneration protocol via adventitious shoot initiation and development from nodal segment. Further, the use of plant tissue culture technique for the purpose of conserving biodiversity and commercial propagation of medicinally important plants which are vulnerable to climatic changes can be considered as an initial work in Bahrain, more specifically in the GCC countries.
2. Materials & Methods
2.1. Explant Source
Stem segments of Heliotropium kotschyi were collected from field grown plants (Figure 1(A)) of Al-Areen Wild Life Park of Bahrain (Figure 2) [12] . Collected stems segments with leaves were cut into 3 - 5 cm (Figure 1(B)) lengths for using surface sterilization and for further use in establishing culture. Nodal stem segments were used as explants source for culture initiation.
2.2. Surface Sterilization of Explants
Stem segments (Figure 1(B)) were surface sterilized following modified method [13] , were washed in running tap water for fifteen minutes to remove dust particles from the surface of stem and leaf. Then stem segments were kept for five minutes successively in 1% of Lux solution, 0.5% of Mercric chloride solution with few drops of Tween 20, 0.1% of Copral solution, while the respective solutions were discarded in each step and were washed with autoclaved distilled water thoroughly (5 - 6 times) in each step to remove the used solutions. Later on, stem segments were kept in 50% (v/v) CLOROX solution (containing 2.625% hypochlorite) for 15 min, then the solution was discarded and explants were washed 5 - 6 times with autoclaved Millipore water to remove hypochlorite nicely. Final washing with 70% ethanol for 30 seconds was performed before transfer in autoclaved Millipore water until final cut of the explants. Finally, the leaves were removed and the stem segments were

Figure 1. Stages of micropropagation of Heliotropium kotschyi. A. Field grown plant; B. Stem segement used in surface sterilization; C. Explants (apical and nodal segments) in culture media transferred for culture initiation; D, Explants in media 1 after 4 weeks. E. Explants showed shoot initiation response in Media 13 after 4 weeks; F. Explants showed shoot initiation response in Media 4 after 2 weeks; G. Explants showed shoot initiation response in media 4 after 4 weeks ; H. Initially developed shoots transferred to multiplication media; I, J, K. Multiplication of shoots in first and second transferd media for 4 weeks. L, M. Micro-shoots developed roots; N. Rooted plantlet taken out from media; L. Plantlet growing in soil pot.
sliced with a sterile scalpel blade into smaller segments (1 - 1.5 cm), each with one node and were used as explants for culture initiation.
2.3. Media and Culture Conditions
The media used in the experiments were modified Murashige and Skoog media [14] containing 0.3% of casein hydrolysate, 3% of sucrose, 1 mg/L Nicotinic acid, 1 mg/L Pyridoxine HCl, 10 mg/L Thiamine HCl. The pH of the medium was adjusted to 5.8 by using 1N NaOH/1N HCl as per requirement. The media were solidified with 0.9% - 0.8% agar and autoclaved at 121˚C, for 20 minutes at 15 psi. Filter (0.22 µm) sterilized plant growth regulators (PGRs) of different combinations and concentrations were added in autoclaved modified MS media and the media were pored in Magenta bottle of equal volume. All cultures were incubated in the culture room maintained at 16/8 hours (light /dark) cycles with a cool white fluorescent light intensity of 2000 - 2500 LUX, temperature of 24˚C ± 2˚C with 70% - 80% humidity.
2.4. Treatments for Shoot Induction, Shoot Multiplication and Rooting
Nodal and apical segments were used as explants (Figure 1(C)) for shoot induction and were cultured in vitro in different media composition: (1) MS; and others were MS media supplemented with (2) 4.44 µM BAP +

Figure 2. Al-Areen wildlife park (Earth, Google, 2013).
2.85 µM IAA; (3) 6.66 µM BAP + 2.85 µM IAA; (4) 8.88 µM BAP + 5.71 µM IAA; (5) 13.32 µM BAP + 5.71 µM IAA; (6) 4.44 µM BAP + 2.68 µM NAA; (7) 6.66 µM BAP + 2.68 µM NAA; (8) 8.88 µM BAP + 5.37 µM NAA; (9) 13.3 µM BAP + 5.37 µM NAA; (10) 4.65 µM KI + 2.85 µM IAA; (11) 6.97 µM KI + 2.85 µM IAA; (12) 9.29 µM KI + 5.71 µM IAA; (13) 13.9 µM KI + 5.71 µM IAA; (14) 4.65 µM KI + 2.68 µM NAA; (15) 6.97 µM KI + 2.68 µM NAA; (16) 9.29 µM KI + 5.37 µM NAA; (17) 13.9 µM KI + 5.37 µM NAA; (18) 0.89 µM BAP; (19) 2.22 µM BAP; (20) 4.44 µM BAP; (21) 8.88 µM BAP.
Based on morphological responses, initially developed shoots in different media were two time sub-cultured for multiplication and regeneration of shoots in modified MS media supplemented with (4) 8.88 µM BAP + 5.71 µM IAA; (22) 8.88 µM BAP + 1.14 µM IAA; (26) 1% charcoal + 7.4 µM IBA; (27) 1% charcoal + 9.84 µM IBA; (28) 0.93 µM KI + 5.71 µM IAA; (29) 2.32 µM KI + 8.56 µM IAA; (30) 2.85 µM IAA.
Newly formed microshoots measuring 1 - 2 cm were cultured in rooting media (1) MS; (4) 8.88 µM BAP + 5.71 µM IAA; (29) 8.56 µM IAA + 2.32 µM KI; (31) 2.85 µM IAA in  MS. Rooted plantlets were removed from culture media, were washed in autoclaved distilled water to remove media from their rooting surface nicely. The plantlets were transplanted in plastic pots containing autoclaved compost soil (1:1 mixture of peat-substrates and potting soil) and were kept in transparent small covered chamber to keep moist. The plants were acclimatized in room conditions at 25˚C ± 3˚C, 16/8 hours photoperiod and watered regularly at 3 days interval.
MS. Rooted plantlets were removed from culture media, were washed in autoclaved distilled water to remove media from their rooting surface nicely. The plantlets were transplanted in plastic pots containing autoclaved compost soil (1:1 mixture of peat-substrates and potting soil) and were kept in transparent small covered chamber to keep moist. The plants were acclimatized in room conditions at 25˚C ± 3˚C, 16/8 hours photoperiod and watered regularly at 3 days interval.
Based on morphological responses of explants, percentage of shoot initiation response, shoot initiation frequency, percentage of shoot proliferation in first and second transfer, their shoot multiplication frequency, plant regeneration capacity, shoot length, root frequency were analyzed. The shoot initiation frequency was measured by calculation of number of shoots initiated/explant. Plant regeneration capacity was calculated per explants by counting total number of shoots initiated and their multiplication by two cycles of proliferation after second transfer.
2.5. Statistical Analysis
The experiments were carried out by using completely randomized design (CRD) with three replications of each experiment and 3 - 5 explants per replication. Data were analyzed using SPSS (SPSS Inc. Version 17.0), statistical package. Means of 3 replicates per treatment were compared with control as well as multiple treatment groups. Mean comparisons were performed at P ≤ 0.05 level of significance using one way analysis of variance (ANOVA) according to Duncan’s multiple range test (DMRT) using JMP (version 9) statistical software.
3. Results and Discussions
3.1. Effect of BAP, IAA, KI and NAA on Shoot Induction
Percentage of multiple shoot induction from nodal and apical segments in modified MS media supplemented with BAP, KI, IAA, NAA has presented in Figure 3. The highest response of 100% shoot initiation from nodal explants was obtained in media containing 8.88 µM BAP with 5.71 µM IAA after 4 weeks of culture. The multiple shoot induction effect of BAP in combination with IAA was 15% higher than KI and IAA. It is clear from this study BAP alone was less effective to induce shoot than its combination with other hormones (IAA, NAA). Differential response in shoot initiation frequency was observed in presence of different combinations and concentrations of PGRs those were statistically analyzed. Interactive effects of culture media on mean number of shoot induction per explants (shoot initiation frequency) were statistically significant (P ≤ 0.05) has shown in Table 1. Analysis of variance revealed different PGRs concentrations and combinations significantly affect shoot induction frequency. The highest shoot initiation frequency of Heliotropium kotschyi was 10.6 on MS medium supplemented with 8.88 µM BAP with 5.71 µM IAA after 4 weeks of culture (Figure 4). Mean comparisons and analysis of variance significantly showed differences in different treatments and the media with 8.88 µM BAP and 5.71 µM IAA was the most effective to induce maximum number of shoot (Table 1). In this media, the nodal explants started shoot initiation within seven days of culture initiation and initially developed shoots showed good growth (Figures 1(F), (G)) while shoot initiated in 13.9 µM KI + 5.71 µM IAA, showed very slow growth after four weeks (Figure 1(E)). MS media without any PGRs showed no shoot initiation response (Figure 1(D)). The surface sterilization of explants is very essential for establishment, as well as for optimum induction of plantlet regeneration under in vitro condition [15] . In several studied different types of explants were compared for shoot regeneration and most of the studied concluded stem internodes were more productive than others [16] [17] . The shoot tip, nodal explants, and roots also produce large number of genetically identical clones in Bixa ovellana L. [18] [19] . The ratio of auxins and cytokinins is an important factor to induce shoots and shoot growth (Figures 1(E)-(G)). Here, 1.5 ratio of BAP/IAA induced the highest shoot initiation frequency. Similarly, the highest shoot regeneration frequency was reported in threatened medicinal plant Salvia lutea L. under similar hormonal balance [20] . While the highest direct multiple shoot induction in medicinal plant Heliotropium indicum L. was observed on MS medium supplemented by 2.0 - 2.5 mg/L BAP with 0.5 - 1.0 mg/L IBA [21] . Similarly, in medicinal plant, Oroxylum indicum, the two cytokinins (BAP and KI) were used for direct in vitro regeneration experiment, where BAP induced significantly higher percentage of shoot initiation and mean number of shoots in compare to KI here [22] . The effect of BAP as the most reliable, useful and effective PGR for shoot proliferation, for breaking dormancy in several medicinal plant species were observed in several other studies [23] -[26] . The lower concentration of IAA (5.71 µM) with higher concentration of BAP (8.88 µM) triggered the breaking of apical dominance by inhibiting the apical peak on the side bud and stimulated proliferation of axillary shoots (Figure 1(G)). Similarly, several studies reported that lower

Figure 3. Effect of various growth regulators to modified MS media on in vitro shoot initiation percentage after two and four weeks of culture. Results are mean ± SE of shoot development per explants.
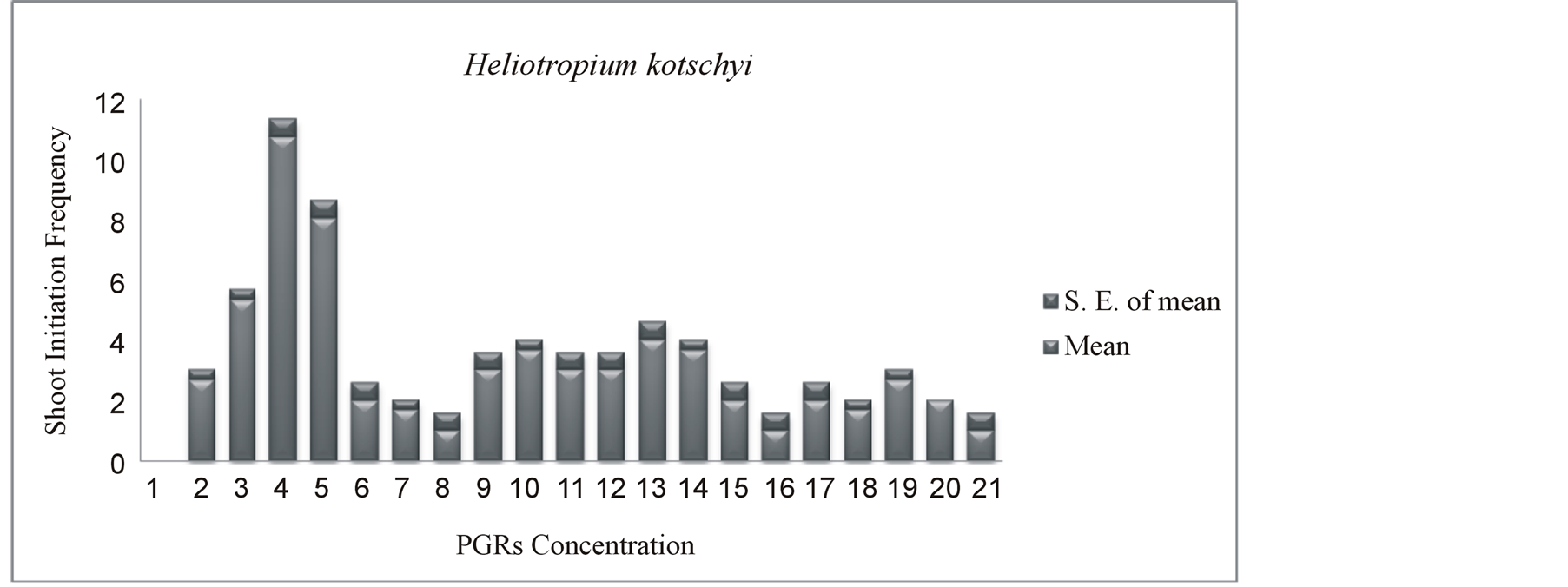
Figure 4. Effect of BAP, KI, IAA, NAA on modified MS media on in vitro shoot initiation frequency from cultured explants after 4 weeks of culture.
concentration of auxins with higher concentration of cytokinins help to overcome apical dominance [17] [27] as observed here.
3.2. Effect of BAP, KI, IAA, IBA and NAA on Shoot Multiplication
The effect of different PGRs in the first time multiplication of initially developed shoots from different media were studied by transferring in different media (Figures 1(H)-(K)). The shoot multiplication frequency was higher in first transfer in compare to initial shoot induction frequency. The highest shoot multiplication was noticed while initial shoots developed in different media were transferred to modified MS media containing 8.88 µM BAP and 5.71 µM IAA for multiplication (Table 2). 100% shoot proliferation associated with the highest shoot multiplication frequency of 15 was observed while initial explants were cultured in 6.66 µM BAP and 2.85 µM IAA, then were transferred to media containing 8.88 µM BAP with 5.71 µM IAA. The lowest shoot multiplication frequency of 2.5 with 75% shoot proliferation response was observed while initial shoots developed in presence of 13.3 µM BAP with 5.37 µM NAA and were transferred to 8.88 µM BAP with 5.71 µM IAA (Table 2). Initial shoots developed in 4.44 µM BAP were transferred (Figure 1(H)) to different proliferation media containing 8.88 µM BAP with 1.14 µM IAA, 4.92 µM IBA, 7.38 µM IBA and 9.84 µM IBA, showed 100% multiplication response with different frequencies of shoot proliferation. The highest shoot proliferation frequency of 20 was found while initial shoots developed in presence of 6.66 µM BAP with 2.68 µM NAA, were transferred to media with 0.93 µM KI + 5.71 µM IAA and final transfer in media without PGRs. The moderately

Table 1. Effect of various growth regulators supplemented to modifed MS media on in vitro shoot initiation response from nodal explants of H. kotschyi after 4 weeks of culture. Results are mean of shoots developed per explant of 3 sets of individual experiments. Means followed by the same letter are not significantly different at P ≤ 0.05 according to DMRT.

Table 2. Effect of BAP, KI, IAA, IBA and NAA on percentage of shoot multiplication frequency after first (column first three from left side) and second (then next three) transfer of initially developed shoots of H. kotschyi.
higher proliferation frequency of 15 was observed in the second transfer, while initial shoots developed in media supplemented with 8.88 µM BAP with 5.71 µM IAA and 8.88 µM BAP with 5.37 µM NAA, then were first transferred to the same media composition of 8.88 µM BAP with 5.71 µM IAA for multiplication and finally in media consisted of 2.85 µM IAA and MS media without any hormone (Table 2). 100% response of shoot multiplication with shoot multiplication frequencies of 7.5 and 4.5 was observed in MS media supplemented with 1% charcoal with 4.92 µM IBA and 7.4 µM IBA, respectively. Several author suggested important role of activated charcoal in culture media, it may remove contaminants from agar [28] help to absorb secondary products secreted by the cultured tissue [29] [30] regulate the supply of certain endogenous growth regulators [31] , stimulate embryogenesis [29] .
The comparative effect of different PGRs in stimulating shoot multiplication response showed more effectiveness of BAP with IAA and it is the fact of total hormonal response that has been gathered from initial culture. Similar results were reported by several researchers in different plants [32] [33] . BAP alone can induce shoot but the frequency of shoot initiation was significantly lower than combine treatment of BAP with others (Table 1). While several other studies reported that BAP alone was effective for shoot induction as well as for lateral shoot multiplication [34] -[36] . It was evident from the obtained data that subculture enhanced both shoot multiplication and shoot length (Figures 1(J), (K)). The shoots produced in second subculture (Figures 1(J), (K)) were longer than those produced in first subculture (Figure 1(I)). The higher concentration of BAP induced more number of shoots but smaller in size in lemon balm [23] , Gentiana kurroo [37] , and water melon were reported [38] . To overcome the growth inhibition effect of BAP, auxins (IAA, NAA, IBA) were combined to trigger shoot multiplication efficiency at the same time to enhance shoot length. As BAP enhanced multiple shoot formation from apical and nodal segments, in this study, the combine effect of BAP with IAA in shoot initiation, first time shoot multiplication and later on transfer in MS media without hormone or presence of auxins helped in shoot elongation and normal development of plantlet was observed.
3.3. Plant Regeneration Capacity
Plant regeneration capacity of Heliotropium kotschyi was calculated based on shoot initiation frequency of explants and multiplication frequency of initially developed shoots after first and second transfer. The highest plant regeneration capacity of Heliotropium kotschyi was observed when shoot initially developed in media containing 8.88 µM BAP with 5.71 µM IAA, then were transferred to the same media and later on to the medium without any hormones. While shoots induced in presence of 8.88 µM BAP with 5.37 µM NAA and similar transfer showed almost half plant regeneration capacity (Figure 5). In the comparative study, although higher shoot multiplication ratio observed in several other media during first and second transfer, but the ultimate conversion of shoot regeneration capacity based on shoot initiation frequency those were lag behind (Table 3). Baksha et al. [39] reported that basal medium containing 4 mg·L−1 BAP and 1 mg·L−1 of IAA, increased the number and growth rate of multiple shoots in Aloe barbadensis [39] . The optimum level of auxin and cytokinins for shoot regeneration, synergistic effect of complex effect to improve the frequency of regeneration was studied

Figure 5. Plant regeneration capacity of H. kotschyi after 2nd transfer in media 1 while shoots were initially developed from explants in different media 1, 4, 6, 7, 8 media and were multiplied in media 4.
in Bupleurum fruticosum [40] . Salvia africana-lutea L. produced 2000 - 3000 plants from one shoot after long subculture [20] . The effectiveness of BAP in axillary shoot regeneration has been reported in a number of plant species [41] -[44] . Mohebalipour et al. [45] reported the addition of 2 mg·L−1 BAP to MS medium significantly increased the in number of shoots per explants in most of the genotypes of Iranian lemon balm (Melissa officinalis L). Plant regeneration ability in the culture medium was usually enhanced by the combine addition of auxins and cytokinins was reported [46] . The in vitro regeneration of shoots is an essential base for most methods of genetic transformation [47] . Nodal segments of Melissa officinalis L. showed in vitro plantlet development in presence of different concentration of BAP and IAA [45] . The stages of regenerated plantlet development of Heliotropium kotschyi in vitro micropropagation has presented in Figure 1.
3.4. Rooting of Microshoots
The frequency of root development varied depending on hormonal combinations and concentration in different media starting from shoot initiation, first and second multiplication media used. The highest root frequency (Root/plantlet) of Heliotropium kotschyi of 5 with 27 mm of shoot length after second transferred was observed when shoot initially developed on modified MS media supplemented with 4.65 µM KI with 2.68 µM NAA then were transferred to modified MS media with 2.85 µM IAA and finally transferred to medium without any hormones has shown in Table 4. The effect of hormone in rooting is the combined result of different hormone, as second transfer in media without any PGRs showed root development in different frequencies (Figure 1(L)). Formation of root occurred in hormone free MS media while previous growing media of axillary shoot contained different concentrations and combinations of auxins (IAA, NAA) and cytokinins (BAP and KI). The similar effect was observed in lemon balm as reported by Mohebalipour et al. [45] . Moreover, the ability of plant tissues to form roots depends on the interactions of different endogenous and exogenous factors [48] . The effect of IAA is more prominent in inducing large number of roots in MS medium containing 5.71 µM IAA than media without any growth hormones as was reported by Ujjwala, 2007 [49] . Similarly, several researcher reported that IBA was more effective in rooting of several plant species [48] [50] [51] . The well development of rooting system of in vitro regenerated plantlets is very important factor for transfer to in vivo condition, particularly for acclimatization, growth and development. Plantlets with well developed roots (Figures 1(L)-(N)) were gradually exposed to normal room conditions in plastic soil pots containing mixture of autoclaved soil (Figure 1(O)) and 60% plantlets survived.
4. Conclusion
This is the first time report of using tissue culture methods in the purpose of endangered plant conservation in Bahrain. Development of axillary shoots, rapid shoot multiplication and high plant regeneration frequency has

Table 3. Effect of different media on plant regeneration ratio of H. kotschyi.

Table 4. Effect of different media for root initiation of H. kotschyi. Roots developed in second transfer in different proliferation media. Results are mean shoot length and root frequency of 3 sets of individual experiments has present.
obtained by culturing apical and nodal explants of Heliotropium kotschyi in presence different hormones with different combinations. The effect of BAP and IAA on shoot initiation, multiplication and plant regeneration is more prominent than other hormones. Considering the great importance of the wild, multipurpose, threatened medicinal plant of Bahrain, the establishment of in vitro culture and plant regeneration protocol is very important initial step of ex situ conservation strategy.
Acknowledgements
The work was supported by College of Graduate Studies, Desert and Arid Zone Sciences Program, Arabian Gulf University, Manama, Kingdom of Bahrain.
NOTES