1. Introduction
The Gram-negative bacterium Neisseria meningitidis (Nm) is one of the main etiologic agents of bacterial meningitis and septicaemia [1]. It is associated with a significant mortality and has a case fatality rate of 10% [2] reaching up to 55% in patients with fulminant septicaemia [3]. Despite significant progress made in the management of patients and the availability of vaccines against serogroups A, C, Y and W135, serogroup B (NmB) disease remains a major public health problem in the UK, Europe and the Australias, accounting for 85% - 90% of meningococcal disease cases in the UK [4].
Nasopharyngeal carriage of Nm has a population prevalence of between 5% and 40%, with the highest rates of carriage in teenagers, household contacts and military recruits [5,6]. In comparison to carriage, disease incidence is relatively rare. One of the factors associated with the relatively low disease prevalence is carriage of commensals, such as N. lactamica, in the nasopharyngeal mucosa [7,8]. In support of these studies, we have more recently shown that N. lactamica induces secretion of broadly protective innate IgM [9] and suppresses inflammation during NmB infection of nasopharyngeal cells in vitro using a toll-like receptor 2 (TLR2)-dependent signalling mechanism [10].
In addition to N. lactamica, other species of commensal lactic acid-producing bacteria have been isolated from the upper respiratory tract (URT) [11-15], including several Lactobacillus (LB) species [11,12,16,17]. Although some LB strains are reported to exhibit direct antimicrobial properties against oral pathogens [11,12], no studies to our knowledge have determined the protective influence of oropharyngeal-derived LB isolates against pathogenic NmB. Studies on LB isolates from the gut [18] and urogenital tract [19-22], however, show that they can promote local barrier function and reduce inflammation [18-21], reduce N. gonnorhoea adhesion [22], E. coli K1 invasion [23] and Group A streptococcal cytotoxicty [24]. In this study, we have evaluated for the first time the influence of a panel of oropharyngeal-derived LB isolates, with known antimicrobial properties [11,12], and evaluated their potential to afford innate immune protection to nasopharyngeal derived epithelial cells; against the nasopharyngeal coloniser and Gram negative pathogen, NmB.
2. Material & Methods
2.1. Bacterial Isolates and Culture Conditions
NmB strain MC58 was cultured from frozen stocks on HBHI agar and enumerated as previously [10]. LB were obtained from the National Collection of Industrial, Marine and Food Bacteria (NCIMB) and include the oral isolates L. casei subsp.casei (NCIMB 8822), L. gasseri (NCIMB 8819), L. plantarum (NCIMB 8825), L. salivarius subsp. salivarius (NCIMB 11975), L. rhamnosus (NCIMB 6375) [11,16,25-27]; and the gastro-intestinal isolate L. rhamnosus GG [28]. LB strains were grown in MRS broth overnight in a shaking incubator at 37˚C. Bacteria were re-suspended in PBS-B (replete with calcium and magnesium) and infectious dose adjusted as required (OD 0.2 at 600 nm ≈ 2.5 × 107 cfu per ml).
2.2. Nasopharyngeal Cell Line
A human nasopharyngeal epithelial cell line, Detroit 562 (ATCC CCL 138, ECACC 87042205), was grown as described elsewhere but in the absence of antibiotics [29]. Cells were used at 80% - 90% confluence and between passage numbers 3 to 10 in all experiments.
2.3. Preparation of Heat Killed Bacteria and Pre-Conditioned Media
Bacteria were inactivated by heating to 65˚C for 30 min in PBS-B. Inactivation was confirmed by overnight culture in MRS broth and performing viable counts. Preconditioned media was generated from LB grown in complete assay medium (M199 + 2% FCS + HEPES) at 37˚C for 6 h in the presence of Detroit 562 cells at the concentration of 200 bacteria/cell, or multiplicity of infection (MOI) 200. Bacteria were removed by centrifugation (15 min, 4000 × g) and the supernatant was sterilized by filtration (0.2 μm), adjusted to pH 7.2 with 0.1 M NaOH, and stored at −20˚C until use.
2.4. Preparation of Bioluminescent N. meningitidis Strain MC58
Plasmid pLKMP, described elsewhere [30], was used to Lux-transform NmB into a bioluminescent organism. In brief, spot overlay assays were performed with NmB overlaid onto a dried spot of plasmid DNA (1 µg) on HBHI agar, which was then incubated for 24 hours at 37˚C and 5% CO2. Transformed bacteria were picked and subsequently re-selected using HBHI agar containing 150 µg/mL kanamycin. The resultant bacterial isolates were confirmed to be NmB using colony morphology, gram-stain and differential biochemical tests. Lux-MC58 was confirmed to have no growth advantage or disadvantage over wild-type MC58 by analysis of viable counts (cfu/ml) and optical density over time. Lux-MC58 was confirmed to have no adhesion or invasion advantage or disadvantage over wild-type MC58 by standard gentamicin protection assay (not shown) [31].
2.5. Cytokine Induction Assays
Cells (105 cells per well) were pre-incubated with live LB at a multiplicity of infection of 2-200 bacterial per cell (MOI 2, 20, 200), heat-killed LB (MOI 20) or conditioned media (0.1ppu or 10%) for 3 h and then challenged with NmB (MOI 200), TLR2 ligand PAM3Cys (100 ng/mL), or early response cytokine IL-1β (10 ng/mL), for an additional 3 hrs at 37˚C and 5% CO2. Cells were washed and cultured for a further 18 h in media containing gentamicin (200 µg/mL), supplemented with IL1-1β or Pam3Cys as required. Supernatants were filtered (0.2 µm) and stored at −80˚C until ready for testing by ELISA, as previously described [10].
2.6. Determination of Cytotoxicity
Cell monolayers were pre-incubated with LB (MOI 2, 20, 200) for 3 hrs and challenged with NmB (MOI 200) for additional 3 hrs, as previously. Cells were washed and incubated in M199 containing gentamicin (at 200 µg/ml) but without phenol red for a further 18 h. Plates were centrifuged and supernatants collected for assessment of extracellular lactate dehydrogenase (LDH) activity, as a measure of cellular necrosis [32], according to the manufacturers’ instructions (Roche Applied Science, UK). Monolayers were then stained with 5 µg/mL propidium iodide [33] and read on a fluorescence microplate reader with excitation at 540 and emission at 620 nm (Fluostar Optima, BMG Labtech). Triton-X (0.1%) and Saponin (1%) were used as positive control treatments. Untreated cells were used as negative controls.
2.7. Adhesion and Invasion—Standard Gentamicin Protection Assay
The number of total cell-associated bacteria (adhesion and invasion) was determined using standard gentamicin protection assay [31]. In brief, confluent monolayers of Detroit 562 cells, were challenged with LB strains (MOI 200) in M199 containing 2% FCS and incubated for 3 hrs in a humidified incubator at 37˚C and 5% CO2. Cells were then washed with Hanks balanced salt solution, lysed in 1% saponin for 10 min, and plated on MRS agar to determine the total number of cell-associated LB bacteria.
To enumerate the intracellular bacteria, cells were challenged for 3 h and incubated with gentamicin (200 μg/mL) for 90 min to kill the extracellular bacteria prior to saponin treatment. The minimum inhibitory concentration of gentamicin for all isolates was previously confirmed as less than or equal to 100 μg/mL. Controls were performed to determine the amount of bacterial growth during the 3 h assay and the amount of bacteria sticking to the side of the wells, which was then subtracted from adhesion and invasion values. Subtraction of the intracellular bacteria from the cell-associated bacteria gives the total number of adherent bacteria.
To examine LB-induced protection against NmB adhesion and invasion, cells were pre-incubated for 3 h with L. plantarum or L. salivarious (MOI 200) prior to NmB challenge (MOI 200) for additional 3 h, as above, or co-incubated together for 3 h. Cells were treated with gentamicin and lysed in 1% saponin, as previously. NmB and LB were cultured on HBHI plates for 24 - 48 hrs at 37˚C in 5% CO2 and colony counts were performed. Bacterial counts for LB were determined on MRS plates. These values were subtracted from total colony counts on HBHI plates to give NmB counts. Direct inhibition of meningococcal growth by LB on the HBHI plates was ruled out using standard spot-dilution assays.
2.8. Bacterial metabolism—Bioluminescence Assay
Using Lux-transformed NmB, we analysed the ability of L. plantarum, L. salivarious (MOI 20), heat-killed organisms (MOI 20), or conditioned media (0.1 ppu) to inhibit light emission and thus metabolism of NmB (MOI 200) over time [34]. A Floustar Optima (BMG Labtech) was used to record the light output and maintain the plates at 37˚C and in 5% CO2.
NmB-Lux was used to examine the effect of LB on NmB metabolism in the presence and absence of cells. A standard gentamicin protection assay was performed, except that Detroit 562 cells were grown in 96-well blacksided tissue culture plates (Genetix). Cells were pre-treated with LB (MOI 20), heat-killed LB (MOI 20) or conditioned media (0.1ppu) for 3 hrs prior to incubation with bioluminescent NmB (MOI 200). As previously, gentamicin was added 3 h after NmB-Lux to kill extracellular bacteria and 1% saponin was added at 4.5 h to allow killing of the intracellular bacteria by the antibiotic. Differences in light output preand 1.5 h post-saponin treatment were used to reflect the levels of metabolising intracellular NmB.
2.9. Statistical Analysis
For comparison of MC58 responses in the presence of increasing doses of each of the different LB strains (L. caseii, L. gasseri, L. plantarum, L. salivarious, L. rhamnosus, L. rhamnosus-GG) One-way ANOVA analysis of variance were performed. For comparison of MC58 responses in the presence and absence of different LB strains (L. caseii, L. plantarum, L. salivarious), conditioned media or heat-killed organisms (CM, HK) paired student t-tests were performed, with Bonferroni correction for multiple testing. All experiments were performed in at least three independent experiments and each of these was performed in triplicate.
3. Results
3.1. LB Show a Differential Ability to Attenuate Epithelial Inflammation in Nasopharyngeal Epithelial Cells
NmB strain MC58 strongly induced IL-6 secretion (400 - 500 pg/ml) and moderately induced TNF-α (20 pg/ml) from nasopharyngeal derived Detroit 562 epithelial cells (Figure 1). In contrast commensal LB strains (oral isolates L. casei subsp. Casei, L. gasseri, L. plantarum, L. salivarius subsp. Salivarius, L. rhamnosus and gut isolate L. rhamnosus GG) only weakly stimulated IL-6 (<10 pg/ml) and TNF-α (5 - 10 pg/ml) secretion from these cells. Importantly oral L. plantarum and L. salivarius were able to attenuate NmB induced IL-6 and TNF-α during co-culture by approximately 50% - 70% compared with NmB alone. Attenuation was apparent whether L. plantarum and L. salivarius were lower or equiva-

Figure 1. L. plantarum and L. salivarious attenuate NmB-induced IL-6 and TNF-α secretion. Detroit 562 nasopharyngeal cells were pre-incubated with Lactobacilli for 3 h (at a multiplicity of infection or moi of 2, 20, 200 bacteria per cell) and then challenged for 3 h with NmB strain MC58 (at moi 200). Cells were washed and cultured for 21 h with antibiotics. Cytokines IL-6 and TNF-α were quantified in cell culture supernatants by ELISA. Cells challenged with NmB (MC58) alone were compared with those pre-treated with Lactobacilli. The data represent mean values ±SEM from three independent experiments each performed in triplicate. *p < 0.05, **p < 0.01, ***p < 0.001.
lent in concentration to NmB (*P < 0.05, **P < 0.01, ***P < 0.001). In contrast the gut-associated LB strain L. rhamnosus GG and oral strain L. rhamnosus were unable to attenuate IL-6 and TNF-α response in NmB-infected nasopharyngeal epithelial cells. Modest reductions were demonstrated with oral strains L. casei and L. gasseri these were not statistically significant.
3.2. Conditioned Media from L. plantarum and L. salivarius Are Sufficient to Attenuate NmB-Induced Inflammation
Heat-killed (HK) L. casei, L. plantarum and L. salivarius significantly attenuated IL-6 responses induced by NmB (Figure 2), albeit to differing degrees (20%, P < 0.01 – 60%, P < 0.05), demonstrating that LB viability is not essential for attenuation of inflammation by these spp. Conditioned media (CM) containing secretions from live L. plantarum and L. salivarius but not L. casei were also found to strongly attenuate the IL-6 response induced by NmB by 42% (P < 0.001) - 62% (P < 0.05), which we suggest contributes to the strong ability of these strains to attenuate in live co-cultures.
3.3. Live and Heat Killed LB Strains but Not LB-Conditioned Media Are Able to Attenuate Inflammation Induced via TLR-2
The specificity of the observed anti-inflammatory effect was further evaluated by stimulating cells with the TLR- 2 ligand Pam3Cys (100 ng/ml) in the presence and absence of LB. There was a marked reduction (60% - 75%) in secretion of IL-6 when cells were treated with live L. plantarum (P < 0.001), L. salivarius (P < 0.01) or L. casei (P < 0.01) prior to stimulation with Pam3Cys (Figure 3) but not when treated with the early response cytokine IL-1β (10 ng/ml) (not shown). Significant IL-6 attenuation was also evident from Pam3Cys-stimulated cells when co-cultured with heat-killed L. plantarum or L. salivarius (P < 0.05) but not with L. casei, or conditioned media from either organism (changes were non-significant), suggesting that conditioned medium from L. plantarum or L. salivarius attenuates inflammation through a TLR-2 independent mechanism.
3.4. LB Strains Are Able to Inhibit NmB-Induced Necrotic Cell Death
NmB is cytotoxic to epithelial cells [35] and has been
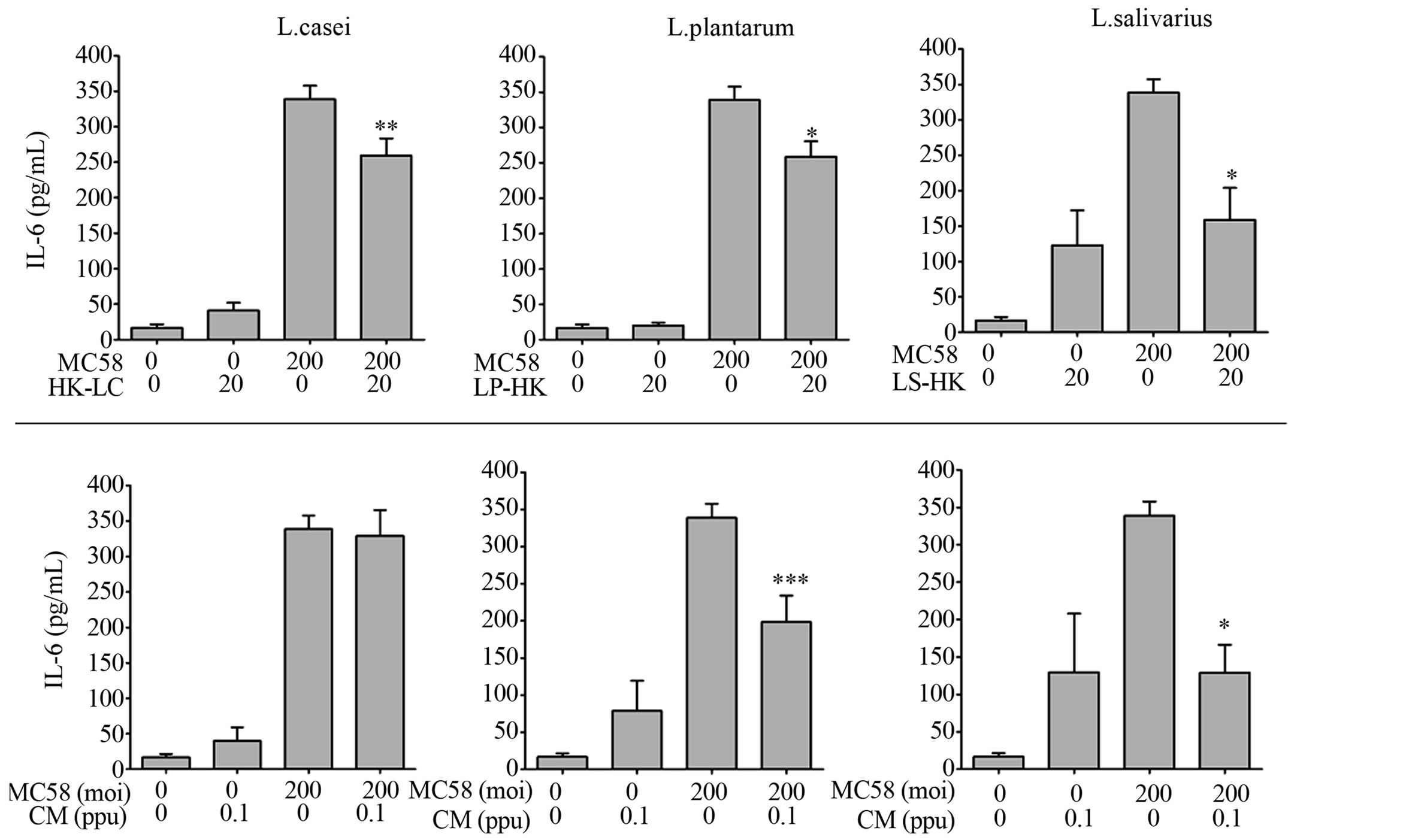
Figure 2. Heat-killed LB and conditioned media from L. plantarum and L. salivarious also attenuate NmB-induced inflammation. Detroit 562 cells were pre-incubated with heat-killed Lactobacilli (HK, moi-20), or conditioned media, (CM, 0.1 part per unit in M199 with 2% FCS, pH = 7.2) and challenged with NmB strain MC58 (moi 200). IL-6 was quantified in cell culture supernatants by ELISA. Cytokine levels from cells challenged with NmB alone were compared with those pre-treated with Lactobacilli. The data represent mean values ±SEM from three independent experiments each performed in triplicate. *p < 0.05, **p < 0.01, ***p < 0.001.

Figure 3. Live LB but not LB conditioned media attenuate inflammation induced through TLR-2 receptor. Detroit 562 cells pre-treated with live LB, heat killed LB (moi-20), or conditioned media (0.1 ppu) for 3 hrs prior to challenge with the TLR2 ligand Pam3Cys (100 ng/ml), or IL1β (10 ng/ml). Cytokines were quantified from supernatants generated over 21 h of culture using ELISA. Cytokine levels from cells challenged with Pam3Cys or IL1β alone were compared with those pre-treated with LB. The data represent mean values ±SEM from three independent experiments each performed in triplicate (*p < 0.05, **p < 0.01, ***p < 0.001).
reported to induce cell necrosis in other cell types [36], which is associated with induction of inflammation and weakening of the mucosal barrier function. We therefore examined nasopharyngeal epithelial cells for necrotic cell death in response to NmB infection and potential cytoprotection afforded by LB. NmB induced a low but significant level of necrotic cell death in Detroit 562 monolayers following 3 h challenge and 21 h culture, which was NmB dose dependent (P < 0.001) (Figure 4), and equivalent to death of approximately 5% - 8% of cells. LB strains alone were not cytotoxic to these cells (data not shown). Culture of cells with NmB in the presence of L. plantarum, L. salivarius or L. casei, however, led to a significant reduction in NmB-induced necrosis, as deter-

Figure 4. LB strains, particularly L. plantarum, inhibit NmB-induced necrotic cell death. Detroit 562 cells were pre-incubated with Lactobacilli (at moi-200) before co-incubation with a range of NmB doses (moi 2, 20, 200, 2000). Cytotoxicity was measured by propidium iodide staining of cells in situ after 24 hr culture and staining intensity was quantified by plate reader giving values in mean fluorescence units (MFU). The enzyme activity of cytosolic lactate dehyrogenase was also quantified from cell culture supernatants and expressed as a percentage of the positive control value to give percentage cytotoxicity. The data represent mean values ±SEM from three independent experiments each performed in triplicate (*p < 0.05). Triton-X treated cells were used as a positive control.
mined by propidium iodide staining (P < 0.05). Using lactate dehydrogenase enzyme release as an alternative and more direct measure of cell necrosis, only L. plantarum was significantly able to reduce necrotic cell death in nasopharyngeal derived epithelial cells challenged with NmB (P < 0.05).
3.5. L. plantarum Inhibits Adhesion to and Invasion of Nasopharyngeal Epithelial Cells by NmB
We postulated that the differential protection afforded by LB strains may be related to their ability to adhere to nasopharyngeal epithelial cells and interfere with the adhesion and invasion of NmB. Using standard gentamicin protection and viable counts [31] we demonstrated that LB isolates, including the gut isolate L. rhamnosus GG, were readily able to adhere to Detroit 562 epithelial cells at levels of approximately 1 to 2 bacteria per cell, or 1% of inoculum (Figure 5(a), grey fill). In comparison L. salivarius and L. rhamnosus were roughly 10 fold less adherent and L. rhamnosus, in particular, was less able to grow in culture medium over the period of the experiment than the other isolates (Figure 5(a), black fill). Only L. casei, L. rhamnosus GG and L. plantarum were found to invade nasopharyngeal epithelial cells, albeit to variable degrees (equivalent to 1.68, 0.86 and 15 per 1000 epithelial cells, respectively) (Figure 5(a), white fill).
In contrast to LB, NmB demonstrated adhesion levels of approximately 10 bacteria per cell (10% - 20% of inoculum) and invasion levels of 2 bacteria per 100 cells, similar to L. plantarum (Figure 5(b), black fill). Coincubation of cells with L. plantarum (MC58 + L. plantarum) or L. salivarius (MC58 + L. salivarious) reduced NmB adhesion to Detroit 562 cells by 65% and 87% respectively (Figure 5(b) and c, grey fill). L. plantarum inhibited recovery of live intracellular NmB by 93% (Figure 5(b), grey fill), where as L. salivarious inhibited recovery by 65% (Figure 5(c), grey fill). In bacteria only controls, NmB growth was also reduced by 43% in the presence of L. plantarum but not L. salivarious (not shown).
3.6. L. plantarum, L. salivarius and Conditioned Media from L. plantarum Inhibit NmB Metabolism
The influence of L. plantarum and L. salivarius on NmB was further explored using Lux-transformed NmB, which

Figure 5. L. plantarum and L. salivarious inhibit NmB adhesion and invasion of nasopharyngeal epithelium. (a) The ability of the different LB strains to adhere and invade nasopharyngeal epithelial cells was determined using standard gentamicin protection assay and viable counts. Monolayers of Detroit 562 cells were incubated with each LB strain (moi 200) for 3 hrs. The dotted line represents the limit of detection of the assay. Data represent means ±SEM from three independent experiments, each performed in triplicate. (b) The number of NmB adhering and invading epithelial cells in the presence (grey fill) and absence of L. plantarum (black fill). (c) The number of NmB adhering and invading epithelial cells in the presence (grey fill) and absence of L. salivarious (black fill). In (b) and (c) Detroit 562 cells were pre-incubated with LB for 3 h (moi 200) and challenged for a further 3 h with NmB (moi 200). Data represent means of two independent experiments each performed in triplicate.
emits light in the visible spectrum and is a highly sensitive reporter of bacterial metabolism that can be monitored in real-time [34]. In bacteria only controls, light output from NmB-Lux was reduced to background levels within 3 - 6 hrs of co-culture with live L. plantarum or L. salivarius (Figures 6(a) and (b), P < 0.001, open circles). Conditioned media from L. plantarum reduced light output, by 60% within an hour (P < 0.05) and maintained this level of reduction over the 12 h time course of study (Figure 6(a), inverted triangles). In contrast no reduction in NmB-Lux light output was observed with heat-killed L. plantarum (closed squares), heat-killed L. salivarius (Figure 6(b), closed squares) or conditioned media from L. salivarius (Figure 6(b), inverted triangles).
3.7. L. plantarum and Conditioned Media Inhibit the Detection of Metabolising Intracellular NmB
Crucially we next examined the effects of LB on NmB metabolism during host epithelial cell invasion, using a standard gentamicin protection assay [31] but measuring light output rather than viable counts [34]. Cells were incubated with bioluminescent NmB for 3 h then treated with gentamicin to kill the extracellular bacteria, so that the light remaining represents metabolising bacteria that have invaded the epithelial cells. Light output from intracellular NmB was almost completely absent in the cells pre-incubated with live L. plantarum compared to cells

Figure 6. L. plantarum and its conditioned media inhibits NmB metabolism directly and during epithelial cell invasion. (a) The direct effect of live L. plantarum (moi 200), heat-killed (HK) or conditioned media (CM, 0.1ppu) on NmB metabolism (moi 200) was determined in microbial co-cultures by measuring light output from MC58 lux+ over time. (b) The effects of L. salivarius (live, HK, CM) on NmB was similarly determined and representative experiments are shown. (c) The effects of L. plantarum, HK and CM on NmB metabolism during epithelial cell invasion were determined using a standard gentamicin protection assay but measuring light output from MC58 lux+ as the endpoint. Epithelial cells were pre-incubated with L. plantarum, HK or CM and challenged 3h later with NmB lux+. (d) Cells were pre-incubated with live L. salivarius, HK, or CM, and challenged with NmB as above. Data for (c) and (d) represent means of three independent experiments each performed in triplicate (*P < 0.05, ***P < 0.001).
treated with NmB alone (Figure 6(c), P < 0.001), mirroring results from the gentamicin protection assay. Using this more sensitive assay, a significant reduction in light output was also observed (60%) when cells were pretreated with conditioned medium from L. plantarum (P < 0.05) but no reduction was afforded by pre-incubation with heat-killed L. plantarum. Live L. salivarious reduced light output by approximately 40%, again mirroring results from the standard gentamicin protection assay. In contrast, neither heat-killed L. salivarious, nor conditioned media from L. salivarious, was able to significantly reduce intracellular NmB light output (Figure 6(d)).
4. Discussion
4.1. Rationale for Study Design
In this study we describe for the first time how oropharyngeal isolates of LB differentially affect innate immune responses at the nasopharyngeal epithelium and afford protection against an important URT pathogen, NmB.
Five oropharyngeal isolates (L. salivarious, L. casei, L. plantarum, L. rhamnosus, L. gasseri) were selected for their antimicrobial activity against oral pathogens in other studies [11], whilst the well-studied gut probiotic L. rhamnosus GG [28], which has been reported to protect against inflammatory colitis [37,38], was selected for initial comparison.
4.2. Inflammation
Our results highlight that, in contrast to gut derived L. rhamnosus GG, oropharyngeal isolates LB can significantly protect nasopharyngeal derived epithelium from excessive IL-6 release during NmB infection (Figure 1). Like other commensals [10] LB were able to attenuate inflammation mediated through TLR-2 receptor on nasopharyngeal epithelial cells (Figure 3, not all shown), as in other tissues [39]. Only L. plantarum and L. salivarious, however, were able to significantly attenuate both IL-6 and TNF-α responses (Figure 1) and none of the LB was able to attenuate responses to the early response proinflammatory cytokine IL1-β. These results highlight a strain and stimulus-dependent protection by LB, which we suggest is enhanced for L. plantarum and L. salivarious against NmB by a factor secreted in their conditioned medium (Figure 2). In our study inflammatory attenuation by their conditioned medium was largely independent of TLR-2 (Figure 3), in contrast to others [40], so is unlikely to involve LB lipoteichoic acid, which can be shed in vesicles by some strains [41] and is highly anti-inflammatory [42,43].
4.3. Cell Death
The immune system is thought to be activated by tissue damage via the release of danger signals [44]. As such, inflammatory cytokine secretion can be triggered by cell death, in particular necrosis, which causes the release of intracellular nucleic acid, uric acid and heat shock proteins. We therefore examined the possibility that our antiinflammatory LB strains, L. plantarum and L. salivarious, may afford protection against known NmB-induced cytotoxicity [35,45]. All LB strains (Figure 4, not all shown), however, were able to abrogate NmB-induced cytotoxicity, as reported for other pathogens [24,46-48], suggesting LB protection against pathogen cytotoxicity is not directly linked to inflammatory attenuation. Using LDH enzyme activity as a more specific measure of necrotic cell death, only L. plantarum significantly reduced NmB cytotoxicity. In our study this associated with reduced pathogen adhesion (Figure 5), as suggested elsewhere [22,48,49] but most clearly associated with protection against NmB invasion (Figure 5) or intracellular survival (Figure 6).
4.4. Adhesion
Despite the differential ability of our LB strains to protect nasopharyngeal cells from NmB-induced inflammation and cytotoxicity, they were all well able to adhere to Detroit 562 cells, as to other respiratory cell lines [13,14]. We observed 65% - 87% reduction in NmB adhesion to nasopharyngeal cells by co-culturing with L. plantarum or L. salivarious (Figures 5(b)and (c)) but found that there were 10 fold fewer LB adhering than NmB, suggesting direct competition for host receptors used for bacterial adhesion is unlikely to account for it. It is possible that L. plantarum and L. salivarious secrete surface associated inhibitory proteins to reduce adhesion [22, 49] which is consistent with their secretions reducing NmBinduced inflammation (Figure 2) and invasion (Figure 6(c)). It is also possible that the dramatic inhibition of NmB metabolism by L. plantarum and L. salivarious in planktonic culture (Figure 6(a)) may reduce NmB virulence prior to adhesion. NmB growth was also inhibited by L. plantarum but only in planktonic culture. It was not observed in agar overlays, in contrast to other studies of intestinal pathogens [50] and Neisseria gonnorhoea [51], suggesting phase-dependent inhibition of NmB.
4.5. Invasion
Crucially L. plantarum abrogated NmB invasion (Figure 5), or recovery of viable intracellular NmB from nasopharyngeal epithelial cells (Figure 6(c)). L. plantarum was equally invasive as NmB (15 - 20 bacteria per cell), suggesting that they may compete for host receptors used for bacterial invasion [22,52]. Studies on E. coli suggest that L. plantarum could induce changes in epithelial cells to indirectly reduce pathogen association, through ERK phosphorylation [53]. In our study, however, L. plantarum uniquely inhibited encapsulated NmB metabolism using secretory factors (Figure 6(a)), including during epithelial cell invasion (Figure 6(c)). Further studies are needed to investigate the mechanism but suggest that secretions from L. plantarum may be used to protect against NmB invasion and survival within the nasopharynx.
5. Limitations
There are a number of limitations to our study including the fact that gentamicin protection may reflect bacteria that are intimately associated with the host cell [54], which precedes invasion, rather than invasion itself. Use of saponin however demonstrated that an intracellular pool of bacteria were present. In this study we used a cell line which may not reflect responses in primary cells [55]. This cell line is however well-characterised [10] and expresses all the relevant receptors for Neisserial interaction [55]. We also used a representative strain of NmB (MC58) which may not reflect the response of all NmB isolates. However its genome has been fully sequenced [56] and it is highly adhesive and invasive (Figure 5) and thus stringently tested our hypothesis. We cannot rule out the possibility that Lux-transformation altered NmB metabolic behaviour, however growth kinetics of the parent and Lux-variants were found to be the same, and results for standard gentamicin protection assay and viable counts were comparable for Lux-MC58 and MC58 (not shown).
6. Conclusions
Taken together we present strong evidence for protection of nasopharyngeal epithelial cells against NmB induced pathogenicity by commensal L. plantarum. We find, however, that not all LBs are equal with regard to their probiotic potential in the nasopharynx. Caution should therefore be used when assessing the efficacy of probiotics (reviewed by Hao and colleagues [57]), as protection shows strain and pathogen specific. We show that oral LB species, in particular L. plantarum, may be used as a means of providing broad prophylactic mucosal protection against the invasive upper respiratory pathogen NmB, through reduced host cell inflammation and cytotoxicity, plus pathogen adhesion, invasion and metabolism. This ability may have developed during coevolution of L. plantarum and Nm, which commonly colonise the same niche. Further studies are needed but suggest that secretions from L. plantarum can protect against highly invasive and fully encapsulated NmB and prevent its invasion and survival within nasopharyngeal epithelium. The latter could remove the NmB mucosal reservoir and thus help reduce transmission, septicaemia and meningitis.
Acknowledgements
The authors would like to acknowledge the Society for Applied Microbiology for a ‘Student into Work’ grant in which the cell death experiments were optimised by AR; the University of the West of England (UWE, Bristol), Faculty of Health and Life Sciences, for a QR PhD studentship for KP (from RAE funding by the Higher Education Funding Council of England), which enabled the bioluminescent transformation of Nm with support from VS, using a plasmid kindly provided by Ann-Beth Jonsson from the Karolinska Institute, Sweden; and UWE, Bristol for provision of an Early Researcher Starter Grant to enable LT and VD to explore the protective properties of oral Lactobacilli. The authors are grateful to GJ MacArthur from the University of Bristol for useful comments on the manuscript intracellular NmB light output (Figure 6(d)).
Ethical Standards
The experiments within this manuscript comply with the current laws of the country in which they were performed.
NOTES