Structural and Surface Morphology Analysis of Nickel Phthalocyanine Thin Films ()
1. Introduction
The materials considered of most importance are the phthalocyanines in the organic dyes and pigments. Phthalocyanines have found applications as semiconducting devices, photovoltoic cells, rectifying devices, liquid crystal displays and gas sensors. The electrical, optical and structural properties of phthalocyanine thin films are dependent on various parameters such as evaporation rate, substrate temperature and post-deposition annealing [1]. Phthalocyanines are found in different phases i.e. α-, β- and γ-phases. Metal-substituted and metal free phthalocyanines are readily used as active layer in several applications such as gas sensors, photocapacitive and photoresistive detectors, Organic Thin Film Transistors (OTFTs), colour filters and organic laser materials. Among the various metal-substituted phthalocyanines, nickel phthalocyanine has received considerably less attention. In the present study, we report the structural properties of nickel phthalocyanine thin films prepared at different substrates temperatures. To explore the potentially interesting properties of the material, structural and morphological characterization is thus considered as a prerequisite for deep insight knowledge of the material. These structural properties mostly depend on the deposition technique, the heat-treatment temperature and the conditions during film deposition. Annealing process is a broadly employed method to enhance the quality of the crystal and exploit structural defects in the material. During thermal annealing, the morphology and structural properties of the material change [2-4]. For semiconductor devices like light emitting diodes the structural properties are very important, therefore, it is essential to investigate how thermal annealing and substrate temperatures process affects these properties [5,6]. In the present work, thin films of NiPc have been fabricated on glass substrates by vacuum evaporation techniques. A detailed study on the effects of heating substrate temperatures on the structural properties and surface morphology has been performed by X-ray diffraction and atomic force microscopy (AFM). Hence, the purpose of our work is to find the optimal temperature for which the film produces best structural properties.
2. Experimental
The NiPc powder used in this study was obtained from Aldrich chemical company and was used as the source material for thermal evaporation. Its molecular structure is shown in Figure 1. Before starting the deposition, the glass substrate was cleaned in an ultrasonic bath for 10 min using acetone, followed by rinsing in distilled water.
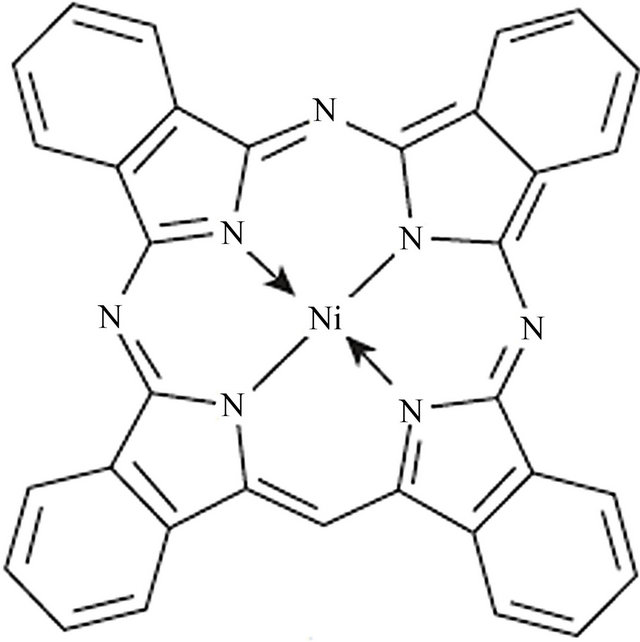
Figure 1. Molecular structure of Ni-phthalocyanine (NiPc).
The substrate was dried in open air in a cleaned room. A molybdenum boat was used as a heating source. Thin films of NiPc, were deposited by vacuum evaporation technique on thoroughly cleaned glass substrate at different substrate temperatures (300, 325, 350, 400, 450) K using Edwards 306 system. During deposition, the pressure in the vacuum chamber was kept constant at about 10−5 mbar and the deposition rates of all the films were kept at about 0.2 nm/s, Also the substrate are placed at a distance of 15 cm from the source. The thickness of the films was measured using Michelson interferometer technique. The thickness of the films was 265 nm. The adhesion of the films to the substrate seems to be extremely good. The structural analysis of films is performed by X-ray diffractometer with Cu Kα radiation (λ = 1.54 Å). The surface morphology of the NiPc films at different substrate temperatures (Ts) is investigated by means of atomic force microscopy (AFM).
The lattice parameters of the films were calculated using the Bragg’s formula:
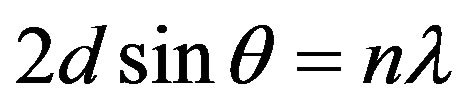 (1)
(1)
The grain size of the crystallites were calculated from the XRD using Scherer’s relation,
 (2)
(2)
Where K = 0.94 is a constant, λ the wavelength of X-ray, β the full width half maximum and θ the diffraction angle. The micro strain (ɛ) and the dislocation density (δ) of films were estimated using the equations
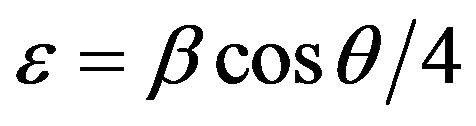 (3)
(3)
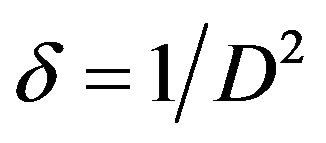 (4)
(4)
3. Results and Discussions
The most common polymorphic crystal phases occur in Ni-phthalocyanines are α, and β, that have two crystallite structures. The α phase is metastable and obtained either as a polycrystalline powder or as a thin film deposited on a cold substrate in vacuum [7]. The phase β can be obtained in single crystal form or as a thin film formed by deposition of phthalocyanine on heated substrates, which are thermodynamically stable. Phase separation could be responsible for the de gradation of the optical properties. The α form is metastable and can be converted to β form by temperature treatments with structures tetragonal and monoclinic respectively. The differences in the d values are attributed to higher X-ray absorption, sample purity, particle size, preferred orientation and crystal texture [4, 7].
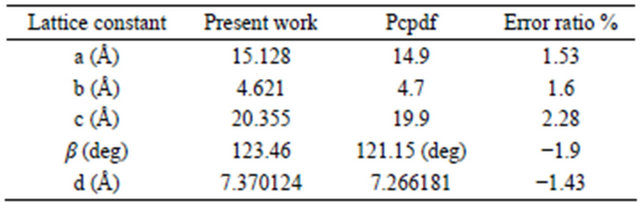
The X-ray diffraction of the NiPc powder used as the source material for sublimation is given in Figure 2. The peaks are identified using the standard JCPDS File No. 11-0744 data. The preferential orientation is found to be in the (100) direction at 2θ = 6.938˚ and that it has an α- crystalline phase with monoclinic system with lattice constants a = 1.513 nm, b = 0.462 nm, c = 2.03 nm and β = 123.46˚. The close agreement showed that the powder is in the monoclinic structure as reported for other phthalocyanines [1-7]. Table 1 gives the lattice constant of present observed and the standard data of NiPc.
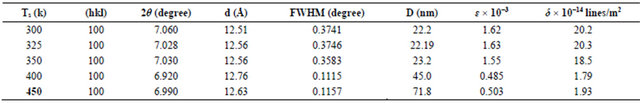
X-ray diffraction pattern of NiPc thin film of thickness (265 nm) prepared at different substrate temperatures (300, 325, 350, 400, 450) K are shown in the Figure 3 respectively. XRD pattern indicates that all the samples are single phase and that it has a β-crystalline phase [4-8]. The difference between β- and α-phases is attributed to the tilt angle of b-axis of the unit cell. The structure of the NiPc thin film is determined as tetragonal with preferential orientation along the (100) direction. Well-defined diffraction peaks by (100) gives the direction of the preferential orientation as deposited film (JCPDS, File No. 11-0744). The preferential orientation is at 2θ = 6.859˚. The spacing between the planes, d = 12.876 Å. The d values obtained from diffractogram are in perfect match with JCPDS record. The diffraction peaks are well matched with the previous observations [1-8]. The increased crystallinity is attributed to the destruction of pseudomorphic layers present in the film upon high substrate temperature. Table 2 shows the structure parameters of NiPc films, The full width at half maximum (FWHM) intensity of preferential orientation, shows that the FWHM are reduced with increasing substrate temperatures, which represents better lattice quality [1,8-10].
From the Table 2 it is observed that the grain size increases which intern decreases strain and dislocation density. The grain size D of the film is calculated using the Scherrer relation, the grain size increases from (22.2 - 71.8 nm) for substrate temperatures (300 - 450 K). This is because the pseudomorphic layers formed at room temperature, which are in the metastable state, are destroyed by increasing the substrate temperatures and lar-

Figure 2. X-ray diffraction of the NiPc powder.
Table 1. The present observed and the standard data NiPc.
ger crystallites are formed or this can be attributed to the onset of phase transition occurring in NiPc. Also this may be attributed to a phase transition or to the regrowth of phthalocyanine. This mean that the crystallization of the films is a function of substrate temperatures and the optimal temperature for which the film produces best structural properties is at 400 - 450 K. Also from Figure 3 two distinct peaks (200) and (314) are visible at high diffraction angles for heating substrate 400 and 450 K respectively, and this is agreement with other literatures [6-9], and this is may be attributed to enhancement and recrystallization these films by increasing the substrate temperatures.
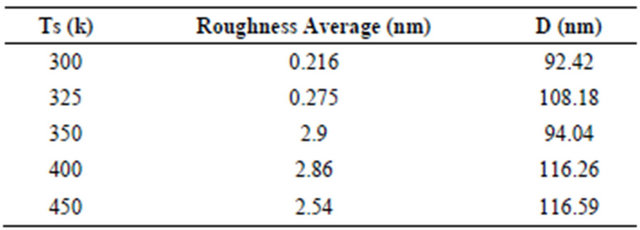
Surface morphology of NiPc thin film is shown in Figures 4 and 5 which prepared at different substrate temperatures (300, 325, 350, 400, 450) K. It shows that the morphology of the NiPc thin film has larger number of grain size and are homogeneously distributed, which indicates the crystalline nature of the film. It is obvious because the films were prepared at different substrate temperatures, due to the heating grain growth will takes place which gives the crystallinity and good surface morphology [2]. These results are in good agreement with XRD characteristics. The crystal morphology and molecular orientation change with substrate temperature. The average grain size increases as temperature increases. Gas sensitivity depends both up on material parameters, such as the crystal phase of the material and film parameters, such as the surface topography of the film. These structural characterizations confirm that the substrate temperature allows us to control the structural organization of nickel phthalocyanine molecules in their solid state, from which a large improvement of the properties can be expected. Table 3 shows that the grain size increased by increasing the substrate temperature.
The morphology of these films, has also confirmed the appearance of polycrystallites and increasing grain size when substrate temperature is raised. This is caused due to the nucleating behavior of the substance [7]. The root mean square (rms) roughness of the film at different substrate temperatures are shown in Table 3 which increases with increasing Ts. The best results can be seen for the film prepared at heating substrate of 400 - 450 K. The NiPc thin films, after increasing substrate temperatures to 400 - 450, show relatively rough and non-uniform surface, suggesting the formation of interpenetrating molecular network of NiPc. 400 - 450 K and above, deterioration feature is observed due to degradation of the film. From the application point of view, the higher efficiency devices are obtained from the film of greater roughness.
4. Conclusions
Thin films of NiPc were fabricated by the thermal
Table 2. Shows the structure parameters of NiPc films.
evaporation technique on glass substrates with thickness (265 nm) prepared at different substrate temperatures successfully. The outcome of this investigation can be summarized as follows:
• X-ray diffraction patterns obtained for NiPc powder showed α-polycrystalline phase with monoclinic system, while the thin film of thermally evaporated NiPc showed it has a single crystalline β-phase film oriented preferentially (100) plane as tetragonal. It can be seen that the intensity, d value and sharpening of the main peak increase with substrate temperature which shows crystallization of the films when substrate temperature increases.
• Diffraction peaks obtained for the films prepared at higher substrate temperatures are sharp and that is due to the increased crystallinity.
• The full width at half maximum (FWHM) intensity of preferential orientation shows that the FWHM are reduced with increasing substrate temperatures and the grain size increases which intern decreases strain and dislocation density, which represents better lattice quality.
• The grain size increases from (22.2 - 71.8 nm) for substrate temperatures (300 - 450 K), this can be attributed to the onset of phase transition occurring in NiPc. Also this may be attributed to a phase transition or to the regrowth of phthalocyanine. This mean that the crystallization of the films is a function of substrate temperatures and the optimal temperature for which the film produces best structural properties is at 400 - 450 K.
• Rearrangements of molecules are taking place with different substrate temperatures. The change in grain size shows that the properties of NiPc are controlled by its structure and morphology depending on increases the substrate temperatures.
• By analyzing the AFM topographic images, we found that there is a significant influence of substrate temperatures on the surface morphology of NiPc films and the crystal morphology and molecular orientation
Table 3. Shows the grain size and roughness of NiPc.
change with substrate temperature. These films showed a larger number of grain size, homogeneously distributed, rough which indicates the crystalline nature of the film. The surface roughness of Ni-Pc thin films is increased by increasing substrate temperatures, which makes it useful for higher efficiency devices. The increase in the temperature beyond 400 K has higher roughness and grain size.
• These structural characterizations confirm that the substrate temperature allows us to control the structural organization of nickel phthalocyanine molecules in their solid state, from which a large improvement of the properties by increasing the substrate temperatures, therefore, the best results can be seen for the film prepared at heating substrate of 400 - 450 K. These results are in good agreement with XRD characteristics.
5. Acknowledgements
The work was supported by Department of Physics college of Science, thin films and opto-electronic laboratories, Baghdad University.