Unsustainable Land-Based Source Pollution in a Climate of Change: A Roadblock to the Conservation and Recovery of Elkhorn Coral Acropora palmata (Lamarck 1816) ()
1. Introduction
Coral reefs are the most diverse and productive marine ecosystems of the world [1] . They constitute heterogeneous habitats that serve as an important source of primary production for multiple species, including fisheries [2] . Reefs also provide protection against strong wave action, protecting coastlines from erosion, constitute an important source of natural products of pharmacological value, and represent a substantial economic base for many countries as the main source of protein and income from multiple recreational activities. Nonetheless, coral reef ecosystems are rapidly declining worldwide due to a combination of multiple anthropogenic factors that include water quality degradation (i.e., sedimentation, turbidity, and pollution), overfishing, diseases, and climate change [3] . Land-Based Source Pollution (LBSP) is one of the most critical concerns for the conservation of coral reefs [4] . It often results from any combination of activities that include mobilization of nitrogen (N) and phosphorus (P) through land clearing, production and applications of fertilizers, sewage pollution, animal production, and combustion of fossil fuels [5] . As a result, surface waters and groundwater throughout developed urban areas often have elevated concentrations of N and P, in comparison to concentrations several decades ago [6] [7] . This has resulted in major eutrophication impacts to coastal waters as elevated dissolved nutrient concentrations stimulate phytoplankton and macroalgal growth, and disrupts the balance between the production and metabolism of organic matter in the coastal zone [4] [8] .
Nutrient enrichment is one of the most significant coral reef stressors, in combination with increased Sea Surface Temperature (SST), increasing human population across coastal habitats, and overfishing [3] [9] [10] . Increasing human pressures lead to increasing non-sustainable construction practices along coastal environments and adjacent steep slopes, increased demand for natural resources, non-sustainable tourism practices, and declining livelihoods of base communities [11] . In the long term, this often results in major LBSP and eutrophication of coastal environments [12] , and in adverse permanent impacts to adjacent coral reef ecosystems [13] [14] . Increasing nutrient concentrations may also be increasing both the prevalence and severity of coral diseases [15] [16] , as well as making corals more susceptible to bleaching as a result of increasing SST trends [17] . Sewage pollution is often associated to eutrophication as two of the most critical synergistic impacts of LBSP. Sewage has been implicated in killing corals [18] -[20] and altering coral reef benthic communities [15] [21] [22] . Evidence also suggests that eutrophication associated to sewage pollution may increase corals susceptibility to bleaching as elevated nutrient availability may increase abundance of susceptible algal symbionts in corals [23] . Eutrophication also results in fostering rapid algal growth and in shifting benthic community structure from Crustose Coralline Algae (CCA) dominance to filamentous turf, and to fleshy macroalgae [4] [24] -[26] . Any combination of eutrophication and declining herbivory may also trigger significant changes in coral reef benthic community structure favoring algal growth [27] . Eutrophication also operates in combination with sedimentation, resulting in further reef decline [28] . Such changes could last across very large temporal scales. Historical changes in land-use patterns have often resulted in long-term degradation and pollution of coral reefs [29] [30] .
Elkhorn coral, Acropora palmata (Lamarck 1816), is a large branching coral with thick and sturdy antler-like branches, and constitutes a mono-specific coral functional group in Caribbean shallow reef front zones as they provide shelter habitat for multiple species (Figure 1) [31] . It is characterized by having fast skeletal growthrates (5 to 9.5 cm∙yr−1), with thick and strong branches, mostly greater than 0.5 m in length, with some colonies even exceeding 2 - 3 m in height and up to 5 - 8 m in diameter which could reach in one to two decades. This coral is usually found in shallow waters (0.5 - 15 m), with high dominance from sea level to about 5 m depth. Acropora palmata is an environmentally sensitive species that requires clear and well circulated water with moderate temperatures (25˚C - 29˚C). Therefore, it is intolerant to prolonged periods of high sedimentation and turbidity stress [32] .
Acroporid coral populations have significantly declined over the past four decades across the Western Atlantic as a consequence of the impact of hurricanes, disease outbreaks, local anthropogenic factors and the impact of climate change [33] . It is estimated that in Puerto Rico A. palmata populations have been reduced by 98% [32] , with major extirpation of populations from most reef localities in Puerto Rico [34] . White-band disease was probably the most important cause of geographic declines [35] . Acropora palmata was eventually listed in May 2006 as a threatened species by the National Marine Fisheries Service (NMFS) under the US Endangered Species Act (ESA), and its Designated Critical Habitat (DCH) was determined by NMFS in November 2008. Recent studies in Puerto Rico have shown that populations have continued to decline across various locations [36] , largely due to local anthropogenic influences linked to changes in land uses [37] [38] and due to the impacts of polluted runoff [22] [39] . Nonetheless, there are still several important remnant population patches along the north coast of Puerto Rico, which have been poorly documented or even remain relatively unknown to science. These include the coastal town of Vega Baja, where there are still remnant patches showing 80% - 90% living tissue cover [40] [41] . Despite this, there are still a number of recurring and cumulative impacts associated to chronic illegal discharges of raw sewage, the construction of a rip-rap structure along the shoreline to reduce beach erosion, and episodic sand dredging along the shoreline which have resulted in a gradual degradation of coral reefs adjacent to the coastal zone [40] -[42] .
This study documents the impact of chronic LBSP on the ecological status of A. palmata dominated coral reefs along a gradient of impacts on the coast of Vega Baja, PR. The null hypothesis of no significant differences in the status of coral reef benthic community structure along a LBSP stress gradient was tested using various multivariate statistical tests to determine whether these factors individually or collectively have any significant impact on coral reefs.
2. Materials and Methods
2.1. Study Sites
Water quality sampling took place along the coastal waters of Vega Baja in the northern coast of Puerto Rico

Figure 1. Example of healthy, high-density Acropora palmata stands at Vega Baja, PR. These reefs still largely unknown to science support outstanding densities of this threatened species.
(Figure 2). Sampling stations were located across a suspected water quality stress gradient downstream from the Cibuco River mouth and downstream of known raw sewage pollution sources across the following sites: Cibuco-CIB (18˚29'05.72"N, 66˚23'04.41"W), Balneario-BAL (18˚29'32.32"N, 66˚23'48.17"W), Malecon-MAL (18˚29'19.82"N, 66˚24'12.31"W), Plazoleta de los Pescadores-PLA (18˚29'20.53"N, 66˚24'19.54"W), Las YolasYOL (18˚29'20.88"N, 66˚24'23.05"W), Playa El Eco-ECO (18˚29'23.19"N, 66˚24'39.31"W), Playa TractoresTRA (18˚29'23.76"N, 66˚24'48.09"W).
The ecological condition of 12 coral reef patches was characterized along a suspected water quality stress gradient in the Vega Baja coast (Figure 3). Reefs were sampled along a distance gradient downstream of a known
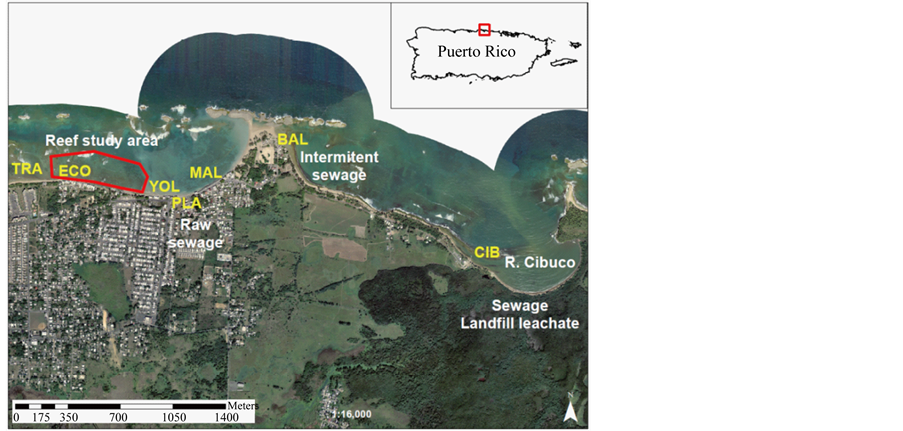
Figure 2. Water quality sampling sites. Known pollution sources are marked at the Cibuco River mouth, at the eastern side of the existing sand tombolo at Balneario Playa Puerto Nuevo—BAL (public beach), and at the storm water runoff outlet at Plazoleta de los Pescadores—PLA. Acronyms described in Methods. Red polygon represents the benthic community sampling range.

Figure 3. Coral reef benthic community sampling design. Eastern inshore: PR-E, PR-W; Eastern offshore: AF-E, AF-W; Mid inshore: PO-E, PO-W; Mid offshore: HI-E, HI-W; West inshore: Eco-In, Eco-Mid; West offshore: Eco-Off-E, Eco-OffW. Acronyms described in Methods.
sewage pollution point source at PLA across three different distance categories: East (<200 m), Middle-MID (200 - 400 m), and West (400 - 700 m). In addition, reefs were subdivided in two segments at increasing distance from the shoreline: inshore (<100 m), and offshore (100 - 250 m). Benthic community sampling sites were subdivided as follows: East-Inshore: Pasarratos-East [PR-E] (18˚29'22.17"N, 66˚24'25.90"W), Pasarratos-West [PR-W] (18˚29'24.35"N, 66˚24'28.44"W), East-Offshore: Afropoint-East [AF-E] (18˚29'29.08"N, 66˚24'30.65"W), Afropoint-West [AF-W] (18˚29'28.96"N, 66˚24'33.41"W); Mid-Inshore: Las Pozas-East [PO-E] (18˚29'24.76"N, 66˚24'34.35"W), Las Pozas-West [PO-W] (18˚29'24.65"N, 66˚24'36.09"W); Mid-Offshore: Hawaii-East [HI-E] (18˚29'29.00"N, 66˚24'44.08"W), Hawaii-West [HI-W] (18˚29'29.05"N, 66˚24'35.37"W); West-Inshore: El EcoInshore [Eco-In] (18˚29'24.07"N, 66˚24'39.41"W), El Eco-Mid [Eco-Mid] (18˚29'25.38"N, 66˚24'38.62"W); West-Offshore: El Eco-Offshore-East [Eco-Off-E] (18˚29'28.02"N, 66˚24'38.00"W), El Eco-Offshore-West [Eco-Off-W] (18˚29'28.00"N, 66˚24'39.33"W).
2.2. Water Quality Sampling Design
Triplicate samples per parameter were obtained at each sampling site during each visit on a nearly monthly basis for eleven months, from July, 2013 to May, 2014. A total of 13 variables were tested, including water temperature, salinity (ppt), conductivity (mS), and dissolved oxygen concentration (mg/L) with a YSI85 digital data logger. The pH and dissolved nutrient concentrations, including phosphates (PO4), nitrates (NO3), nitrites (NO2), ammonium (NH3 and ), and total nitrogen were analyzed with a LaMotte Smart 2 colorimeter. A Turner Designs fluorometer was used to measure in vivo chlorophyll-a concentration (μg/L) and optical brighteners (OABs) concentration (ppm).
), and total nitrogen were analyzed with a LaMotte Smart 2 colorimeter. A Turner Designs fluorometer was used to measure in vivo chlorophyll-a concentration (μg/L) and optical brighteners (OABs) concentration (ppm).
2.3. Benthic Community Sampling Design
Coral reef benthic community structure was characterized across twelve individual coral reef patches located along an increasing east-west distance, and along an inshore-offshore distance gradient from a known pollution source at PLA (Figure 3). Data were collected via 6 replicate haphazard 10 m-long transects, and 5 replicate 0.75 × 0.50 m (0.375 m2) high-resolution, non-overlapping images from each transect at two replicate reefs per distance (east, mid, west) and per segment (inshore, offshore). A total of 35 regularly-spaced points were digitally projected over each image to determine %cover of all benthic components (i.e., corals, sponges, zoanthids, algal functional groups, cyanobacteria, open reef substrates), species richness (S), coral species diversity index (H’c) [43] , evenness (J’c) [44] , and the variation in taxonomic distinctness (sΔ+) [45] .
2.4. Statistical Analyses
Water quality data was analyzed using a suite of multivariate statistical methods in the statistical package PRIMER v6.1.16 + PERMANOVA v1.0.6 (Plymouth Marine Lab, UK) [45] [46] . The null hypothesis of no significant difference in water quality condition among sampling sites was tested using a 3-way permutational analysis of variance (PERMANOVA), with site (n = 7), date (n = 9), and season (n = 4) as main variables. Also site × date, site × season, date × season, and site × date × season interactions were tested. Spatio-temporal patterns were illustrated following a Bray-Curtis ordination cluster analysis [47] [48] using Euclidean distance and Multi-Dimensional Scaling (MDS) ordination [45] . Principal Component Ordination (PCO) was also used to identify spatio-temporal patterns of water quality trends and indicator parameters of spatio-temporal clustering patterns [46] . Data were normalized prior to analysis. A total of 10,000 permutations were used for multivariate tests.
The null hypothesis of no significant difference in coral reef benthic community structure among surveyed sites was also tested using a 3-way PERMANOVA, with distance (n = 3), segment (n = 2), and site (n = 12) as main variables. Also distance × segment, distance × site, segment × site, and distance × segment × site interactions were tested. A similar approach was used to test for S, H’c, J’c, and sΔ+. Spatio-temporal patterns were illustrated following a Bray-Curtis ordination cluster analysis and MDS ordination. PCO was used to identify spatial patterns of coral reef benthic community structure clustering and indicator taxa of such clustering patterns. A combination of linear and non-linear regression models were fit using Sigma Plot v11.0 (Systat Software, Inc.) to test the relationship of individual coral reef taxa and increasing linear distance from the pollution source. In addition, a Spearman rank correlation was carried out between individual coral reef benthic components and individual water quality parameters to test for any significant relationship.
3. Results
3.1. LBSP Stress Gradient on Coastal Waters
Sea Surface Temperature (SST) was significantly lower (p < 0.0001) at PLA due to the discharges of freshwater from an underground storm sewer pipe directly to the beach (Figure 4). Otherwise, SST and its mean fluctuation were statistically similar among all other sites. Mean pH was also significantly lower (p < 0.0001) at PLA, and just slightly lower at CIB and BAL, due to freshwater influences either from river outflow or from polluted discharges. Salinity and conductivity were also significantly lower (p < 0.0001) at PLA due to continuous freshwater discharges. CIB and BAL showed significantly lower salinity due to influence from the Cibuco River mouth and sporadic illegal raw sewage discharges, respectively. Although dissolved oxygen concentration declined at PLA to about 3 mg/L, and to about 4 - 5 mg/L at CIB, BAL, and YOL, difference among sites was not significant (Figure 5). Water turbidity was significantly higher at CIB in comparison to other locations due to the continuous direct influences of the Cibuco River mouth. Also, chlorophyll-a concentration was significantly higher at PLA with approximately 25 μg/L, followed by BAL with about 9 μg/L, and by CIB with 5 μg/L. These are sites known for receiving continuous raw sewage-polluted discharges. Optical brighteners (OABs) concentration was significantly higher at CIB and PLA with approximately 200 ppm, followed by BAL with about 90 ppm. These sites showed major influences by LBSP.
Phosphate (PO4) concentration was significantly higher at PLA, with up to 15 μM, followed by BAL with 12 μM, and YOL, with nearly 10 μM (Figure 6). These sampling stations received continuous or intermittent raw sewage discharges. YOL was located about 100 m downstream of PLA. Nitrate (NO3) concentration was significantly higher (p = 0.0051) at TRA, with a mean value slightly below 30 μM, followed by ECO, with about 22 μM, and by CIB, with about 20 μM. Locations with higher NO3 concentrations were located downstream of PLA and adjacent to multiple houses and an apartment complex with either septic tanks adjacent to the shoreline or malfunctioning sewer pipes right within the phreatic level within a karstic, highly permeable soil. These could represent a significant source of nitrogen to adjacent coastal waters, particularly during low tides which produce seepage of coastal phreatic freshwater to adjacent coastal waters. Nitrite (NO2) concentration was just slightly higher at YOL, ECO, and TRA, but trends were not significant.
Ammonium (NH3) concentration was just short of significance (p = 0.0568) with slightly higher values at ECO and TRA, with 120 - 125 μm. However, ionized ammonium ( ) concentration was significantly higher (p = 0.0009) at these two sites, probably as a result of polluted groundwater seepage. Total nitrogen concentration was also short of significance (p = 0.0633).
) concentration was significantly higher (p = 0.0009) at these two sites, probably as a result of polluted groundwater seepage. Total nitrogen concentration was also short of significance (p = 0.0633).
These results suggest that nitrogen concentration seems to be significantly influenced by the trophic condition of groundwater and by groundwater seepage dynamics.

Figure 4. Water quality parameters: (a) Temperature; (b) pH; (c) Salinity; and (d) Conductivity. Mean ± 95% confidence intervals.

Figure 5. Water quality parameters: (a) Dissolved oxygen concentration; (b) Turbidity; (c) Chlorophyll-a concentration; and (d) Optical brighteners (OABs) concentration. Mean ± 95% confidence intervals.

Figure 6. Dissolved nutrients concentration: (a) Phosphate (PO4); (b) Nitrate (NO3); (c) Nitrite (NO2); (d) Ammonium (NH3); (e) Ionized ammonium ( ); and (f) Total nitrogen (N). Mean ± 95% confidence intervals.
); and (f) Total nitrogen (N). Mean ± 95% confidence intervals.
3.2. Water Quality Decline Association to Raw Sewage
A Principal Component Ordination (PCO) analysis using the entire matrix of water quality parameters showed a strong spatio-temporal water quality gradient that resulted in a significant clustering of sampling sites PLA and BAL which was largely explained by high chlorophyll-a concentration, low conductivity, and in a lesser extent by high PO4 and OABs concentration (Figure 7). PLA received a continuous mixture of illegal raw sewage discharges and storm waters, while BAL received intermittent raw sewage discharges during the early months of the study in 2013. There were also two minor clusters composed by CIB explained by high turbidity and in a lesser extent by low dissolved oxygen concentration. The rest of the sampling sites, particularly those adjacent to houses along the shoreline were characterized by higher NH3,  , and lower salinity and pH. The PCO analysis explained approximately 55% of the spatio-temporal variation in water quality parameters and confirmed the existence, at least during prolonged periods of time, of a strong water quality gradient along the Vega Baja coast which is influenced by: 1) highly turbid outflow from the Cibuco River; 2) intermittent raw sewage discharges at BAL; 3) continuous raw sewage discharges mixed with storm water at PLA; and 4) groundwater seepage with potential raw sewage infiltration from malfunctioning septic tanks and sewer pipes along the coastline, particularly during low tides. Littoral currents tend to carry pollutants downstream to the west along existing coral reefs, affecting critical reef resources. A three-way permutational analysis of variance (PERMANOVA) showed highly significant differences (p < 0.0001) among sites, dates, season, and all possible interactions (Table 1).
, and lower salinity and pH. The PCO analysis explained approximately 55% of the spatio-temporal variation in water quality parameters and confirmed the existence, at least during prolonged periods of time, of a strong water quality gradient along the Vega Baja coast which is influenced by: 1) highly turbid outflow from the Cibuco River; 2) intermittent raw sewage discharges at BAL; 3) continuous raw sewage discharges mixed with storm water at PLA; and 4) groundwater seepage with potential raw sewage infiltration from malfunctioning septic tanks and sewer pipes along the coastline, particularly during low tides. Littoral currents tend to carry pollutants downstream to the west along existing coral reefs, affecting critical reef resources. A three-way permutational analysis of variance (PERMANOVA) showed highly significant differences (p < 0.0001) among sites, dates, season, and all possible interactions (Table 1).

Figure 7. Principal Component Ordination (PCO) of spatio-temporal variation in LBSP across coastal waters.

Table 1. Three-way permutational analysis of variance (PERMANOVA) of overall water quality parameters.
*Degrees of freedom.
3.3. Spatial Patterns in Benthic Categories
Coral reef benthic communities along the known LBSP stress gradient were showing unequivocal signs of decline, but not all coral reef biological parameters were impacted in a similar fashion. Mean species richness ranged from about 3 to 6 species/transect, depending on the distance from the land-based pollution source and on dominance by Acropora palmata (Figure 8). Mean H’c ranged from about 0.75 to 1.50, and J’c from about 0.5 to nearly 0.9. But none of these parameters were significantly different among all distance (east, mid, west) or segment (inshore, offshore) combinations. Nonetheless, variation in taxonomic distinctness (sΔ+) was significantly different among sites, with a consistent declining trend with increasing distance from LBSP. This suggests that recurrent disturbance by water quality stressors is driving coral assemblages to increased states of disturbance and increased variability in phylogenetic diversity due to difference in tolerance to environmental stress by different coral species.
Percent living coral cover showed a highly significant increase with increasing distance from LBSP, from values of about 18% in eastern inshore reefs to about 93% across western offshore reef segments (Figure 9). Also, dominance by A. palmata increased with increasing distance from LBSP, with values of <1% in eastern inshore reefs to about 80% across mid and western offshore reef segments. However, Brain coral, Pseudodiploria clivosa, showed a contrasting and significant dominance in reefs adjacent to pollution sources, with up to 14% in eastern inshore reefs, in comparison to <1% in western offshore reef segments. This suggest that under a lack of interference competition by A. palmata shading P. clivosa can become a significant opportunist and dominate open reef substrates under high wave energy conditions. Pseudodiploria strigosa followed a similar but non-significant trend.
Green, red and brown macroalgae spatial distribution also followed a non-significant trend of increasing cover in close proximity to LBSP probably as a result of large variability and patchiness across the reef system (Figure 10). A larger sample size or an increased spatial resolution might have better illustrated their spatial trends. But percent cover of filamentous algal turfs was significantly higher adjacent to LBSP, with values reaching up to 60%, in comparison to values below 10% in A. palmata-dominated reef zones. Percent CCA cover showed very low percent cover (<0.5%), although higher values were observed under areas exposed to higher wave energy (2.5%). But the pattern was also non-significant. Percent cover of bare substrates composed of Sand, Pavement and/or Rubble (SPR) was also significantly higher adjacent to LBSP. Local inshore reef segments

Figure 8. Coral reef benthic community parameters across LBSP stress gradient: (a) Species richness (S); (b) Species diversity index (H’c); (c) Evenness (J’c); and (d) Variation in taxonomic distinctness (sΔ+). Mean ± 95% confidence intervals.

Figure 9. Coral reef benthic community parameters across LBSP stress gradient: (a) Percent coral cover (S); (b) Percent Acropora palmata cover; (c) Percent Pseudodiploria clivosa cover; and (d) Percent P. strigosa cover. Mean ± 95% confidence intervals.

Figure 10. Coral reef benthic community parameters across LBSP stress gradient: (a) Percent green magraoalgal cover (S); (b) Percent red macroalgal cover; (c) Percent brown macroalgal cover; (d) Percent filamentous algal turf cover; (e) Percent Crustose Coralline Algal (CCA) cover; and (f) Percent cover of Sand, Pavement and Rubble (SPR). Mean ± 95% confidence intervals.
are also known to be impacted by significant sediment bedload due to east-west littoral drift and significant shoreline erosion.
3.4. A Gradient of Coral Reef Ecological Conditions
The spatial gradient of reef conditions was evident from regression analyses. There was a highly significant (p = 0.0022) non-linear regression between coral species richness and linear distance from LBSP (Figure 11(a)). There was also a highly significant (p = 0.0057) non-linear regression between sΔ+ and linear distance from LBSP (Figure 11(b)). In addition, there was a highly significant (p = 0.0098) linear regression between percent living coral cover and linear distance from LBSP (Figure 12(a)), and between percent Acropora palmata cover and linear distance from LBSP (p = 0.0137) (Figure 12(b)). PCO analysis also showed that coral reef benthic community structure clustered in four different spatial patterns (Figure 13). The reef most adjacent to LBSP source

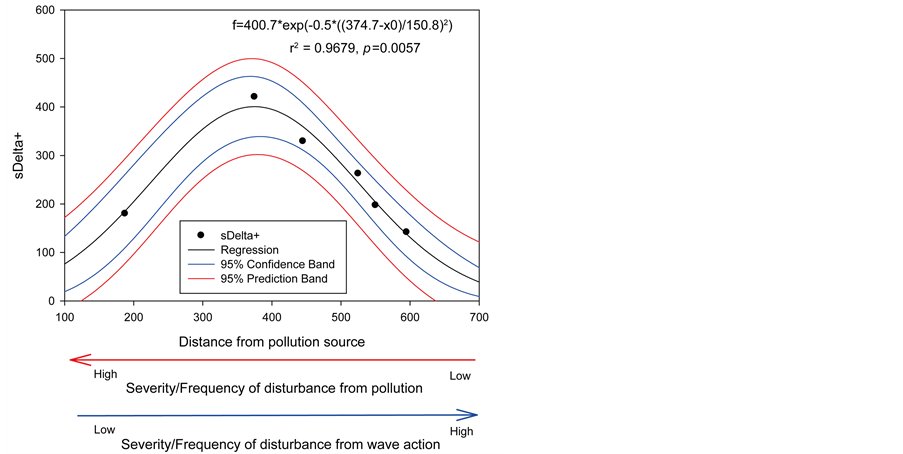
Figure 11. Non-linear correlation between coral parameters and distance from pollution source: (a) Species richness (S); and (b) Variation in taxonomic distinctness (sΔ+).


Figure 12. Linear correlation between coral parameters and distance from pollution source: (a) Percent coral cover; and (b) Percent Acropora palmata cover.
(PR-E) was dominated by a sand veneer across the reef surface and brown macroalgae. There was a second cluster of three eastern reefs which were dominated by filamentous algal turfs and opportunistic Pseudodiploria clivosa. A third cluster was composed by a combination of mid and western reefs with moderate to high dominance by Acropora palmata, and a final cluster of mid and western offshore reefs almost exclusively dominated by A. palmata. This PCO analysis showed a solid resolution (80.6%) of the observed spatial variation in benthic community structure, which also resulted statistically significant (Table 2).
There was also a highly significant correlation between percent cover of A. palmata and water quality parameters such as salinity, and the concentration of dissolved oxygen, OABs, PO4,  , NO3, NO2, and total N (Table 3). Acropora palmata distribution was marginally correlated with conductivity. Pseudodiploria clivosa distribution was highly correlated with salinity, and the concentrations of OABs,
, NO3, NO2, and total N (Table 3). Acropora palmata distribution was marginally correlated with conductivity. Pseudodiploria clivosa distribution was highly correlated with salinity, and the concentrations of OABs,  , NO3 and NO2. Green macroalgae was significantly correlated salinity, and the concentrations of OABs,
, NO3 and NO2. Green macroalgae was significantly correlated salinity, and the concentrations of OABs,  , NO3, NO2, and total N.
, NO3, NO2, and total N.

Figure 13. Principal Component Ordination (PCO) analysis of spatial variation of coral reef benthic community structure.

Table 2. Three-way permutational analysis of variance (PERMANOVA) of spatial patterns of variation in benthic community structure.
*Degrees of freedom.

Table 3. Significant and marginally significant correlations (Pearson) of selected benthic community parameters and water quality parameters.
There was also a significant correlation between the distribution of filamentous algal turf and salinity, conductivity, and the concentration of dissolved oxygen, OABs,  , NO3 and NO2. No other significant correlations were documented among other parameters. These results clearly suggest that chronic coastal water quality degradation due to LBSP is significantly impacting coral reef benthic community structure, which in the long term could have paramount adverse ecological implications for the conservation and sustainability of threatened Acropora palmata populations across the zone.
, NO3 and NO2. No other significant correlations were documented among other parameters. These results clearly suggest that chronic coastal water quality degradation due to LBSP is significantly impacting coral reef benthic community structure, which in the long term could have paramount adverse ecological implications for the conservation and sustainability of threatened Acropora palmata populations across the zone.
4. Discussion
4.1. Hypertrophic Pulses and Raw Sewage Pollution in Coral Reefs
Coral reefs have significantly declined across an east-west LBSP gradient. Coral species richness and several species diversity indices have declined in proximity to LBSP, are high across moderately disturbed areas, and decline again at remote reef segments far from LBSP disturbance due to dominance by Acropora palmata. This pattern followed the classical intermediate disturbance hypothesis [40] . A particular concern is the rapid decline in remnant populations of A. palmata. Vega Baja coral reef communities have been unequivocally and severely impacted by a combination of illegal point and non‐point sources of raw sewage pollution, by recurrent sewage‐polluted, nutrient‐loaded, sediment‐laden, turbid storm water runoff pulses, and most probably by organic-polluted groundwater seepage along coastal properties. Also, sediment bedload has been a key concern further enhancing long-term negative impacts of chronic water quality decline. Chronic coastal water pollution has produced significant recurrent coral physiological stress pulses that have resulted in extensive coral colony mortalities and major phase shifts in benthic community structure often favoring non-reef building taxa. Recurrent highly turbid and polluted pulses from illegal discharges of raw sewage to adjacent Playa Puerto Nuevo public beach and through the Cibuco River discharge have been previously documented to precede coral mortality events [41] -[43] .
Local non-point source sewage pollution sources documented in this study across the Playa Puerto Nuevo area were numerous and included raw sewage spills from sewer pipes that illegally empty at the beach, illegal connections from private properties to storm water sewers, broken sewage pipes by corrosion or beach erosion from a road along the shoreline, overloaded and leaking septic tanks from properties at nearly sea level, and illegal dumping of sewage from septic tank cleaning trucks [41] . Leaking septic tanks have been identified as key non-point sources of sewage pollution impacting adjacent coral reef systems in multiple studies [8] [21] [37] [38] . The Vega Baja municipal landfill is also located in the eastern bank of the Cibuco River alluvial plain, further polluting the river estuary through chronic leachate infiltration through groundwater and to the river over an intermittent creek. Nutrient-enriched groundwater can have elevated PO4 (up to 2.75 mM) and NO3 + NO2 concentrations (up to 2.89 mM) [6] . The overloaded Vega Baja sewage treatment plant effluents empty at Caño Cabo Caribe, which in turn empties to the east at Cibuco River, also contributing significant amounts of sewage and nutrients to coastal waters. Caño Cabo Caribe also connects to the west to an extensive system of wetlands that includes Laguna Tortuguero Natural Reserve. Data not included in this study previously showed mean chlorophyll-a concentration of up to 35 - 60 μg/L and OABs concentration of up to 423 - 1350 ppm at Caño Cabo Caribe creek within less than 100 m off the sewage plant outfall (Hernández-Delgado, unpubished data). Also, PO4 concentration reached up to 60 μM, NO3 up to 178 μM, NO2 up to 22 μM, NH3 up to 80 μM,  up to 79 μM, and total nitrogen up to 281 μM.
up to 79 μM, and total nitrogen up to 281 μM.
A key concern associated to sewage and eutrophication impacts was the elevated mean chlorophyll-a concentration range found in this study on sites known to be polluted (5 - 25 μg/L), which represented up to 17 - 83 times higher than the recommended concentration for coral reef waters (0.3 - 0.5 μg/L) [12] [49] . Mean chlorophyll-a concentration was up to 3 - 10 times higher than the recommended concentration even in areas farther downstream from LBSP. Chlorphyll-a concentration at Caño Cabo Caribe creek was 117 - 200 times higher than recommended values for its receiving waters with adjacent coral reefs. Also, nutrient concentrations were dangerously high for the survival and persistence of coral reef ecosystems. Previous studies have suggested that PO4 concentration on coral reef habitats should not exceed 0.3 μM [12] , and that any concentration above 1.5 μM would be too high for coral reefs [50] . Our study showed mean PO4 concentration to range between 2 and 15 μM, which resulted 7 - 50 times above the recommended concentration for healthy coral reefs. PO4 concentration at Caño Cabo Caribe creek was 200 times higher than the recommended value for adjacent coral reefs. Similarly,  concentrations above 24 μM were deemed as too high, when the recommended concentration for coral reefs should not exceed 0.1 μM [12] . We obtained mean
concentrations above 24 μM were deemed as too high, when the recommended concentration for coral reefs should not exceed 0.1 μM [12] . We obtained mean  concentrations that ranged from 60 to 124 μM, which was 600 - 1240 times higher than the recommended acceptable level.
concentrations that ranged from 60 to 124 μM, which was 600 - 1240 times higher than the recommended acceptable level.  concentration at Caño Cabo Caribe creek was 263 times higher than the recommend concentration for adjacent coral reefs. Further, the recommended acceptable ratio of total dissolved nitrogen to PO4 is 7:1 for coral reefs [12] . But ratios across coastal waters ranged from 17:1 to 70:1, which exceeded by far the recommended ratio for coral reefs. In addition, mean dissolved oxygen concentration in this study ranged from 2.3 to 6 mg/L, when any concentration below 2.5 mg/l is considered hypoxic [50] . Mean turbidity in waters adjacent to surveyed reefs ranged from 2 to 5 NTU, but in waters adjacent to Cibuco River mouth reached a mean of 13 NTU, when recommended values should be around 0.5 NTU [12] . This was 4 - 10 times higher than the recommended value. This was unequivocal evidence that surface water, storm water runoff and groundwater that empty to the Vega Baja coastal waters were severely polluted by raw sewage from a malfunctioning treatment facility and from multiple non-point sources, producing recurrent hypertrophic pulses that moved across local coral reefs with drifting currents.
concentration at Caño Cabo Caribe creek was 263 times higher than the recommend concentration for adjacent coral reefs. Further, the recommended acceptable ratio of total dissolved nitrogen to PO4 is 7:1 for coral reefs [12] . But ratios across coastal waters ranged from 17:1 to 70:1, which exceeded by far the recommended ratio for coral reefs. In addition, mean dissolved oxygen concentration in this study ranged from 2.3 to 6 mg/L, when any concentration below 2.5 mg/l is considered hypoxic [50] . Mean turbidity in waters adjacent to surveyed reefs ranged from 2 to 5 NTU, but in waters adjacent to Cibuco River mouth reached a mean of 13 NTU, when recommended values should be around 0.5 NTU [12] . This was 4 - 10 times higher than the recommended value. This was unequivocal evidence that surface water, storm water runoff and groundwater that empty to the Vega Baja coastal waters were severely polluted by raw sewage from a malfunctioning treatment facility and from multiple non-point sources, producing recurrent hypertrophic pulses that moved across local coral reefs with drifting currents.
4.2. A Shift in the Frequency and Severity of LBSP Disturbances
Poor water quality pulses are often discrete events in time and space that can become chronic and/or widespread depending on their frequency and severity. In the particular case of BAL in this study, pulse impacts were dramatic as a raw sewage pipe was deliberately opened to free local sewers from Playa Puerto Nuevo community from obstructions and spillovers, but impacts were discrete in time and so were coral mortality pulse events. In the particular case of PLA, raw sewage was chronically mixed with storm waters, emptied just east of the Acropora palmata reef system (Figure 3), and was carried out through the reef by prevailing east-west littoral drift. Sewage pollution across coastal waters is not only confined to the immediate vicinity of receiving waters and has been shown to potentially extend with tidal and wind-driven currents through large geographic areas across the shelf [21] , negatively impacting extensive coral reef communities [22] [42] [43] . Sewage impacts are often associated to eutrophication and turbidity [4] [12] [19] [50] , to altered coral-associated microbial community composition [51] , to an increased prevalence of Black Band Disease and White Plague-Type II in coral colonies [16] , and to an increased prevalence of White Pox in A. palmate [20] . It has also been associated to declining coral survival rates [18] [19] , skeletal extension rates [52] , to declining coral reproduction [53] , and larval settlement rates [54] .
Sewage impacts often result in a combination of systemand species-specific responses, as well as cascading direct and indirect effects that could result in major long-term phase shifts in benthic community structure, favoring dominance by fleshy macroalgae and non-reef building taxa [22] [42] . Such phase shifts could be irreversible in long-term scales [55] . Sewage-associated eutrophication impacts can also result in an accelerated reef decline often due to a combination of synergistic impacts, mostly from sediments and turbidity [4] [26] [28] . This combination of factors and their rapid shift from pulse to chronic events may explain why some local coral reefs at Vega Baja showed such a rapid phase shift.
4.3. Turbidity and Sediment Bedload
A nearly continuous strong east-west LBSP gradient, in combination with high turbidity and sediment bedload smothering corals, has also been documented in the past to affect coral reefs adjacent to the shoreline, with extreme pulse turbidity events reaching local reefs with values as high as 125 NTU [41] [42] . Sediment bedload stress has been shown also to kill corals [43] along the Vega Baja coast due to continuous wave backwash and sediment resuspension along the existing rip-rap area just east of inshore coral reefs. Sediment deposition might be minimal on high-energy habitats such as Vega Baja’s reefs. But sediment bedload abrasive effects can be substantial and deleterious to corals, and should be thoroughly studied in the future. There is also a particular concern from potential pollutant resuspension from sediments under such conditions. Sediments can have a long-term influence on the structure of coral reef benthic communities [56] , may smother reef organisms [57] , and in combination with eutrophication, may reduce light available for photosynthesis [58] . But sedimentation effects can be highly variable depending on the taxa [59] , on the local oceanographic conditions, topographic complexity, environmental history of coral reefs, on the spatial and temporal scales of impacts, and on impacts by other stressors. So is the ability of coral reefs to survive and recover from combined and synergic sediment, turbidity, eutrophication, and sewage pollution stress. Deforestation, poor land use patterns, coastal development, engineering projects, dredging, construction of rip-raps, beach renourishment and other sort of anthropogenic activities can further trigger a widespread increase in the frequency, duration and/or severity of stressful events for corals. They may further result in a reduced time span for coral reefs to recover from recurrent disturbances, therefore resulting in a net long-term phase shift in community structure favoring non reef-building taxa.
4.4. Unsustainable Decline of a Coupled Social-Ecological System in a Climate of Change
A final concern is the role of chronic LBSP on potentially magnifying the severity of climate change-related impacts over already stressed out coral reef resources. Coral reefs should be viewed as a coupled social-ecological system in the context that multiple human activities and social structures can profoundly influence the dynamics of factors such as human population growth, environmental conditions, and natural resource availability, distribution, and condition. Climate change forecasts suggest increased SST and recurrence of massive coral bleaching events across the wider Caribbean region [60] , which in combination with increasing atmospheric carbon dioxide (CO2) concentration [61] may result in major irreversible changes in coral reef assemblages at global scales in the near future. There is evidence that massive coral bleaching has already had paramount negative and permanent impacts on coral reefs across the Wider Caribbean [62] -[65] . Modeling efforts have further suggested increasing coral reef decline across the Wider Caribbean with increasing SST and CO2 trends, resulting in irreversible losses of major reef-building taxa [66] and in widespread socio-economic losses [67] . We argue that poor governance often results in poor land use practices, which in turn often results in increased chronic LBSP impacts. The environmental condition of coastal waters in Vega Baja, Puerto Rico, is biologically unsustainable for coral reefs, and may result in the long-term decline of the coral reef coupled social-ecological system. It may represent an increased exposure to human-driven stress, an increased sensitivity to disturbance, a potentially reduced adaptive capacity to climate change, and an increased state of vulnerability of both ecosystems and local human populations.
Chronic LBSP leads to increased exposure of coral reefs to cumulative and synergistic stress. This could be affected by the severity, frequency, duration and spatial extent of pulse (discrete), cumulative and synergistic events. Such combined impacts may lead to multiple species-specific threshold responses that still remain largely unknown for most taxa, and may lead to increased sensitivity to environmentalor climate-related stress. Sensitivity could be described as the degree to which any stress level may modify a response of populations to ecosystems. A key example could be the combination of SST above the mean monthly maximum (annual highest SST) for any given locality and the chronic exposure to sediment-laden, nutrient-loaded, sewage-polluted runoff. Higher SST may lead to altered benthic microbial communities, but the combination of high SST + high dissolved nutrient loads may lead to potentially stronger alterations, which may lead to increased states of sensitivity, and eventually to a magnified state of vulnerability.
Under predicted escalating climate change impacts the narrow window still available to protect threatened coral reef resources, such as Elkhorn coral, Acropora palmata, is rapidly being closed by human negligence. Natural resource management has become a major challenge in degraded ecosystems due to the increasing vulnerability to climate change, combined with declining ecological conditions of key resources, and weak adaptive capacity and governance. Losing A. palmata may predispose Caribbean reefs to a dangerous and potentially irreversible loss of resilience in a human time scale [68] . There is a need for immediate novel unprecedented management actions, probably across spatial and temporal scales never thought before [69] , in order to burrow some extra time to prevent coral reef ecosystems from collapsing [70] . But weak governance and neglecting impacts by human stressors, including climate change, probably constitute the largest roadblock to the conservation and rehabilitation of coral reef ecosystems.
5. Conclusions
Chronic LBSP has resulted in a significant decline of extensive stands of threatened Elkhorn coral, A. palmata. Sporadic pulse impacts of nutrient-loaded, turbid and polluted runoff documented in the past [41] -[43] have become chronic and unsustainable, largely affecting adjacent reef ecosystems. Remote reefs exposed to stronger wave action and surface circulation still support more prolific coral growth, but in isolated patches. Even large bottom extensions across remote reef segments which have large amounts of hurricaneor winter swell-generated A. palmata fragments are also showing significant reattachment, tissue regeneration and regrowth. But benthic communities adjacent to known pollution sources support a depauperate coral community, with nearly absence of Acropora palmata, and dominance by macroalgae and filamentous algal turf. Adverse conditions were predominant through areas under the strong influence of a LBSP gradient characterized by high nutrient, chlorophyll-a, and OABs concentrations, and by raw sewage pollution. These conditions are biologically unsustainable for coral reefs. This situation has received little attention from Commonwealth and US Federal regulatory agencies to prevent or minimize further damage to these unique resources. It further points out at the paramount importance of implementing specific water quality legal limits for coral reef waters for chlorophyll-a (0.3 μg/L), PO4 (0.3 μM),  (0.1 μM), and a ratio of PO4:Total N of 7:1 or lower.
(0.1 μM), and a ratio of PO4:Total N of 7:1 or lower.
The most critical corrective management action identified in this study was the imperative need to eliminate illegal raw sewage dumping from septic tank effluents, malfunctioning sewers, and other non-point sources at nearby Puerto Nuevo Public Beach by improving sewer systems and upgrading existing sewage treatment plant facilities. A first step was already taken with the expansion of the sewage pipes system to connect a large portion of the community. Nonetheless, there is still a need to prepare an inventory of septic tanks, their condition, and their potential leakage to saline groundwater on properties located along the shoreline. There is also a need to eliminate illegal connections of private houses to storm sewers and other illegal raw sewage discharges directly to coastal waters. There is a need to improve the implementation of erosion-sedimentation controls on any construction activity across adjacent watersheds or coastal waters. A moratorium in the implementation of existing plans to expand the adjacent rip-rap as a method to control beach erosion is also strongly recommended. Instead, the alternative of using semi-artificial reef technology to reduce or modify wave action and littoral drift should be thoroughly evaluated and implemented to reduce beach erosion, but also to reduce further impacts of turbidity and sediment bedload to adjacent coral reefs.
There is also a need to implement a permanent long-term ecological monitoring program for A. palmata, in combination with a standard water quality monitoring program. Plans are also already underway to expand the existing Community-Based High-Energy Reef Rehabilitation Program led by NGOs Vegabajeños Impulsando Desarrollo Ambiental Sustentable (VIDAS) and Sociedad Ambiente Marino (SAM), in collaboration with the Coral Reef Research Group of the Center for Applied Tropical Ecology and Conservation (CATEC) at the University of Puerto Rico, aimed at propagating A. palmata and reintroducing the species at nearby depleted reefs.
Also, the community-based plan to designate the Vega Baja-Manatí Submarine Gardens Natural Reserve was already submitted for final evaluation by the PPRDNER as a critical strategic tool to manage conflicting uses along the coastal areas of concern that still support significant natural resources, including A. palmata. This should provide an unprecedented opportunity to improve community-based participation in decision-making processes regarding management of the proposed reserve. However, there are critical issues that still need to be addressed, including the implementation of effective pollution controls of storm water runoff from Vega Baja downtown urban areas and from the adjacent landfill. There is also a need to study and model complex local ocean circulation dynamics in order to understand pollutants movement, as well as the connectivity between terrestrial and marine systems. In addition, a strong and continuous community outreach and educational program still needs to be implemented to improve local support for the proposed natural reserve designation, but also as a tool to educate local communities and decision makers regarding the critical importance of the conservation and rehabilitation of local coral reef ecosystems. This could become a major behavior-modifying and community-based engaging tool to foster improved participation through a co-management model.
In the context of forecasted increasing impacts from sea-surface warming trends and oceanic acidification associated to climate change, there is no time to waste to implement immediate actions to protect A. palmata stands and rehabilitate coral reef resilience, ecological functions, benefits and services. Failing to implement rapid and effective solutions may represent an irreparable loss of a significant population of an already severely depleted species through the entire Atlantic. Chronic unsustainable LBSP and lack of governance constitute a major roadblock to the conservation and rehabilitation of coral reefs in face of current and forecasted climate change impacts. The combination of climate-related effects and uncontrolled local anthropogenic stressors would be devastating for these coral reefs and may result in the unprecedented permanent loss of the first line of defense against storm swells and sea-level rise. The price of no action would be too high for a small island with limited economic resources. The conservation and rehabilitation of declining coral reef resources must be one of the top critical priorities for implementing any climate change mitigation and adaptation plan, but roadblocks to conservation need to be eliminated first.
Acknowledgements
This work was possible thanks to funding provided by the National Science Foundation (HRD #0734826) through the Center for Applied Tropical Ecology and Conservation (CATEC), and the University of Puerto Rico’s Central Administration to E.A. Hernández-Delgado. Our appreciation for the logistical field support provided by Ricardo Laureano (Vegabajeños Impulsando Desarrollo Ambiental Sustentable—VIDAS), and to Jaime Fonseca and Alex Mercado (Department of Biology, University of Puerto Rico, Río Piedras Campus) for laboratory and logistical support. Our thanks also to volunteer members of NGO Sociedad Ambiente Marino (SAM) and its Students Chapter (CESAM) for their support. This publication was part of the B.Sc. Honors Thesis project of G. Díaz-Ortega and a contribution from CATEC’s Coral Reef Research Group and SAM’s collaborative Coral Reefs Conservation and Rehabilitation Project.
NOTES
*Corresponding author.