1. Introduction
Appropriate expression of proteins in epidermis is a basic condition for the formation of a competent epidermal barrier (EB). Keratinocytes undergo different molecular changes during the transition from the basal layer into the spinous layer, the granular and finally to the stratum corneum. This process includes the synthesis of basal (K5 and K14) and suprabasal keratins (K1 and K10).
Different proteins, such as filagrin, loricrin, trichohyalin, small proline-rich proteins, involucrine and keratines, are located in the cornified cell envelop and are cross-linked by the action of different transglutaminases [1] -[5] . In order to assure an optimal structure and function of epidermis, it is important that both differentiation and proliferation of keratinocytes are well balanced. Variations in these processes, as well as genetic abnormalities in the genes encoding proteins of the cornified cell envelop components can lead to an abnormal keratinization, which is typical from certain skin conditions, like Atopic Dermatitis [6] -[8] .
Human skin models (in vivo studies) are the best way to analyze the main functions of skin, including the EB structure. However, the studies in human volunteers have major limitations to evaluate EB such as analysis by biopsy, because there is no ethic in humans. For this reason indirect parameters are evaluated (transepidermal water loss, skin capacitance, erythema, skyn surface analysis and/or SCORAD) [9] -[15] instead of localization and expression of proteins. Furthermore, other drawbacks including the genetic variation among individuals, can lean to different effects of environmental factors on individuals from the same population [2] [16] -[20] , and of course the number of volunteers needed to obtain significant results [21] .
There is a considerable amount of new topical products on the market aimed to improve or to relieve certain symptoms associated to skin diseases like the EB in Atopic Dermatitis [9] [11] -[15] [18] [22] -[25] . Many of them are formulated with different active substances which have been analyzed by in vitro approaches or in animal models [22] [25] -[32] , but not in the pool of ingredients of the final product. This is important because of the complexity of the formulas, where ingredients of different mechanism of action can overlap and do not give significant results [22] .
Nowadays, in vitro models are important tools for the evaluation of safety and efficacy of topical or dermatological products [33] . A reconstructed skin model should show protective barrier function and the same behavior to environmental contaminants. In this context, validated models that show a structure similar to human skin, with proliferative basal layer, spinous, granular and cornified layer and also similar expression and distribution of structural proteins such as keratins, constitute an excellent tool for studying the effects on the structure and function of different agents, chemicals or physicals [34] . This fact allows quantification measures out of direct parameters, like localization and protein expression or regeneration levels.
The aim of this work was to demonstrate that the product REPAVAR ATOPIC SKIN BODY CREAM EXTREME, containing “SKIN CALM COMPLEX”, mix of scientifically tested efficacy actives, improves EB at structural level.
2. Methods
2.1. In Vitro Model
In vitro Reconstructed Human Epidermis of 0.33 cm2, Age Day 10 (12 samples) or Age Day 17 (12 samples), produced by Skin Ethic laboratories (Nice, France) were used. The tissues were cultured and kept in chemically defined medium, provided by the same laboratory, during the assays. Tissue cultures were performed in an incubator with 5% of CO2 at 37˚C. The tissue manipulation process was carried out in sterile conditions.
2.2. Treatments
4 samples per group were used, 1) negative control (Skin Ethic Maintenance Medium-Skin Ethic laboratories, Nice, France), 2) positive control of proliferation and differentiation treatment with 100 nM Vitamin D (1,25- dihidroxivitamina D3) (Sigma, St. Louis, MO) [35] -[37] , diluted in maintenance medium for 24 hours or 7 days and 3) Product, REPAVAR ATOPIC SKIN BODY CREAM EXTREME, applied topically to the epithelium (enough to cover the tissue).
Immature tissues (Age Day 10) were treated for 7 days (7 days-treatment) and the mature tissues were treated for 24 hours (24 hours-treatment).
2.3. Histological Processing
Samples (3 negative controls, 3 positive controls and 3 treated tissues, for 24 hours or 7 days of treatment) were fixed by immersion in 3.7% formaldehyde (Panreac, Barcelona, Spain) in phosphate buffered saline (PBS) (HyClone Laboratories, Inc., South Logan, UT) for 12 hours at 4˚C. Subsequently, they were washed and dried in increasing ethanol (Panreac, Barcelona, Spain) series before inclusion in paraffin. Finally, histological sections (4 μm thickness) were placed on electrostatically charged slides (Superfrost® Plus) (Menzel-Gläser, Braunschweig, Germany).
2.4. Hematoxylin-Eosinand Inmuno-Staining
For morphological evaluation, sections were deparaffinized for 15 minutes in xylene (Panreac, Barcelona, Spain), and hydrated in decreasing ethanol series and washed with distilled water. All samples were stained with hematoxylin/eosin (H&E), according to standard protocols, and examined under the microscope (Olympus BX61 coupled to a digital camera Olympus DP50).
For inmunostaining, sections were deparaffinized, hydrated and washed with distilled water. Inhibition of endogenous peroxidase was carried out with hydrogen peroxide (Panreac, Barcelona, Spain) at 3% in methanol (Panreac, Barcelona, Spain). Afterwards, the sections were washed with PBS. Next, antigen retrieval was performed in citrate buffer at pH 6 (0.25% citric acid and 0.038% sodium citrate in water). Sections were blocked with non-immune serum and incubated with the primary antibody against the proteins under study, antiE-cadherin (BD Transduction Laboratories, Franklin Lakes, NJ), anti-Involucin, anti-Loricrin and anti-Filagrin (Sigma, St. Louis, MO), anti-Keratin-10 (Dako, Glostrup, Denmark) and anti-Keratin-5 (Abcam, Cambridge, UK) overnight. For immnunohistochemistry (IHQ), the excess primary antibody was washed with PBS and sections were incubated with biotinylated antiserum for 25 minutes at room temperature with streptavidin-peroxidase, developed with diaminobenzidine and counterstained with Harris hematoxylin for nuclear contrast. The processing for immunofluorescence (IF), sections were incubated with corresponding secondary antibody, mounted with ProLong® Gold Natifade Reagent (Invitrogen, Frederick, MD) with DAPI for nuclear contrast and observed under the fluorescence microscope.
2.5. MTT Assay
Cellular proliferation was assessed by MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl-2H-tetrazolium bromide) assay (Sigma, St. Louis, MO), a method based on the activity of mitochondrial dehydrogenases [38]. Stock solution of MTT (5 mg/ml) in PBS, was diluted (1 mg/ml) in Skinethic culture medium, and then the samples were incubated at 37˚C, 5% CO2, 95% humidified atmosphere, for 3 hours. The resulting formazan crystals were dissolved by the addition of isopropanol (Sigma, St. Louis, MO), at room temperature, for 2 hours with gentle agitation. Optical Densities (OD) were measured at 570 nm wavelength.
2.6. Optical Microscopy
Microscopic observations were carried out using an Olympus BX61 epifluorescence microscope equipped with filter sets for fluorescence microscopy: ultraviolet (UV, 365 nm, exciting filter UG-1), blue (450 - 490 nm, exciting filter BP 490), and green (545 nm, exciting filter BP 545). Photographs were obtained with the digital camera Olympus CCD DP70 and processed using the Adobe PhotoShop CS5 extended version 12.0 software (Adobe Systems Inc., USA). The expression of some proteins was quantified using ImageJ 1.37v (http://rsb.info.nih.giv/ij). The relative fluorescence intensity was determined from, at least, 20 areas of 500 μm2 for each treatment condition.
2.7. Statistical Analysis
The statistical analysis was performed using the SPSS 15.0, 1 software (SPSS Inc., Chicago, IL), using Student’s t test and analysis of variance (ANOVA), and considering a p <0.05 as statistically significant.
3. Results and Discussion
3.1. Morphological Analysis
Figure 1 shows the morphological analysis, by hematoxylin-eosin staining, of the treated reconstructed epidermis (Vitamin D positive control group, product REPAVAR ATOPIC SKIN BODY CREAM EXTREME) and untreated epidermis (negative control) for 1 or 7 days.
Tissue samples, in all cases, showed a normal stratification, appreciating the different layers, basal (B), spinous

Figure 1. Histological sections of a representative reconstructed control (negative control), Vitamin D-treated (positive control) or REPAVAR ATOPIC SKIN BODY CREAM EXTREMEtreated (product) epidermis for 24 hours (left panel) or 7 days (right panel), stained with H&E. All samples show a good arrangement of the layers: basal (B), spinous (S), granular (G) and cornified layer (C). Note the increased epidermal thickness in the treated group (Product). Scale bar: 50 μm.
(S), granular (G) and cornified (C). Similarly, in all cases, a good organization was observed in the different cell layers of the epidermis.
Interestingly, Vitamin D-treated epidermis as much as Product-treated tissues showed a thick increase compared to negative control, being greater in the case of Product treatments (24 hours and 7 days).
When assessing the epidermal thickness (cornified layer not included) by morphological evaluation, the results above described were confirmed. It is shown a statistically significant increase in the thickness of the epidermis treated for 24 hours (p < 0.05, Student t-test) (Figure 2).
All evaluations were carried out on immature (7 days-treatment) and mature tissues (24 hours-treatment) to confirm the effect of the product in both types of tissues but, because of thickness of cornified layer was smaller in all sections in immature epidermis, the analyses of this study were focused on mature epidermis treated for 24 hours (and non-treated).
All these results were confirmed by quantitative analysis. Product REPAVAR ATOPIC SKIN BODY CREAM EXTREME promotes an increase in epithelial proliferation of about 50% over the negative control (untreated tissue) and 30% more than Vitamin D, used as positive control (Figure 3).
3.2. Analysis of Protein Expression
The key proteins expression in organization and differentiation of epidermis was analyzed by IHC and immunofluorescence IF (Figures 4-7).
E-cadherin is a very important protein in epithelial cell organization, participating in cell-cell adhesion [5] [7] . The tissues treated for 24 h with Product show a good expression and localization of this protein on cell membrane, especially in the keratinocytes of the spinous layer and granulosum and lower in layer baseline (Figure 4). There were no differences in the amount of protein expression between the different sample groups, treated and controls.
An important protein which is frequently altered in Atopic Dermatitis and other skin conditions is Filaggrin [2] [39] [40] . This protein is produced from its precursor, the functionally inactive profilaggrin, which is proteolytically processed to Filaggrin during the transition from the granular layer of the cornified layer. Filaggrin is, therefore, an excellent marker of epidermal differentiation. Figure 5 shows the high expression of this protein in

Figure 2. Measuring thickness of reconstructed epidermis: untreated tissues (control, Ctrl), positive control (Vitamin D, vit D-treated tissues, vit D) or Product (REPAVAR ATOPIC SKIN BODY CREAM EXTREME-treated tissues) for 24 hours (left) or 7 days (right). There is a significant increase of thickness in Vitamin D-treated and Product-treated epidermis for 24 hours (p < 0.05).

Figure 3. Cell viability (%) estimated by the MTT assay: untreated tissues (Ctrl), positive control (Vitamin D, vit D-treated tissues) or product (REPAVAR ATOPIC SKIN BODY CREAM EXTREME-treated tissues) for 24 hours. The Product-treated sample shows an increase in cell viability around 60%.
the keratinocytes located between the upper region of the granular layer, in contact with the keratin layer, in the reconstituted epidermis exposed to Vitamin D and Product, with a more precise localization, in both epidermis in relation to the untreated control, which is also located in spinous layer. This protein is not expressed in the stratum corneum. Greater thickness of Product-treated samples is noteworthy and good differentiation is shown by the marker Filaggrin.
Loricrin is the precursor protein of the stratum corneum, expressed mainly in the upper granular layer. Loricrin appears well defined and localized, with higher expression in Product-treated epidermis in relation to the Vitamin D-treated (positive control) for 24 hours and untreated (negative control) (Figure 5).
Involucrin expression was determinated by immunofluorescence, because no conclusions could be drawn by immunochemistry. A lower expression of the protein in the basal layer and a suitable location in suprabasal layers were detected in the case of Vitamin D-treated and Product-treated tissues (data not shown).
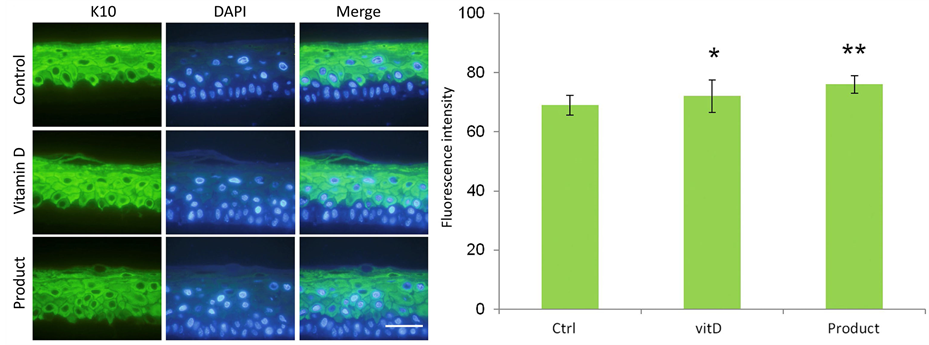
Finally the expression of two keratins, K10 and K5 were analyzed, by immunofluorescence. Keratin 10 (Figure 6) preferably located in the suprabasal layers. Meanwhile, keratin 5 (Figure 7) is preferentially expressed in the basal layer. The results show excellent labeling in all cases (treated and control samples) with Keratin 10, especially in Product-treated equivalents (Figure 6(a)). In this case, again, greater thickness is notable and good differentiation is shown by this new marker. Moreover, the measure of fluorescence intensity (Figure 6(b)) indicates a higher expression in suprabasal layers of treated epidermis compared to control
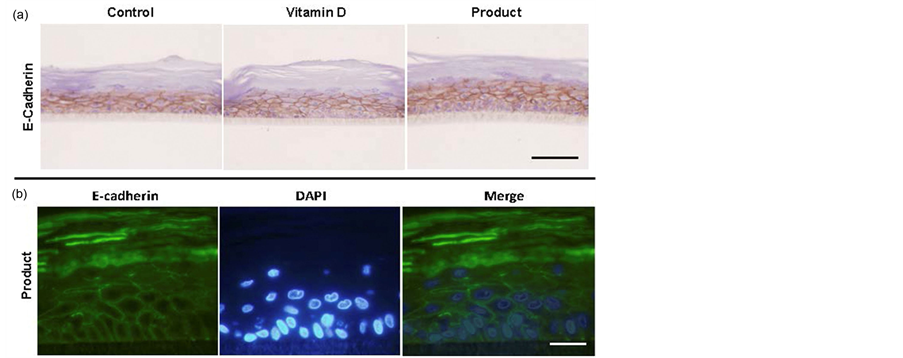
Figure 4. (a) Histological sections of representative reconstructed control (negative control), Vitamin D-treated (positive control) or REPAVAR ATOPIC SKIN BODY CREAM EXTREME-treated (Product) epidermis for 24 hours, and exposed to E-cadherin antibody for determination by immunohistochemistry. Scale bar: 50 μm; (b) Indirect immunofluorescence for the adhesion molecule E-cadherin in reconstituted REPAVAR ATOPIC SKIN BODY CREAM EXTREME-treated (Product) epidermis for 24 hours. Excellent membrane localization appreciated in spinous and cornified layers. In the basal layer is slightly lower. Scale bar: 25 μm.

Figure 5. Immunohistochemistry for Filaggrin and Loricrin in representative reconstructed control (negative control), Vitamin D-treated (positive control) or REPAVAR ATOPIC SKIN BODY CREAM EXTREME-treated (Product) epidermis for 24 hours. An excellent location for all the proteins studied can be appreciated, both in positive control and Product-treated group. Scale bar: 50 μm.
samples. Keratin 5 (K5) analysis, by indirect immunofluorescence, revealed increased expression of this protein in the basal layer of both Vitamin D-treated and Product-treated epidermis. No striking differences were observed between Vitamin D and Product expression and localization.
All these results were confirmed after 7 days of treatment on immature (10 day aged) tissues.
Skincalm Complex® is a mix of scientifically tested efficacy actives that includes a skin-identicallipidiccomplex (SK-influx®), γ linoleic acid (Railinol®), botanical hidrolized protein, pool of 7 botanical extracts, Dihydroavenanthramide D and Peumusboldus extract. It restores EB [41] [42] and increases synthesis and functions of epidermal proteins [43] .
Evaluating structural proteins involved in Atopic Dermatitis is not possible by human biopsy. For this reason
 (a) (b)
(a) (b)
Figure 6. (a) K10 expression by indirect immunofluorescence in reconstructed normal, Vitamin D-treated (positive control) or REPAVAR ATOPIC SKIN BODY CREAM EXTREME-treated (Product) epidermis for 24 hours. It shows a greater thickening of the treated epidermis, especially the tissues exposure to product. Scale bar: 25 μm; (b) Quantification of K10 expression using imagenJ program. The differences are significant (p < 0.05), between Vitamin D-treated epidermis (positive control) and negative control, and between Product-treated epidermis and other groups.

Figure 7. K5 expression by indirect immunofluorescence in reconstructed normal, Vitamin D-treated (positive control) or REPAVAR ATOPIC SKIN BODY CREAM EXTREME-treated (Product) epidermis for 24 hours. There is greater fluorescence intensity in the basal layer of the epidermis (white arrow) due to the increased expression of the protein in Vitamin D- (differentiation enhancer) or Product-treated epidermis, in relation to the untreated control. Scale bar 25 μm.
animal models are used. Nowadays, reconstructed human skin models allow this approach [34] , representing a step forward in the evaluation of efficacy of human structural proteins involved in Atopic Dermatitis. The results obtained after treatment for 24 hours show that the product REPAVAR ATOPIC SKIN BODY CREAM EXTREME induces, compared to Vitamin D-treated and untreated tissues, 1) an excellent effect on the organization of the epidermal layers, showed in the histological sections stained with H/E; 2) enhances epidermal proliferation, showed in the increased thickness quantified by MTT assay; and, 3) promotes differentiation process, showed in the increased expression of the analyzed proteins Fillagrin, Loricrin and Keratins (10 and 5), and optimal distribution of all of them, as well as E-cadherin and Involucrin (by immunofluorescence or inmunochemistry analysis).
4. Conclusion
As a final conclusion, the study demonstrated that the product REPAVAR ATOPIC SKIN BODY CREAM EXTREME is able to enhances skin regeneration, balancing both proliferation and differentiation of skin layers and thus, improving the epidermal barrier effect.