Computer Aided Design and Performance Analysis of Inverse Fluidized Bed Biofilm Reactors with Special Reference to Bioplastic Synthesis ()
Received 3 March 2016; accepted 12 April 2016; published 15 April 2016

1. Introduction
Inverse fluidization technology is not a new concept, though the application of this technology to the design of bioreactors has been investigated only during the recent past. In the most conventional design, inverse fluidized beds are composed of particles that are of lower density than the liquid (feed solution). Consequently, though the liquid is fed from the top and executes downflow, these particles remain suspended in the descending stream of liquid and thus form a fluidised bed. In the present study, particles made of polymer composites (that are lighter than water) each surrounded by a biofilm (a thin film of microbial solution) are used and these particle-biofilm aggregates remain fluidised in the down-flowing substrate solution (feed solution).
Studies on hydrodynamic characteristics of liquid-solid inverse fluidised beds have been reported by Ulaganathan and Krishnaiah [1] and Banerjee, Basu and Ganguly [2] . Both of these investigators have considered flow of Newtonian fluids (water and aqueous solutions) through inverse fluidised beds composed of polymer beads and granules. Non-Newtonian flow (flow of CMC-carboxy methyl cellulose-solutions) in inverse fluidized beds has been analysed Vijayalakshmi [3] et al. For the estimation of minimum inverse fluidization velocity,
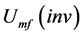 , a modified form of the correlation proposed by Ulaganathan and Krishnaiah [1] has been used in the present study:
, a modified form of the correlation proposed by Ulaganathan and Krishnaiah [1] has been used in the present study:
 (1)
(1)
where,
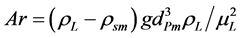 (2)
(2)
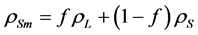 (3)
(3)
 (4)
(4)
 (5)
(5)
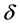 = biofilm thickness (assumed constant).
= biofilm thickness (assumed constant).
In the above equations, 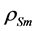 and
and 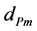 are density and effective size respectively of particle-biofilm aggregates and
are density and effective size respectively of particle-biofilm aggregates and 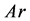 is the modified Archemedes number. The parameter f represents the volume fraction of biofilm in particle-biofilm aggregate. The above correlation is valid for
is the modified Archemedes number. The parameter f represents the volume fraction of biofilm in particle-biofilm aggregate. The above correlation is valid for 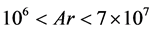 and
and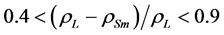 . The operating velocity of the feed solution,
. The operating velocity of the feed solution, 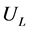 , is then chosen as at least 20% higher than this minimum.
, is then chosen as at least 20% higher than this minimum.
The above authors [1] have also proposed a correlation for the estimation of expanded bed height .
.
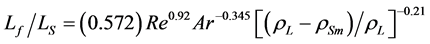 (6)
(6)
where,
![]() (7)
(7)
The voidage of the bed, ![]() (which is equal to the fractional liquid holdup,
(which is equal to the fractional liquid holdup,![]() ) could be then estimated from a solid balance as
) could be then estimated from a solid balance as
![]() (8)
(8)
where ![]() is the voidage of the original static bed (of height,
is the voidage of the original static bed (of height,![]() ).
).
As stated earlier, reported studies on design and analysis of inverse fluidised bed bioreactors are scarce in literature. Performance characteristics of immobilized enzyme inverse fluidized bed bioreactors of modified design that employ nanosilica particles as support media have been analysed by Narayanan [4] et al. The superior performance features of such reactors have been effectively highlighted by these authors.
The specific process considered in the present study is synthesis of lactic acid from molasses and cheese whey. Lactic acid is the starting material for the manufacture of PLLA (Poly Laevo Lactic Acid) which is the most promising bioplastic (green plastic) in the present context. Lactic acid synthesis from pure lactose, glucose or sucrose is exhorbitantly expensive and hence cannot be recommended for the commercial manufacture of PLLA. We have therefore selected molasses and cheese whey as the starting materials which are waste effluents discharged from cane sugar manufacturing industries and milk processing plants and thus are available practically free of cost. Our earlier investigations [5] - [7] have successfully demonstrated that these two are promising raw materials for commercial manufacture of polymer grade lactic acid in fluidised bed biofilm reactors [5] , in semifluidized bed biofilm reactors [6] and in fluidised bed biofilm reactors of diverging-converging geometry [7] . In the present project, design and analysis of inverse fluidised bed bioreactors have been attempted for lactic acid synthesis from molasses and cheese whey permeate (the permeate left behind after the separation of proteins from raw cheese whey by ultrafiltration).
2. Mathematical Modeling
A schematic view of Inverse fluidised bed biofilm reactor is shown in Figure 1. Assuming dispersed flow of substrate solution through the reactor column, the performance equation for the bioreactor is,
![]() (9)
(9)
where, ![]() = operating superficial velocity of feed solution (selected as at least 20% higher than the minimum, as stated earlier)
= operating superficial velocity of feed solution (selected as at least 20% higher than the minimum, as stated earlier)
![]() (10)
(10)
![]() = volumetric flow rate of feed solution employed, m3/s.
= volumetric flow rate of feed solution employed, m3/s.
![]() = axial dispersion coefficient, m2/s.
= axial dispersion coefficient, m2/s.
![]() = effectiveness factor, dimensionless.
= effectiveness factor, dimensionless.
![]() = intrinsic rate of bioconversion of sucrose/lactose, g/(L.s).
= intrinsic rate of bioconversion of sucrose/lactose, g/(L.s).
In the case of sucrose from molasses being fermented to lactic acid using Enterococcus faecalis as biocatalyst or lactose from cheese whey being fermented to lactic acid using Lactobacillus helveticus as biocatalyst, the intrinsic kinetics of bioconversion follows Monod-type kinetic equation. Thus,
![]() (11)
(11)
where,![]() (12)
(12)
![]() = biomass concentration in the biofilm (assumed constant).
= biomass concentration in the biofilm (assumed constant).
![]()
Figure 1. A schematic diagram of inverse fluidized bed bioreactor.
![]() = overall yield coefficient for cell mass production.
= overall yield coefficient for cell mass production.
The values of kinetic constants reported by Anjana and Kumar [8] for lactic acid synthesis from molasses are, ![]() and
and![]() .
.
In case of lactic acid synthesis from cheese whey permeate using a microbial culture of Lactobacillus helveticus, the kinetic constants reported by Schepers [9] et al. are, ![]() ,
, ![]() ,
,![]() .
.
All the above values of kinetic constants have been verified experimentally by us and then utilised in the present analysis.
The parameters ![]() have been assumed to remain more or less unaltered during the entire period of op-
have been assumed to remain more or less unaltered during the entire period of op-
eration of the bioreactor. This, in fact, is a well-justified assumption. Cell growth and multiplication do take place in the biofilm. And so are cell death or cell decay. However, as the biomass concentration exceeds a particular value, the biofilm gets detached from the particle surface (the phenomenon being known as sloughing) and it gets replenished immediately by a new layer of microbial cells. Also, as dead cells fall out from the bio-
film, they are at once replaced by living cells. As a consequence, both the biofilm thickness ![]() as well as the biomass concentration in the biofilm
as well as the biomass concentration in the biofilm ![]() remain more or less constant during the entire operation of the bioreactor.
remain more or less constant during the entire operation of the bioreactor.
It is also to be noted that due to the high magnitude of cell mass concentration in the biofilm (the value of ![]() often exceeds 5 - 10 times the substrate concentration in the feed solution), the rate of bio-conversion attained shall also be substantially large.
often exceeds 5 - 10 times the substrate concentration in the feed solution), the rate of bio-conversion attained shall also be substantially large.
The effectiveness factor ![]() is computed from the correlation proposed by Gottifreddi and Gonzo [10] :
is computed from the correlation proposed by Gottifreddi and Gonzo [10] :
![]() (13)
(13)
where,
![]() (14)
(14)
![]() (15)
(15)
![]() (16)
(16)
![]() (17)
(17)
![]() = effective diffusivity of substrate into biofilm.
= effective diffusivity of substrate into biofilm.
The substrate concentration at the biofilm surface (at the liquid-biofilm interface), namely![]() , depends on the rate of substrate transfer from the liquid bulk to the biofilm surface. However, due to the large degree of turbulence in the fluidized bed, the liquid to particle mass transfer coefficient has been observed to be significantly large. As a result, without any serious error, it could be assumed that substrate concentration (sucrose
, depends on the rate of substrate transfer from the liquid bulk to the biofilm surface. However, due to the large degree of turbulence in the fluidized bed, the liquid to particle mass transfer coefficient has been observed to be significantly large. As a result, without any serious error, it could be assumed that substrate concentration (sucrose
concentration/lactose concentration) at the liquid-biofilm interface ![]() is more or less equal to that in the liquid bulk at any cross-section of the bioreactor
is more or less equal to that in the liquid bulk at any cross-section of the bioreactor![]() . In other words, in the case of the bioreactor under consideration, we may safely assume that:
. In other words, in the case of the bioreactor under consideration, we may safely assume that:
![]() (18)
(18)
Computation of effectiveness factor ![]() from the above equations, Equations (12) to (15), has been performed based on the above assumption. An experimental value of axial dispersion coefficient (
from the above equations, Equations (12) to (15), has been performed based on the above assumption. An experimental value of axial dispersion coefficient (![]() ) is used in computations.
) is used in computations.
Equation (9) is solved numerically using a modified form of Runge-Kutta method starting from the bottom (where z = 0,![]() ). Computations were continued upward until
). Computations were continued upward until ![]() and
and![]() . The fractional conversion of sucrose/lactose is then given by:
. The fractional conversion of sucrose/lactose is then given by:
![]() (19)
(19)
The software package is re-executed at different values of feed flow rate (![]() ) and at different
) and at different ![]() -values. In each case, the fractional substrate conversion (
-values. In each case, the fractional substrate conversion (![]() ) attained and the required height of reactor column (
) attained and the required height of reactor column (![]() ) are computed. Typical results are illustrated graphically in Figures 2-6.
) are computed. Typical results are illustrated graphically in Figures 2-6.
3. Experimental Study
The purpose of experimental work was to test the accuracy and reliability of the software package developed (described above). For this, the numerical values of system/operating parameters (expanded bed height, fractional substrate conversion) computed using the developed CAD software were compared against the experimental data complied on pilot plant scale. Out of 25 data points collected on a pilot plant bioreactor 0.5 m in diameter, 1.5 m height, composed of PP composite beads each surrounded by 0.3 mm biofilm, with clarified molasses as feedstock, 15 of them deviated by 12% (max) from the results (![]() -values) computed from the developed software package and the remaining deviated by less than 10%. With cheese whey as charge stock (cheese whey collected from dairy farms was subjected to ultrafiltration, after preliminary treatments, to separate all the milk proteins and the UF permeate is then used as the feedstock for lactic acid synthesis), the computed values and the experimental values of
-values) computed from the developed software package and the remaining deviated by less than 10%. With cheese whey as charge stock (cheese whey collected from dairy farms was subjected to ultrafiltration, after preliminary treatments, to separate all the milk proteins and the UF permeate is then used as the feedstock for lactic acid synthesis), the computed values and the experimental values of ![]() deviated by 15% (max) and 5% (min). The developed software could be thus treated as reasonably accurate and reliable.
deviated by 15% (max) and 5% (min). The developed software could be thus treated as reasonably accurate and reliable.
The biomass concentration in the biofilm (![]() ) was maintained at 500 g/L during experiments with clarified molasses as the feed solution and a value of
) was maintained at 500 g/L during experiments with clarified molasses as the feed solution and a value of ![]() = 100 g/L is employed when experiments were conducted using cheese whey permeate as the substrate. Concentration measurements were performed using a spectrophotometer (at 600 nm wave length) and HPLC (High pressure Liquid Chromatograph, Agilent Technologies 1200 series, CA, USA). Readings were repeated three to four times at each flow rate to ascertain the experimental accuracy.
= 100 g/L is employed when experiments were conducted using cheese whey permeate as the substrate. Concentration measurements were performed using a spectrophotometer (at 600 nm wave length) and HPLC (High pressure Liquid Chromatograph, Agilent Technologies 1200 series, CA, USA). Readings were repeated three to four times at each flow rate to ascertain the experimental accuracy.
4. Results and Discussion
Two typical plots of expanded bed height versus feed flow rate (![]() ) are shown in Figure 2 and Figure 3. Within the range of feed flow rate handled, the increase in
) are shown in Figure 2 and Figure 3. Within the range of feed flow rate handled, the increase in ![]() with increase in liquid flow rate is not large (
with increase in liquid flow rate is not large (![]() varies within 2.35 to 2.5 m). This is true for all the three particle sizes considered. (
varies within 2.35 to 2.5 m). This is true for all the three particle sizes considered. (![]() = 11 mm, 12 mm, 12.5 mm). The variation, however, is close to linear.
= 11 mm, 12 mm, 12.5 mm). The variation, however, is close to linear.
4.1. Effect of Feed Flow Rate on Reactor Performance
The dependence of fractional conversion of sucrose/lactose attained on feed flow rate (![]() versus
versus![]() ) is illustrated in Figures 4-7. Figure 4 and Figure 5 are for fermentation of molasses with
) is illustrated in Figures 4-7. Figure 4 and Figure 5 are for fermentation of molasses with ![]() = 500 g/L (cell mass concentration in biofilm being maintain constant at 500 g/L) and for different support particle sizes such as
= 500 g/L (cell mass concentration in biofilm being maintain constant at 500 g/L) and for different support particle sizes such as ![]() = 12 mm, 12.5 mm. The biofilm thickness remains the same (
= 12 mm, 12.5 mm. The biofilm thickness remains the same (![]() = 0.3 mm) for particles of all sizes. It can be seen that the bioreactor provides more than 75% conversion of sucrose at feed flow rates exceeding 55,000 L/hr when the particle size (
= 0.3 mm) for particles of all sizes. It can be seen that the bioreactor provides more than 75% conversion of sucrose at feed flow rates exceeding 55,000 L/hr when the particle size (![]() ) employed is 12.5 mm. With 12 mm particles, the conversion attained is slightly lower (though more than 65%) at comparable flow rates. As stated earlier, the expanded height of the bed (effective height of reactor column) is less than 2.5 m, the column diameter being 0.5 m. It can be thus inferred that this bioreactor is capable of providing large degree of bioconversion (large yield of lactic acid) within a low reactor volume. The fractional substrate conversion attained is higher at larger particle size.
) employed is 12.5 mm. With 12 mm particles, the conversion attained is slightly lower (though more than 65%) at comparable flow rates. As stated earlier, the expanded height of the bed (effective height of reactor column) is less than 2.5 m, the column diameter being 0.5 m. It can be thus inferred that this bioreactor is capable of providing large degree of bioconversion (large yield of lactic acid) within a low reactor volume. The fractional substrate conversion attained is higher at larger particle size.
The observation is exactly similar in the case of fermentation of cheese whey permeate as well (Figure 6 and Figure 7). Here also, the fractional conversion of lactose (to lactic acid) attained is more than 75% even at large feed flow rates (more than 56,000 L/hr) when ![]() = 12.5 mm. No doubt,
= 12.5 mm. No doubt, ![]() (attained) is lower at lower particle size (
(attained) is lower at lower particle size (![]() = 12 mm). The cell mass concentration maintained in the biofilm (
= 12 mm). The cell mass concentration maintained in the biofilm (![]() ) in this case is 150 g/L. To note that lactose concentration in the cheese whey permeate (feed solution) is 9.0 g/L, while
) in this case is 150 g/L. To note that lactose concentration in the cheese whey permeate (feed solution) is 9.0 g/L, while ![]() = sucrose concentration in feed molasses = 150 g/L.
= sucrose concentration in feed molasses = 150 g/L.
The magnitude of ![]() (attained) does increase, but sluggishly, with increase in
(attained) does increase, but sluggishly, with increase in![]() , as can be seen from Figures 4-7. This is due to the increase in expanded bed height (
, as can be seen from Figures 4-7. This is due to the increase in expanded bed height (![]() ) and a simultaneous increase in
) and a simultaneous increase in ![]() (bed voidage).
(bed voidage).
4.2. Effect of Cell Mass Concentration in Biofilm on Reactor Performance
Figure 8 effectively demonstrates that a large value of cell mass concentration in biofilm (![]() ) helps in attaining larger degree of substrate (here, sucrose) conversion at any selected feed flow rate (
) helps in attaining larger degree of substrate (here, sucrose) conversion at any selected feed flow rate (![]() ). The situation is analogous with lactose fermentation as well (Figure 9). Though the plots shown are for
). The situation is analogous with lactose fermentation as well (Figure 9). Though the plots shown are for ![]() = 12 mm, the phenomenon has been observed to be identical at all particle sizes considered. This observation is not surprising, since larger the biomass concentration, larger shall be the intrinsic rate of bioconversion,
= 12 mm, the phenomenon has been observed to be identical at all particle sizes considered. This observation is not surprising, since larger the biomass concentration, larger shall be the intrinsic rate of bioconversion, ![]() , as is
, as is
evident from Equations (11) and (12). However, too large biomass concentration in the film could make the biofilm unstable and it shall have pronounced tendency to get detached from the particle surface.
4.3. Operating Cost of Bioreactor
The downflow mode of operation is another exquisite advantage of inverse fluidized bed bioreactors. This brings down the operating cost of the reactor as compared to the conventional fluidized bed/semifluidized bed reactors where the fluid is to be pumped upward. Another advantage due to the downflow mode of operation is that problems related to entrainment of particles or particle-biofilm aggregates in the outgoing product solution are absent in bioreactors of this category.
5. Conclusions
1) Performance characteristics of inverse fluidized bed biofilm reactors are analysed with special reference to synthesis of polymer grade lactic acid from waste effluents such as molasses and cheese whey.
2) A multi-parameter software package has been developed which predicts the performance of the bioreactor satisfactorily. The reliability of the package has been verified by comparing with pilot plant data.
3) This bioreactor provides more than 75% conversion of sucrose/lactose within a reactor height of 2.4 m at large feed flow rates (large capacities) exceeding 56,000 L/hr.
4) The fractional substrate conversion does increase with increase in cell mass concentration in the biofilm, though large values of ![]() are difficult to maintain in industrial bioreactors.
are difficult to maintain in industrial bioreactors.
5) Since the feed solution is not required to be pumped upward and it executes downflow under gravity, the operating cost of this bioreactor is lower than that of fluidized bed/semifluidized bed bioreactors which handle an ascending stream of fluid. This downflow mode of operation also inhibits entrainment loss of particles in the product solution discharged.
6) This bioreactor also permits use of large size support particles and thus larger size particle-biofilm aggregates, though the particle material must be so chosen that its density is sufficiently lower than the substrate solution handled.
6. Future Research Areas
At large liquid velocities, the particles experience large hydrodynamic forces and as a result, these particles could have a tendency to aggregate (or accumulate) at the bottom of the column, just above the bottom porous plate and thereby tend to form a compact packed bed there (similar to what occurs in a semifluidized bed). Such a rearrangement of particles, if occurs, could very much affect the overall performance of the reactor. More elaborate studies are required to throw more light on this feasible phenomenon.
Acknowledgements
We are grateful to all the fellow members of our International Research Group and to a number of consultancy firms of India whose assistances were invaluable for the successful completion of this project.
Nomenclatures
![]() modified Archemedes number.
modified Archemedes number.
![]() substrate concentration in product solution, g/L.
substrate concentration in product solution, g/L.
![]() substrate concentration in feed solution, g/L.
substrate concentration in feed solution, g/L.
![]() diameter of support particle, m.
diameter of support particle, m.
![]() diameter of particle-biofilm aggregate, m.
diameter of particle-biofilm aggregate, m.
![]() effective diffusivity of substrate into biofilm, m2/s.
effective diffusivity of substrate into biofilm, m2/s.
![]() axial dispersion coefficient, m2/s.
axial dispersion coefficient, m2/s.
![]() volume fraction of biofilm in particle-biofilm aggregate, dimensionless.
volume fraction of biofilm in particle-biofilm aggregate, dimensionless.
![]() Monod’s kinetic constant, g/L.
Monod’s kinetic constant, g/L.
![]() height of fluidised bed, m.
height of fluidised bed, m.
![]() characteristic dimension, m.
characteristic dimension, m.
![]() height of original static bed, m.
height of original static bed, m.
![]() substrate flow rate, L/h.
substrate flow rate, L/h.
![]() modified Reynolds number.
modified Reynolds number.
![]() intrinsic rate of bioconversion, g/(L.s).
intrinsic rate of bioconversion, g/(L.s).
![]() superficial velocity of liquid, m/s.
superficial velocity of liquid, m/s.
![]() minimum inverse fluidization velocity, m/s.
minimum inverse fluidization velocity, m/s.
![]() biomass concentration in biofilm, g/L.
biomass concentration in biofilm, g/L.
![]() overall yield coefficient, mg/mg.
overall yield coefficient, mg/mg.
![]() fractional conversion, dimensionless.
fractional conversion, dimensionless.
![]() biofilm thickness, m.
biofilm thickness, m.
![]() total voidage (=liquid holdup) of fluidised bed.
total voidage (=liquid holdup) of fluidised bed.
![]() total voidage (=liquid holdup) of original static bed.
total voidage (=liquid holdup) of original static bed.
![]() effectiveness factor, dimensionless.
effectiveness factor, dimensionless.
![]() maximum specific growth rate, s−1.
maximum specific growth rate, s−1.
![]() viscosity of fluid, kg/m・s.
viscosity of fluid, kg/m・s.
![]() density of fluid, kg/m3.
density of fluid, kg/m3.
![]() density of support particle, kg/m3.
density of support particle, kg/m3.
![]() density of particle-biofilm aggregate, kg/m3.
density of particle-biofilm aggregate, kg/m3.
![]() Thiele-type modulus (defined in Equation (15)), dimensionless.
Thiele-type modulus (defined in Equation (15)), dimensionless.
NOTES
![]()
*Paper presented in the ISHMT-ASTFE International Heat and Mass Transfer Conference held at Trivandrum, India during December 17-20, 2015.