Hot Spring Hydro Therapy Regulates Peripheral Leukocyte Together with Emotional Hormone and Receptor Positive Lymphocytes According to Each Constitution/Condition* ()
Throughout the world, the phenomenon of the natural hot hydro therapy spawned in the central Asia and East Asia and Europe. Mongolia has a long history of socalled immersion therapy recorded in Chinese or Mongolian medical works. In Gallo-Roman France, a most important number of documents, coins, various china, and votive stones survive from the province of Aquitaine. Even after two thousand years the therapeutic qualities of several mineral hot waters springs of that era are still recognized. This impressive history expands up into Germany, the Netherlands and through the whole of middle and Eastern Europe, extending as far as the well-known bathing centers and customs of Turkey. Japanese geological position on the edge of the Pacific Plate has given its volatile underpinnings, always changing and bursting forth in volcanic and earthquake activity. The legacy of this activity is the plethora of hot springs scattered all through its abundant islands. For centuries, these hot springs have been therapeutic destinations for the islands’ inhabitants.
Traditionally, hot spring therapy has its own character and efficacy for various complaints. Through the years, each water source was evaluated for its specific properties and with the advent of better transportation in our mountainous land, even remote springs in the mountains were visited for their specific medicinal effect. For example, the hot spring of Fukatani, which is located just below our research facility, was known all over the area to cure hemorrhoids. However, almost all the judgments of efficacy are VAS (visual analog scale). The proprietors of the hot spring inns say that many of the guests came to cure that sort of ailment since about 20 years ago. Now in Japan, hot spring hydrotherapy is often used as a supplementary therapy for many diseases [24]. It has shown to reduce surgical complaints such as shoulder pain, amyloidosis and various rheumatic problems. It can also lighten the burden to the heart and improve the condition of patients who suffer from emphysema as well as other respiratory ailments. Hot springs have also traditionally functioned as places to relax and enjoy oneself, even though resort-type hot springs always exist along with those for illnesses. In recent years, trends have been seen that even remote hot springs, such as the one below our facility, transform into fashionable resorts for the healthy to visit for relaxation and stress release. It is interesting to note that this historical duplicity encompassed by hot springs has also entered the world of Western medicine as the release of mental stress and physical fatigue has been shown to be essential for good health. Put into other words: the relaxation side of the hot spring promotes prevention, including health enhancement, and the medicinal side, the treatment of illness.
Recently, hot spring hydrotherapy has sparked more and more interest and many objective studies have been established. What is the mechanism of hot spring hydrotherapy that affects humans? It is known that the function of the immune system is closely related to the nervous and endocrine systems, and varies with physical conditions and changes in the surroundings [1-12]. It seems reasonable that hot spring hydrotherapy should also influence the immune system, but so far no objective studies have been made. Finding the answer to this query became the focus of our investigation.
In order to investigate the influence of hot spring hydrotherapy on the immune system, we examined over 20 groups that consisted with 10 - 15 individuals/group during 1999-2010. Our results showed that within 24 hours after hot spring hydrotherapy, the white blood cells in peripheral blood had changed significantly, not only in cell count but also cell function. In Japan, centuries of tradition have shown that alternative therapies like hot spring hydrotherapy, acupuncture, and herbal medicine enhance the QOL of empirically healthy individuals. Evidence has been accumulating that this may be the result of immune system regulation. The scientific basis, however, has not yet been established.
In our investigation, we measured the number of leukocyte subsets, granulocyte and lymphocytes regulated before and after hot spring hydrotherapy. The propose of moreover, every individual exposes to the risk of immunodeficiency in daily life with both internal and externals stimuli. In this report, we tried to report the suitable menu for regulating the immune system modulate and discuss how we can compare and select each menu by evidencebased manner more than VAS: visual analog scale.
The major purpose of this script is concerned with the following issues. One is to confirm the effect of hot spring hydrotherapy within a short time is crucial or not. In other words, the reputational results are possible or not. The regulation was constitution/condition dependent manner and regulated the ideal value to each individual from the condition before the menu. The second was that this regulatory bias coincided with other factors influenced by pituitary nervous system, such as emotional hormone and the hormone receptor positive lymphocyte. The third one was that reputational effect can be expressed by the linear function and could compare each other with the value of slope. We tried to discuss more, the possibility of the exhibition and comparison between, each menu and established medicine in East and West.
The results showed that these subsets could reflect the number and function of immuno-competent cells. For example, in an individual with a low granulocyte number, the number increased after treatment, while it decreased in another individual with a higher cell number. Our results led us to believe that leukocyte subsets could be an interesting indicator for the evaluation of alternative therapies. Many systems are in place to evaluate Western therapies that aim at healing the symptoms of an illness. However, when the purpose of a therapy is to enhance the QOL of healthy people, such as some alternative medical therapies, it is not widely-accepted. Evaluation system has been established. To fill this lack, we would like to propose the umber and functions of leukocyte subsets as indicators for the evaluation of alternative therapies.
2. Subjects and Methods
2.1. Subjects
We found 12 healthy volunteers (age, 41 ± 15.2 years) and informed consented according to The Ethics Committee of Kanazawa Medical University.
In addition, the hydrotherapy group was further divided into two groups according to the variation after hydrotherapy. After preliminary results showed that a critical change in up and down regulation occurred at around 35 years old, the participants were divided according to age: the younger group was 35 years old and under, and the older group was over 36. According to the percentage of lymphocytes or granulocytes in the total leukocytes, the hydrotherapy group was further divided into two types: subjects whose granulocyte number was over 60% belonged to the G type and those whose lymphocyte number was over 40% belonged to the L type.
2.2. Hot Spring Hydrotherapy
We sampled peripheral blood from the 12 volunteers before and after hot hydrotherapy, at the same time on each day, in consideration of circadian rhythm [25-27] of leukocyte. These subjects participated in this study after

Figure 1. Experimental design of this report: We sampled peripheral blood from the 12 volunteers before and after hot hydro therapy, at the same time on each day, in accordance with the consideration of circadian rhythm of leukocyte. The spring quality is a weak sodium chloride with sodium carbohydrate of the water temperature 41˚C ± 1˚C. During the night and in the morning of the next day, they had a bath in the hot spring two or three times, for 20 - 30 minutes each time. Time interval of blood sampling between before and after hot-spring hydrotherapy was approximately 24 hours. The total and differential leukocyte counts were measured by the automated hematology analyzer.
giving their informed consent. We conducted the experiment at Chugu Onsen Spa (Oguchi Village, Ishikawa Pref., Japan) in one night of the day. The spring quality is a weak sodium chloride with sodium carbohydrate of the water temperature 41˚C ± 1˚C. During the night and in the morning of the next day, they had a bath in the hot spring two or three times, for 20 - 30 minutes in each time. Time interval of blood sampling between before and after hot-spring hydrotherapy was approximately 24 hours. Measurements of the total and differential leucocyte counts and 3 catecholamines levels in the peripheral blood were done (Figure 1).
We depended on the laboratory of Ishikawa Prefecture Preventive Medicine Association about the total and differential leukocyte counts and the levels of 3 catecholamines (adrenaline, noradrenaline, and dopamine) in the peripheral blood from the subjects. The total and differential leukocyte counts were measured by the automated hematology analyzer XE-2100 (Sysmex, Inc., Kobe, Japan). The levels of catecholamines were measured by high performance liquid chromatography (HPLC) system (Tosoh Co. and Hitachi High-Technologies Co., Japan).
2.3. Assessment of Emotional Hormone as by Stander Witness of Short Term Regulation of Peripheral Leukocyte
The analysis of β2-AR expressing cells and CD positive cells by FCM was measured by gating in the lymphocytes region on the scattered gram. Figure 2 shows an example analysis. Nonspecific reaction of the PE fluorescence was found in the isotype control. Therefore, the real values of the AR expressing cell counts were calculated by subtracting the control values. The CD19+ cells were observed nonspecific reaction which seems to response of the second antibody. However, the significant variation was not seen from the comparison of β2-AR expressing cells before and after hot-spring hydrotherapy. The mean % of β2-AR expressing cells in the lymphocyte subsets was 18% - 19% in CD3+ cells, 5% in CD4+ cells, 57% - 63% in CD8+ cells, and 93% - 95% in CD56+ cells. That in CD19+ cells was approximately 100% (data not shown), but we were not able to be confirmed because it was very likely to be the nonspecific reaction.
The adrenaline and noradrenaline are secreted by various stress (stimulation); the former from the adrenal medulla and the latter from both the adrenal medulla and the end of sympathetic nerve. They express these functions through the adrenergic receptors (ARs) and regulate the target organ. The adrenergic receptor has two types of α and β, and there are multiple subtypes (α1, α2, β1, β2, β3) [4]. These subtypes present the vascular (α1), the presynaptic terminal (α2), the heart (β1), the bronchial muscle of the lung (β2), fat cells (β3), respectively. The α-receptor stimulation causes bronchodilatation andvasodilaHormonal Level & Vector
tation, the β1-receptor stimulation causes an increase in heart rate and lipolysis, and the β2-receptor stimulation causes bronchoconstriction, vasodilatation, and muscle glycogen resolution. The adrenaline provides heart activation by effect on α- and β-receptors, and the noradrenaline provides blood pressure rises by strongly effect on α-receptors. In addition to the above, it has been reported that the ARs are present on leucocyte membranes [20,21] and that the level of expression of β-ARs in lymphocytes was examined by radioligand (125I-iodopindolol) binding, but the details of subtype of β-receptors have not been disclosed. The subtypes of the AR are able to be analyzed by flow cytometry (FCM) method. In this study, we described how hot spring hydrotherapy influences leukocyte, lymphocyte subpopulations expressing β2-AR and the levels of catecholamine in human peripheral blood.
2.4. Assessment of the β2-AR Expression in Lymphocyte Subsets by FCM
The whole blood obtained from the subjects by blood collection tube contain an anticoagulant EDTA-2K. 100 μl of whole blood were added the anti β2-AR antibody (Santa Cruz Biotechnology, Inc. USA) of the primary antibody and were incubated for 30 minutes at 4˚C. Mouse IgG1-isotype control antibody was used as negative controls. After two times washing by phosphate buffered saline solution (PBS, pH 7.2), the samples were added biotin-labeled goat anti-mouse IgG1 antibodies (Santa Cruz Biotechnology) as a secondary antibody and were incubated for 30 minutes at 4˚C. After washing with PBS, the suspensions were added phycoerythrin (PE)- conjugated streptavidin (Beckman Coulter Inc. France) and fluorescence monoclonal antibody: peridinin chlorophyll protein-cyanin 5.5 (PerCP-Cy5.5)-conjugated CD3, fluorescein isothiocyanate (FITC)-conjugated CD4, FITCconjugated CD19 (each Becton Dickinson Co. USA), allophycocyanin (APC)-conjugated CD8, and APC-conjugated CD56 (each Beckman Coulter). The negative controls were added PE-conjugated streptavidin and the isotype control antibodies to the CD antibodies. After incubation for 30 minutes at 4˚C, these samples were hemolyzed using a 10-times dilution FACS Lysing Solution (Becton Dickinson). After washing with PBS, the cell suspensions were fixed using a 10-times dilution CellFIX (Becton Dickinson) and analyzed by flow cytometer FACS Caliber (Becton Dickinson) [27-29].
2.5. Statistical Analysis
The statistical comparisons between two groups (before and after hot-spring hydrotherapy) for the test of significant difference were performed using paired t-test and wilcoxon signed-ranks test. Further, the test of the correlation was performed a spearman’s correlation coefficient by rank test. Data are expressed as means ± standard error of mean (SE). A p value < 0.05 was considered to be statistically significant.
3. Results
3.1. Twenty-Four Hours Change of Total Peripheral Leukocyte Count on the Basis of Each Individual
Each volunteer prepared their blood before start to visit hot spring village after informed consented to the experimental purpose by written Ethics of the Committee in Kanazawa Medical University. After lodging the hotel, volunteers enjoyed Japanese type hot spring bath system, with no under wear in the bath room/pool separately prepared for ladies and gentleman. We suggested to take a bath totally 2 - 3 times within two days for the aim of trial. Beside the advance of VAS: visual analog scale in our investigation, we measured the total number of leukocyte and two major subsets, granulocyte and lymphocytes regulated before and after hot spring hydrotherapy.
We tried to express the effect of peripheral total leukocyte number by individual level of change and plot in the x-axis as in each age. As was in Figure 3, the groups were separated into two, up-regulated individuals and down-regulated one. The correlation of change was expressed as a linear function. Figure 3 was ideal change of effect with hot spring hydrotherapy, showing the best inclination. The results showed that these subsets could reflect the number and function of immuno-competent cells. For example, in an individual with a low granulocyte number, the number increased after treatment, while it decreased in another individual with a higher cell number (Figure 3).
We sampled peripheral blood from the 12 volunteers before and after hot sand/hydro therapy, at the same O’clock on the day, with the respect of circadian rhythm [25,27] of leukocyte. These subjects participated in this study after giving their informed consent. We conducted the experiment at Chugu Onsen Spa (Oguchi Village, Ishikawa Pref., Japan) in one night of the day. The spring quality is a weak sodium chloride with sodium carbohydrate of the water temperature 41˚C ± 1˚C. During the night and in the morning of the next day, they had a bath in the hot spring two or three times, for 20 - 30 minutes each time. Hot sand spa had performed and bathing for 40 min. Time interval of blood sampling between before and after hot-spring hydrotherapy was approximately 24 hours. Measurements of the total and differential leukocyte counts and 3 catecholamines levels in the peripheral blood are done.
Relative value of total leukocytes
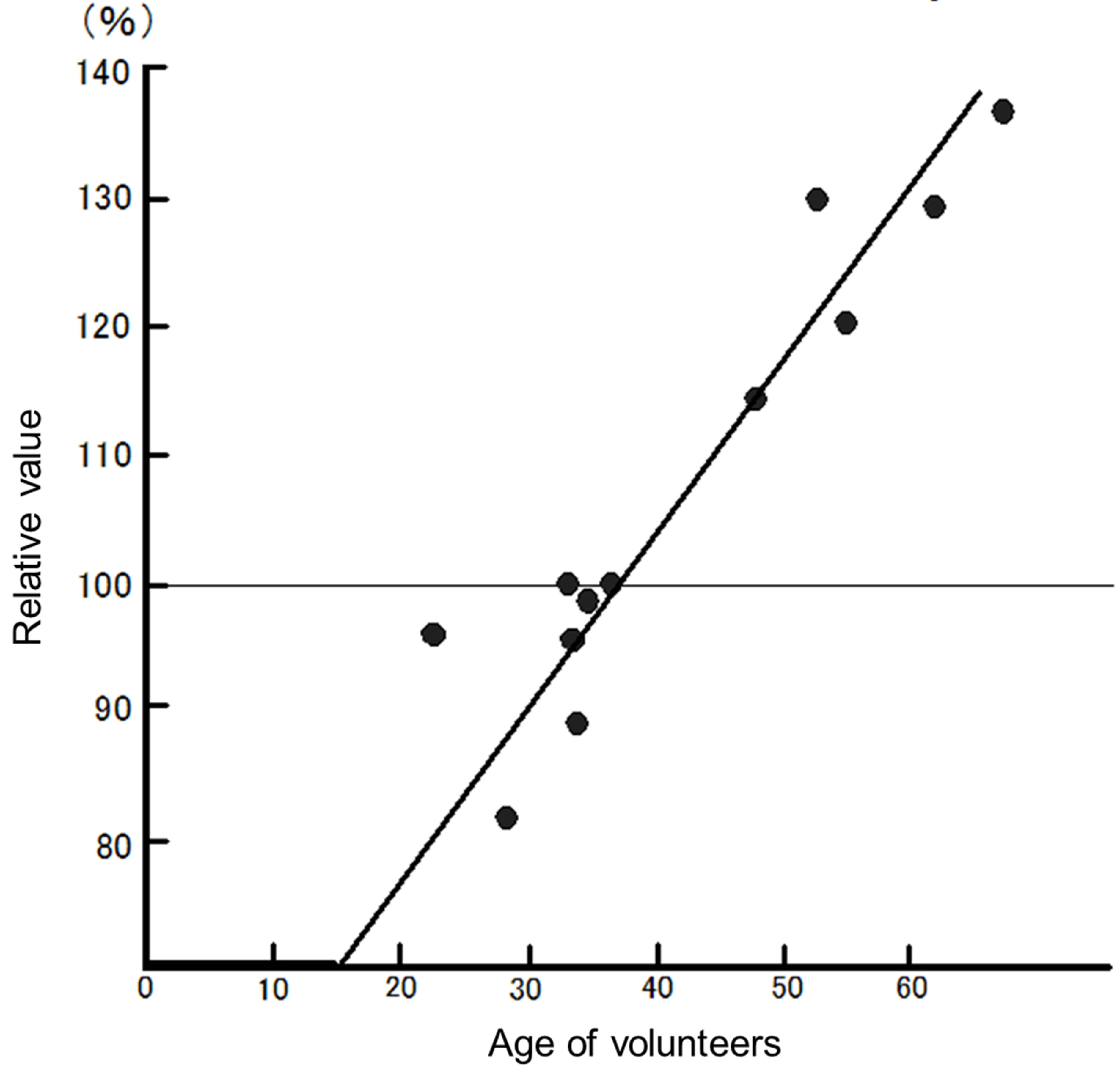
Figure 3. Twenty-four hours change of total peripheral leukocyte count on the basis of each constitution/condition: We tried to express the effect of peripheral total leukocyte number by individual level of change and plot in the x-axis as in each age of volunteer. Variations in leukocyte subpopulations in the peripheral blood before and after hot spring hydrotherapy.
We ordered on the laboratory of Ishikawa Prefecture Preventive Medicine Association about the total and differential leukocyte counts and the levels of 3 catecholamines (adrenaline, noradrenaline, and dopamine) in the peripheral blood from the subjects. The total and differential leukocyte counts were measured by the automated hematology analyzer XE-2100 (Sysmex, Inc., Kobe, Japan). The levels of catecholamines were measured by high performance liquid chromatography (HPLC) system (Tosoh Co. and Hitachi High-Technologies Co., Japan).
3.2. Twenty-Four Hours Change of Leukocyte Counts on the Bases of Group Comparison between Pre/Post Hydrotherapy
Despite the expression in the section 3.1., when we tried to explain these values into the grouped value before and after the hydrotherapy, we got only one positive result from the granulocyte change that was the major population of peripheral leukocytes. The other factor did not exhibit as positive result (Figure 4).
The total and differential leukocyte counts in the peripheral blood of 12 subjects tended to decrease after hotspring hydro-therapy. The statistically significant result only obtained that the granulocyte counts significantly decreased (p < 0.05). Another minor group lymphocyte did not changed significantly by judging from simple

Figure 4. Twenty-four hours change of leukocyte counts on the bases of group comparison between pre/post Hydrotherapy: We sampled peripheral blood from the 12 volunteers before and after hot spring hydrotherapy, at the same time on each day, in accordance with the consideration of circadian rhythm of leukocyte In this figure, we tried to show the date simply pooled and make mean, then compared.
sum up and made average (Figure 4).
3.3. Emotional Hormones Also Regulated with Condition/Constitution Dependent Manner
We had been observed the reputational effect of hydrotherapy could be evidenced within a short period. But the possibility still remain that the leukocyte change was happen to emerge for the dairy life as accidental factor, such as stress and so on. In order to avoid such possibility, then we tried to show the change of the peripheral leukocyte number was the result of another network system of the inner system such as peripheral leukocyte, endocrine and brain system. We sampled peripheral blood from the 12 volunteers before and after hot sand/hydro therapy, at the same time on the next day, with the respect of circadian rhythm (of leukocyte [25-27]. These subjects participated in this study after giving their informed consent. We conducted the experiment at Chugu Onsen Spa (Oguchi Village, Ishikawa Pref., Japan) in one night of the day. The spring quality is a weak sodium chloride with sodium carbohydrate of the water temperature 41˚C ± 1˚C. During the night and in the morning of the next day, they had a bath in the hot spring two or three times, for 20 - 30 minutes each time. Time interval of blood sampling between before and after hot-spring hydrotherapy was approximately 24 hours. Measurements of the total and differential leukocyte counts and 3 catecholamines levels in the peripheral blood (Figures 2 and 5)
The adrenaline and noradrenaline are secreted by various stress (stimulation); the former from the adrenal medulla and the latter from both the adrenal medulla and the end of sympathetic nerve. They express these functions through the adrenergic receptors (ARs) and regulate the target organ. The adrenergic receptor has two types of α and β, and there are multiple subtypes (α1, α2, β1, β2, β3) [21]. These subtypes present the vascular (α1), the presynaptic terminal (α2), the heart (β1), the bronchial muscle of the lung (β2), fat cells (β3), respectively. The α-receptor stimulation causes bronchodilatation and vasodilatation, the β1-receptor stimulation causes an increase in heart rate and lipolysis, and the β2-receptor stimulation causes bronchoconstriction, vasodilatation, and muscle glycogen resolution. The adrenaline provides heart activation by effect on α- and β-receptors, and the noradrenaline provides blood pressure rises by strongly effect on α-receptors. In addition to the above, it has been reported that the ARs are present on leucocyte membranes [22-24] and that the level of expression of β-ARs in lymphocytes was examined by radioligand (125I-iodopindolol) binding, but the details of subtype of β-receptors have not been disclosed. The subtypes of the AR are able to be analyzed by flow cytometry (FCM) method. In this study, we described how hot spring hydrotherapy influences leukocyte, lymphocyte subpopulations expressing β2-AR Thyoid Hormones
 age T4(μg/dl)
age T4(μg/dl) 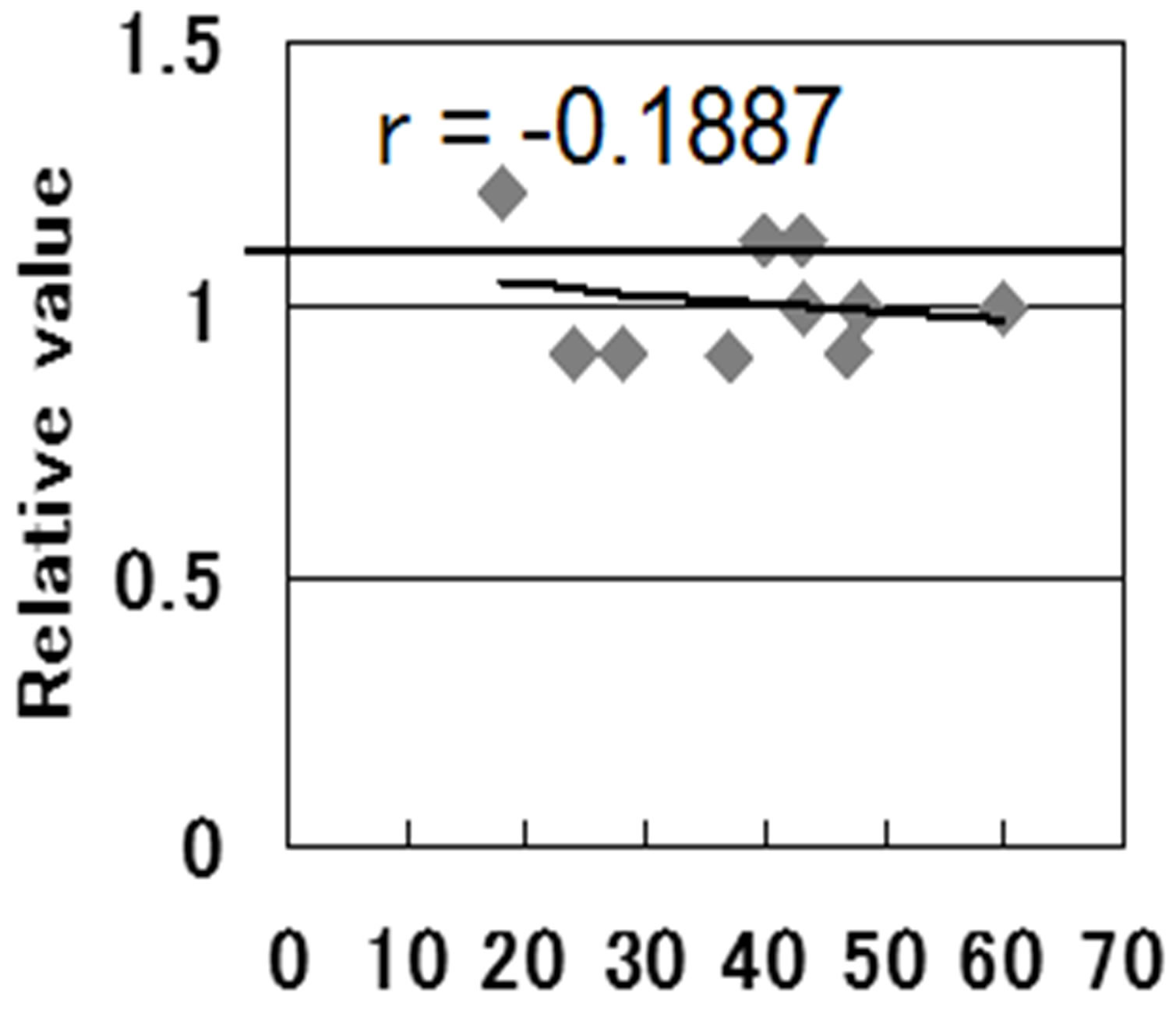 age T3(μg/dl)
age T3(μg/dl)  age TSH(μU/ml)
age TSH(μU/ml)
Figure 5. The constitution dependent analysis for (adrenaline and dopamine). Each spot were obtained from the calculation comparing relative value from before and after levels in the serum, catecholamines levels in the peripheral blood relative value 1 shows no change before and after the menu. The constitution dependent analysis, the detail change and vector of each change could find from individual data, showing higher value volunteer down regulated much more than lower leveled one.
and the levels of catecholamine in human peripheral blood.
3.4. CD Positive Lymphocytes Correlate with Serum Adrenaline Level, as a Result of the Regulation by Hot Spring Hydrotherapy
The analysis of CD positive cells by FCM was measured by gating in the lymphocytes region on the scattered gram. Figure 6 shows an example analysis. Nonspecific reaction of the PE fluorescence was found in the isotype control. Therefore, the real values of the AR expressing cell counts were calculated by subtracting the control values. The CD19+ cells were observed nonspecific reaction, which seems to response of the second antibody. The comparison of each lymphocyte subpopulations before and after hot spring hydrotherapy showed that the CD8+ cell and CD56+ cell counts tended to increase (Figures 5 and 7). However, the significant variation was not seen from the comparison of β2-AR expressing cells before and after hot-spring hydrotherapy. The mean % of β2-AR expressing cells in the lymphocyte subsets was 18% - 19% in CD3+ cells, 5% in CD4+ cells, 57% - 63% in CD8+ cells, and 93% - 95% in CD56+ cells. That in CD19+ cells was approximately 100% (data not shown), but we were not able to be confirmed because it was very likely to be the nonspecific reaction (Figures 6, 8 and 9).
We examined the correlation with the rate of change in adrenaline levels and the rate of change in β2-AR expressing cell counts of each subset or that in each CDpositive cell counts before and after hot-spring hydrotherapy. In the CD-positive cells, the rate of change in adrenaline levels was a positive correlation with that in the levels of CD56+ cells, CD8+ cells, and CD3+ cells; in particular, a correlation with CD56+ and CD8+ cells was high (Figure 5). In β2-AR expressing cells, the rate of change in adrenaline levels was a positive correlation with the rate of change in the levels of β2-AR+ CD56+ cells (Figures 8 and 10). These results suggested that the variation in adrenaline levels is correlated with CD56+ cells.
3.5. β2-AR+ Receptor Positive Lymphocytes Correlate with Adrenaline Level, as a Result of the Regulation by Hydrotherapy
The adrenaline and noradrenaline are secreted by various stress (stimulation); the former from the adrenal medulla and the latter from both the adrenal medulla and the end of sympathetic nerve. They express these functions

Figure 6. Relationship between the rate of change in adrenaline levels and the rate of change in the levels of CD-positive cells before and after hydrotherapy.

Figure 7. FACS analyses of CD positive cells in lymphocyte subpopulations before and after hot spring hydrotherapy:The analysis of CD positive cells by FCM was measured by gating in the lymphocytes region on the scattered gram. Fig. shows an example analysis. Nonspecific reaction of the PE fluorescence was found in the isotype control.

Figure 8. FACS analyses of β2-AR+ cells in lymphocyte subpopulations before and after hot spring hydrotherapy: The analysis of β2-AR expressing cells and CD positive cells by FCM was measured by gating in the lymphocytes region on the scattered gram.
through the adrenergic receptors (ARs) and regulate the target organ. The adrenergic receptor has two types of α and β, and there are multiple subtypes (α1, α2, β1, β2, β3). These subtypes present the vascular (α1), the presynaptic terminal (α2), the heart (β1), the bronchial muscle of the lung (β2), fat cells (β3), respectively. The α-receptor

Figure 9. Variations in the number of β2-AR+ cells in lymphocyte subpopulations: And adrenaline levels in the peripheral the analysis of β2-AR receptor expressing cells and CD positive cells by FCM was measured by gating in the lymphocytes region on the scattered gram. Fig. shows an example analysis.

Figure 10. Individual comparison of adrenalin levels and CD positive cells in the peripheral blood time interval of blood sampling between before and after hot-spring hydrotherapy was approximately 24 hours. Measurements of the total and differential leukocyte counts and 3 catecholamines levels in the peripheral blood.
stimulation causes bronchodilatation and vasodilatation, the β1-receptor stimulation causes an increase in heart rate and lipolysis, and the β2-receptor stimulation causes bronchoconstriction, vasodilatation, and muscle glycogen resolution. The adrenaline provides heart activation by effect on α- and β-receptors, and the noradrenaline provides blood pressure rises by strongly effect on α-receptors. In addition to the above, it has been reported that the ARs are present on leucocyte membranes and that the level of expression of β-ARs in lymphocytes was examined by radioligand (125I-iodopindolol) binding, but the details of subtype of β-receptors have not been disclosed. The subtypes of the AR can be analyzed by flow cytometry (FCM) method. In this study, we described how hot spring hydrotherapy influences leukocyte, lymphocyte subpopulations expressing β2-AR and the levels of catecholamine in human peripheral blood.
The analysis of β2-AR expressing cells and CD positive cells by FCM was measured by gating in the lymphocytes region on the scattered gram. Figure 2 shows an example analysis. Nonspecific reaction of the PE fluorescence was found in the isotype control. Therefore, the real values of the AR expressing cell counts were calculated by subtracting the control values. The CD19+ cells were observed nonspecific reaction, which seems to response of the second antibody. The comparison of each lymphocyte subpopulations before and after hot spring hydrotherapy showed that the CD8+ cell and CD56+ cell counts tended to increase (Figures 5 and 7). However, the significant variation was not seen from the comparison of β2-AR expressing cells before and after hot-spring hydrotherapy. The mean % of β2-AR expressing cells in the lymphocyte subsets was 18% - 19% in CD3+ cells, 5% in CD4+ cells, 57% - 63% in CD8+ cells, and 93% - 95% in CD56+ cells. That in CD19+ cells was approximately 100% (data not shown), but we were not able to be confirmed because it was very likely to be the nonspecific reaction.
We examined the correlation with the rate of change in adrenaline levels and the rate of change in β2-AR expressing cell counts of each subset or that in each CDpositive cell counts before and after hot-spring hydrotherapy. In the CD-positive cells, the rate of change in adrenaline levels was a positive correlation with that in the levels of CD56+ cells, CD8+ cells, and CD3+ cells; in particular, a correlation with CD56+ and CD8+ cells was high (Figure 5). In β2-AR expressing cells, the rate of change in adrenaline levels was a positive correlation with the rate of change in the levels of β2-AR+ CD56+ cells. These results suggested that the variation in adrenaline levels is correlated with CD56+ cells (Figures 6 and 8).
4. Discussion
4.1. Acquired Immunodeficiency in Daily Life
Every creature in the world including human exposes to the risk of immunodeficiency in daily life [1-10]. The factors that influence the acquired immune activity are systemic metabolic disorder such in diabetes meritus, malnutrition, extreme exhaustion and stress, senile and side effect by in cancer. So we have to select daily an appropriate menu to regulate immune function through leukocyte storage. The menu had been summarized and listed as CAM: complementary and alternative Medicine. One of the major menu is TCM in western medicine world, some trying o integrate Western Medicine and Eastern Medicine.
We have been trying to regulate the immune responsiveness through much mature for fragile daily condition from circumstance stress and so on. The main menu were, acupuncture, hot-spring hydrotherapy, light exercise etc. In this article, we would like to show the regulatory mechanism of the hot spring hydrotherapy. The circumstance, the balneotherapy using the effectiveness of hotsprings hydrotherapy, except for cases of contraindication, has been medically useful approved to be effective in many stress-related disorders and the improvement of dysfunction of the biological rhythm disturbance as well as chronic disease. The mechanism of effects has been reported in many studies, but many things are still unclear. Balneotherapy needs to be treated in general a period of time, but the effectiveness has been suggested even if the short duration hot-springs hydrotherapy. We examined the effect of hot spring hydrotherapy for a short duration on immune system and reported about the quantitative and qualitative variation of immuno-competent cells [13,14,18,20]. The mechanism is suggested the association with an autonomic nervous system and the endocrine system. Hot spring hydrotherapy for a short duration was expected to stimulate sympathetic nerve or parasympathetic nerve and to change the levels of catecholamines (adrenalin, noradrenaline, and dopamine), which are neurotransmitters and hormones, as well as the number and function of immune cells.
4.2. The Constitution/Condition Dependent Regulation of Hot Spring Hydrotherapy
Our results showed that within 24 hours after hot spring hydrotherapy, the white blood cells in peripheral blood had changed significantly, not only in cell count but also cell function. We hope that our work will attract more attention to the mechanisms of which hot spring hydrotherapy regulates the human immune system. Abo reported that according to the lymphocyte subset content, lymphocyte rich type showed over 40% on the other hand granulocyte rich type show over 60% of granulocyte [25,26]. Each type exhibited different character even in the same age, sexuality and each age. In the figure, within the same age and the sex, even in mankind can sorts out as G-rich type (granulocyte 60%), and L-rich type (lymphocyte 40%). On the other hand, as a stand point of sex difference, the lady belongs to L-rich type but the gentleman belongs G-rich type. According to the age-related change, G-rich type of young man changes to L-rich type according getting older.
We have been trying to regulate the immune responsiveness through much mature for fragile daily condition from circumstance stress and so on. The main menus were, acupuncture, hot-spring hydrotherapy, light exercise etc. In this article, we would like to show the regulatory mechanism of the hot spring hydrotherapy. The circumstance, the balneotherapy using the effectiveness of hotsprings hydrotherapy, except for cases of contraindication, has been medically useful approved to be effective in many stress-related disorders and the improvement of dysfunction of the biological rhythm disturbance as well as chronic disease. The mechanism of effects has been reported in many studies, but many things are still unclear. Balneotherapy needs to be treated in general a period of time, but the effectiveness has been suggested even if the short duration hot-springs hydrotherapy. We examined the effect of hot spring hydrotherapy for a short duration on immune system and reported about the quantitative and qualitative variation of immuno-competent cells [4,13,14,18,20]. The mechanism is suggested the association with an autonomic nervous system and the endocrine system. Hot spring hydrotherapy for a short duration was expected to stimulate sympathetic nerve or parasympathetic nerve and to change the levels of catecholamines (adrenalin, noradrenaline, and dopamine), which are neurotransmitters and hormones, as well as the number and function of immune cells.
The adrenaline and noradrenaline are secreted by various stress (stimulation); the former from the adrenal medulla and the latter from both the adrenal medulla and the end of sympathetic nerve. They express these functions through the adrenergic receptors (ARs) and regulate the target organ. The adrenergic receptor has two types of α and β, and there are multiple subtypes (α1, α2, β1, β2, β3) [21]. These subtypes present the vascular (α1), the presynaptic terminal (α2), the heart (β1), the bronchial muscle of the lung (β2), fat cells (β3), respectively. The α- receptor stimulation causes bronchodilatation and vasodilatation, the β1-receptor stimulation causes an increase in heart rate and lipolysis, and the β2-receptor stimulation causes bronchoconstriction, vasodilatation, and muscle glycogen resolution. The adrenaline provides heart activation by effect on α- and β-receptors, and the noradrenaline provides blood pressure rises by strongly effect on α-receptors. In addition to the above, it has been reported that the ARs are present on leucocyte membranes [21-24] and that the level of expression of β-ARs in lymphocytes was examined by radioligand (125I-iodopindolol) binding, but the details of subtype of β-receptors have not been disclosed. The subtypes of the AR are able to be analyzed by flow cytometry (FCM) method. In this study, we described how hot spring hydrotherapy influences leukocyte, lymphocyte subpopulations expressing β2-AR and the levels of catecholamine in human peripheral blood.
Abo reported that according to the lymphocyte subset content, lymphocyte rich type showed over 40% on the other hand granulocyte rich type show over 60% of granulocyte. Each type exhibited different character even in the same age, sexuality and each age.
In the figure, within the same age and the sex, even in mankind can sorts out as G-rich type (granulocyte 60%), and L-rich type(lymphocyte 40%).
On the other hand, as a standpoint of sex difference, the ladiy belongs to L-rich type but the man belongs Grich type. According to the age-related change, G-rich type of man changes to L-rich type within the same sex (Figure 11).
4.3. The Factor Circadian Rhythm
In order to assess correctly the changes after the menu, it

Figure 11. The constitution/condition dependent regulation of hot spring hydrotherapy. Abo reported that according to the lymphocyte subset content, lymphocyte rich type showed over 40% on the other hand granulocyte rich type show over 60% of granulocyte. Each type exhibited different character even in the same age, sexuality and different age of the same individual. In the figure, within the same age and the sexuality, even in gentleman can sorts out as G-rich type (granulocyte ≧60%), and lady can do L-rich type (lymphocyte ≧40%). Within a same individual G-rich type moves to L-type by getting older.
is important to conscious of circadian rhythm. Abo reported that it was possible to sort the constitution, granulocyte-rich individual and lymphocyte-rich one with the peripheral leukocyte [25,26].
Each population of subset is depending on a circadian rhythm. Within a same individual, granulocyte increase in the daytime, on the other hand, lymphocyte increased in the nighttime in a cycle 24 hrs. So we have to compare the effect of each menu for the peripheral leukocyte on the same time before and after the menu.
It were reported that the leukocyte subset, granulocyte and lymphocyte regulated by various factors. One major point is that they are regulated by autonomous nervous system, resulting in circadian rhythm [25,27]. Therefore, in order to access the effect within a short time, it is necessary to consider this factor to adjust the time to collect the sample. For example, efficacy and impact of walking exercise have been widely recognized. However, the majority of walkers did not have a scientific background to know the best exercise menu for the one QOL; quality of life. The purpose of this study was to demonstrate the best menu of walking that regulates the peripheral white blood cells in number and function as a marker of QOL expression.
4.4. Suggested Expression of Efficacy in Each Menu for Comparison
Traditionally, each heat therapy has its own character and efficacy for various complaints. Through the years, each water source was evaluated for its specific properties and with the advent of better transportation in our mountainous land, even remote springs in the mountains were visited for their specific medicinal effect. For example, the hot spring of Fukatani, which is located just below our research facility, was known all over the area to cure hemorrhoids. However, almost all the judgments of efficacy are VAS (visual analog scale). The proprietors of the hot spring inns say that many of the guests came to cure that sort of ailment since about 20 years ago. Now in Japan, hot spring hydrotherapy is often used as a supplementary therapy for many diseases [28-30]. It has been shown to reduce surgical complaints such as shoulder pain, amyloidosis and various rheumatic problems. It can also lighten the burden to the heart and improve the condition of patients who suffer from emphysema as well as other respiratory ailments. Hot springs have also traditionally functioned as places to relax and enjoy oneself, even though resort-type hot springs always exist along with those for illnesses. In recent years, trends have been seen that even remote hot springs, such as the one below our facility, transform into fashionable resorts for the healthy to visit for relaxation and stress release. It is interesting to note that this historical duplicity encompassed by hot springs has also entered the world of Western medicine as the release of mental stress and physical fatigue has been shown to be essential for good health [31- 33]. Put into other words: the relaxation side of the hot spring promotes prevention, including health enhancement, and the medicinal side, the treatment of complains and illness [13-20].
5. Conclusions
We have measured the number of leukocyte subsets, granulocyte and lymphocyte ratio after hot spring hydrotherapy. The conclusions were as following:
1) Hot spring hydrotherapy for a short duration proved to regulate the total number of peripheral leukocyte.
2) Hot spring hydrotherapy for a short duration proved to regulate the ratio of leukocyte subsets, granulocyte/ lymphocyte.
3) The emotional hormones, adrenaline & dopamine also regulate in the serum level.
4) The mode of change was constitution/condition dependent manner. The adrenalin and dopamine were also regulated by heat therapy.
5) The quantitative effect of hot spring hydro therapy on the regulatory effects of leukocyte and β2-adrenergic receptors (β2-ARs) positive lymphocyte.
6) It was also proved by the β2-AR of lymphocyte subsets by combining with fluorescent monoclonal antibodies (CD3, CD4, CD8, CD19, and CD56) for flow-cytometry and changed correlatively to adrenaline level.
7) The expression is possible as linear function and slant.
REFERENCES
- S. Kurashige, T. Yoshida and S. Mitsuhashi, “Immune Responsein Sarcoma 10-Bearing Mice,” Annual Report of Gunnma University, Vol. 1, 1980, pp. 36-44.
- S. Miyazaki, “Immunodificiency in Clinical Origin,” Clinical Pediatrics, 1977, pp. 1001-1006.
- K. Kishida, S. Miyazaku, H. Take, T. Fujimoto, H. Shi, K. Sasaki and N. Goya, “Granial Irradiation and Lymphocyte Subpopulation in Acute Lymphatic Leukemia,” Journal of Pediatrics, Vol. 92, No. 5, 1978, pp. 785-786. doi:10.1016/S0022-3476(78)80155-3
- N. Yamaguchi, T. Takei, R. Chen, P. Wushuer and W. Hsin Wu, “Maternal Bias of Immunity to Her Offspring: Possibility of an Autoimmunity Twist out from Maternal Immunity to Her Young,” Open Journal of Rheumatology and Autoimmune Diseases, Vol. 3, No. 1, 2013, pp. 40-55. doi:10.4236/ojra.2013.31008
- R. A. Murgita and T. B. Tomasi Jr., “Suppression of the Immune Response by Alpha-Fetoprotein,” The Journal of Experimental Medicine, Vol. 141, No. 2, 1975, pp. 269- 286. doi:10.1084/jem.141.2.269
- G. Paul, S. Margaret, Y. F. Liew and M. M. Allan, “CD4+ but Not CD8+ T Cells Are Required for the Induction of Oral Tolerance,” International Immunology, Vol. 7, No. 3, 1995, pp. 501-504. doi:10.1093/intimm/7.3.501
- H. Koshimo, H. Y. Miyazawa, Y. Shimizu and N. Yamaguchi, “Maternal Antigenic Stimulation Actively Produces Suppressor Activity in Offspring,” Developmental & Comparative Immunology, Vol. 13, No. 1, 1989, pp. 79-85. doi:10.1016/0145-305X(89)90020-7
- M. Zoeller, “Tolerization during Pregnancy: Impact on the Development of Antigen-Specific Help and Suppression,” European Journal of Immunology, Vol. 18, No. 12, 1988, pp. 1937-1943. doi:10.1002/eji.1830181211
- R. Auerback and S. Clark, “Immunological Tolerance: Transmission from Mother to Offspring,” Science, Vol. 189, No. 4205, 1975, pp. 811-813. doi:10.1126/science.1162355
- S. Shinka, Y. Dohi, T. Komatsu, R. Natarajan and T. Amano, “Immunological Unresponsiveness in Mice. I. Immunological Unresponsiveness Induced in Embryonic Mice by Maternofetal Transfer of Human-Globulin,” Biken Journal, Vol. 17, No. 2, 1974, pp. 59-72.
- J. M. Aase, G. R. Noren, D. V. Reddy and J. W. Geme Jr., “Mumps-Virus Infection in Pregnant Women and the Immunologic Response of Their Offspring,” The New England Journal of Medicine, Vol. 286, No. 26, 1972, pp. 1379-1382. doi:10.1056/NEJM197206292862603
- D. V. Cramer, H. W. Kunz and T. J. Gill III, “Immunologic Sensitization Prior to Birth,” American Journal of Obstetrics & Gynecology, Vol. 120, No. 3, 1974, pp. 431- 439.
- X.-X. Wang, Kitada, K. Matsui, S. Ohkawa, T. Sugiyama, H. Kohno, S. Shimizu, J.-E. Lai, H. Matsuno and N. Yamaguchi, “Variation of Cell Populations Taking Charge of Immunity in Human Peripheral Blood Following Hot Spring Hydrotherapy Quantitative Discussion,” The Journal of Japanese Association of Physical Medicine, Balneology and Climatology, Vol. 62, 1999, pp. 129134.
- H. Matsuno, X.-X. Wang, W. Wan, K. Matsui, S. Ohkawa, T. Sugiyama, H. Kohno, S. Shimizu, J. E. Lai and N. Yamaguchi, “Variation of Cell Populations Taking Charge of Immunity in Human Peripheral Blood Following Hot Spring Hydrotherapy Qualitative Discussion,” The Journal of Japanese Association of Physical Medicine, Balneology and Climatology, Vol. 62, 1999, pp. 135-140.
- N. Yamaguchi, H. Hashimoto, M Arai, S. Takada, N. Kawada, A. Taru, A.-L. Li, H. Izumi and K. Sugiyama, “Effect of Acupuncture on Leukocyte and Lymphocyte Subpopulation in Human Peripheral Blood-Quantitative Discussion,” The Journal of Japanese Association of Physical Medicine, Balneology and Climatology, Vol. 65, No. 4, 2002, pp. 199-206.
- W. Wan, A.-L. Li, H. Izumi, N. Kawada, M. Arai, A. Takada, A. Taru, H. Hashimoto and N. Yamaguchi, “Effect of Acupuncture on Leukocyte and Lymphocyte Subpopulation in Human Peripheral Blood Qualitative Discussion,” The Journal of Japanese Association of Physical Medicine, Balneology and Climatology, Vol. 65, No. 4, 2002, pp. 207-211.
- X.-X. Wang, S. Katoh and B.-X. Liu, “Effect of Physical Exercise on Leukocyte and Lymphocyte Subpopulations in Human Peripheral Blood,” Cytometry Research, Vol. 8, 1998, pp. 53-61.
- Y. Kitada, W. Wan, K. Matsui, S. Shimizu and N. Yamaguchi, “Regulation of Peripheral White Blood Cells in Numbers and Functions through Hot-Spring Bathing during a Short Term Studies in Control Experiments,” Journal of Japanese Society Balneology Climatology Physiological Medicine, Vol. 63, 2000, pp. 151-164.
- N. Yamaguchi, T. Takahashi, T. Sugita, K. Ichikawa, S. Sakaihara, T. Kanda, M. Arai and K. Kawakita, “Acupuncture Regulates Leukocyte Subpopulations in Human Peripheral Blood,” eCAM, Vol. 4, No. 4, 2007, pp. 447- 453.
- N. Yamaguchi, S. Shimizu and H. Izumi, “Hydrotherapy Can Modulate Peripheral Leukocytes: An Approach to Alternative Medicine, Complementary and Alternative Approaches to Biomedicine,” Kluwer Academic/Plenum Publishers, New York, 2004, pp. 239-251.
- D. B. Bylund, D. C. Eikenberg, J. P. Hieble, S. Z. Langer, R. J. Lefkowitz and K. P. Minneman, “Intenational Union of Pharmacology Nomenclature of Adrenoceptors,” Pharmacological Review, Vol. 46, No. 2, 1994, pp. 121-136.
- B. H. Dulis and I. B. Wilson, “The β-Adrenergic Receptor of Live Human Polymorphonuclear Leukocytes,” Journal of Biological Chemistry, Vol. 255, 1980, pp. 1043-1048.
- A. S. Maisel, T. Harris, C. A. Rearden and M. C. Michel, “Beta-Adrenergic Receptors in Lymphocyte Subsets after Exercise. Alterations in Normal Individuals and Patients with Congestive Heart Failure,” Circulation, Vol. 82, 1990, pp. 2003-2010. doi:10.1161/01.CIR.82.6.2003
- V. M. Sanders, R. A. Baker, D. S. Ramer-Quinn, D. J. Kasprowicz, B. A. Fuchs and N. E. Street, “Differential Expression of the β2-Adrenaergic Receptor by Th1 and Th2 Clones,” Journal of. Immunology, Vol. 158, No. 9, 1997, pp. 4200-4210.
- T. Abo, T. Kawate, K. Itoh and K. Kumagai, “Studies on the Bioperiodicity of the Immune Response. 1. Circadian Rhythms of Human T, B and K Cell Traffic in the Peripheral Blood,” Journal of Immunology, Vol. 126, 1981, pp. 1360-1363.
- T. Abo and T. Kumagai, “Studies of Surface Immunoglobulins on Human B Lymphocytes. Ⅲ. Physiological Variations of Sig+ Cells in Peripheral Blood,” Clinical Experimental Immunology, Vol. 33, 1978, pp. 441-452.
- S. Suzuki, S. Toyabe, T. Moroda, T. Tada, A. Tsukahara and T. Iiai, “Circadian Rhythm of Leukocytes and Lymphocytes Subsets and Its Possible Correlation with the Function of the Autonomic Nervous System,” Clinical Experimental Immunology, Vol. 110, No. 3, 1997, pp. 500-508. doi:10.1046/j.1365-2249.1997.4411460.x
- J. R. Ostberg, R. Patel and E. A. Repasky, “Regulation of Immune Activity by Mild (Fever-Range) Whole Body Hyperthermia: Effect on Epidermal Langerhans Cells,” Cell Stress Chaperones, Vol. 5, No. 5, 2000, pp. 458-461. doi:10.1379/1466-1268(2000)005<0458:ROIABM>2.0.CO;2
- Y. H. Huang, A. Haegerstrand and J. Frostegard, “Effect of in Vitro Hyperthermia on Proliferative Responses and Lymphocyte Activity,” Clinical Experimental Immunology, Vol. 103, No. 1, 1996, pp. 61-66. doi:10.1046/j.1365-2249.1996.00932.x
- H. Mats, E. Orion and R. Wolf, “Balneotherapy in Dermatology,” Dermatological Therapy, Vol. 16, No. 2, 2003, pp. 132-140. doi:10.1046/j.1529-8019.2003.01622.x
- L. J. Ignarro and C. Colombo, “Enzyme Release from Polymorphonuclear Leukocyte Lysosomes: Regulation by Autonomic Drugs and Cyclic Nucleotides,” Science, Vol. 180, No. 4091, 1973, pp. 1181-1183. doi:10.1126/science.180.4091.1181
- I. J. Elenkov and G. P. Crousos, “Stress Hormones, Th1/ Th2 Patterns, Pro/Anti-Inflammatory Cytokines and Susceptibility to Disease,” TEM, Vol. 10, 1999, pp. 359-368.
- R. M. A. Landmann, F. B. Muller, C. Perini, M. Wesp, P. Erne and F. R. Buhler, “Changes of Immunoregulatory Cells Induced by Psychological and Physical Stress: Relationship to Plasma Catecholamines,” Clinical Experimental Immunology, Vol. 58, 1984, pp. 127-135.
Abbreviations
ACTH: Adrenocorticotropic hormone is secreted by pituitary gland, located below the brain;
AR: Adrenaline receptor;
β2-AR+: β2 Adrenaline receptor positive lymphocyte;
β2-AR-: β2 Adrenaline receptor negative lymphocyte;
CAM: Complementary and alternative medicine, beside the western medicine, there are many traditional medicine and/or health promoting menu all over the world;
CD: Cluster of differentiation. Each lymphocyte has name that expressed CD number, for example CD2, CD4, etc.;
FCM: Flow cytometry;
G-rich type: The individual that exhibit over 60% of granulocyte in peripheral blood, finding many in young gentleman;
L-rich type: The individual that exhibit over 40% of lymphocyte in peripheral blood, finding lot in ladies and senile;
METS: Metabolic rate(s);
T3: This hormone has another name triiodothyronine, This one acts in energy management and a variety of biochemical and metabolic reactions and functions;
T4: Thyroxine known as T4, This hormone concerns in energy management and a variety of biochemical and metabolic reactions and functions;
TCM: Traditional Chinese medicine;
QOL: Quality of life;
VAS: Visual analog scale.
NOTES
*Conflict of Interests: No conflict of interest exists in this work.
#Corresponding author.