Effect of Surfactant Concentration on Aqueous LiBr Solution Absorption Rate ()
1. Introduction
From the beginning of the 19th century, absorption cooling systems attracted increasing interest, since it is possible to recover energy by using waste heat and thermal solar energy with these systems for cooling applications. Absorption chillers differ from the more prevalent compression chillers in the way that the cooling effect is driven by heat energy rather than mechanical energy. Vapour absorption systems have been less popular than vapour compression systems because of higher cost and lower efficiency.
It is well known that the vapour absorption rate into the absorbent solution is the key process to improve the thermal coefficient of performance (COP) of vapour absorption air conditioning systems. The enhancement of the absorption process leads to reducing the heat exchanger size of the absorber.
For the continuous control of any absorption system, it is important to monitor the properties of the binary solution at different points in the cycle and under different refrigeration loads.
Various techniques are available to measure the concentration of the binary water/lithium bromide solution. The most commonly used method is the titration method [1], which can give a high accuracy when the procedure is performed by an experienced and careful researcher. The drawback of this method is that it tends to be time consuming, as it requires drawing a sample from the system solution.
The continuous measurement of concentration using radioactive tracer detectors or mass spectrometers is very expensive [2].
This study describes an accurate procedure for obtaining the concentration of the aqueous lithium bromide solution based on the measurement of electrolyte conductivity.
The study also presents an experimental investigation to study the effect of the surfactant concentration on the absorption rate.
2. Measurement Techniques
The working ranges for vapour absorption systems based on water/lithium bromide are (40% - 65%) for the concentration of the lithium bromide in the binary solution and (20˚C - 100˚C) for the temperature of the solution. These are the ranges investigated in this study. The concentration samples of the binary solutions are prepared in a glass beaker and immersed in a constant temperature water bath. A commercially available conductivity meter with a built-in temperature probe is then used to measure the density and conductivity of the solution [3].
2.1. Sample Preparation
Lithium Bromide is a white cubic crystal or crystalline powder. It is apt to decompose in the moist air, dissolve into water, alcohol or glycol, and able to dissolve into ether or pentanol to become colourless aqueous solution or pale yellow transparent liquid.
As the lithium bromide is extremely hygroscopic [4], it is important to follow a working procedure to ensure that no moisture from the atmosphere is absorbed unintentionally when preparing the measuring sample. The samples were prepared from a pure dry lithium bromide powder and deionised water. A digital mass balance was used to measure the masses of the lithium bromide and the deionised water before mixing.
2.2. Temperature Measurement and Control
The reaction of the lithium bromide powder and water is exothermic; this produces a significant rise in the temperature of the solution (20˚C to 60˚C). The temperature of the solution was controlled by a thermostatically regulated water bath to the desired value [3].
The temperature of the solution was measured using the conductivity meter with the built-in temperature probe.
2.3. Conductivity Measurement
For a cell of a uniform cross section (A), with electrodes at either end separated by distance (l), the conductivity (K) is related to the conductance (G), by Equation (1) and has the units Micro-Siemens (W−1∙m−1 or Sm−1) [5].
 (1)
(1)
2.4. Apparatus
In principal, the measurement of the conductivity could be carried out in a cell, with rectangular electrodes of known area A m2 positioned l m apart. However, in practice a number of complicated corrections would have to be made for getting the exact value of the conductivity. Rather than doing this for all measurements, use is now made of definitive conductivity measurements for certain standard solutions carried out under very carefully controlled conditions in specially designed cells.
An extensive and thorough survey of the market led the author to conclude that Omega bench-top CDB-420 conductivity-meters were the most suitable. Conductivity span ranges from 0 to 19.99 S with resolution 0.01 S and accuracy ±0.5%, the temperature range is −10˚C to 105˚C with accuracy ±0.5˚C. It is also concluded that the epoxy conductivity cell model CDE-430-10-EP with constant (K = 10) and built-in temperature probe is the most suitable probe to link with the conductivity meter.
2.5. Calibration
A sample of known electrolyte conductivity (e.g. 0.745 g/litre Potassium Chloride solution) has been used to calibrate the instrument. Dissolving 0.745 grams of dried Analar Grade Potassium Chloride (KCl) into 1 litre of deionised water. The solution has been placed into a water bath set to 25˚C. When the temperature of the calibration standard solution has reached steady state, the electrode has been placed in the solution and left for 5 - 10 minutes. The range of 0 - 2000 m Siemens on the conductivity meter has been selected, and then the instrument reading has been adjusted to 1413 m Siemens.
3. Data Correlation and Empirical Modelling
The experimental data presented in Table 1 shows that for a constant concentration, the conductivity (m Siemens) decreases with temperature (˚C) for concentrations up to 55% LiBr. For concentrations higher than 55%, the conductivity increases as the temperature increases.
In order to develop an empirical model of the concentration as a function of temperature and conductivity of the aqueous lithium bromide solution, numerical curve fits have been produced for the experimental points of conductivity versus temperature at particular concentrations. The best-fit equations are logarithmic of the form
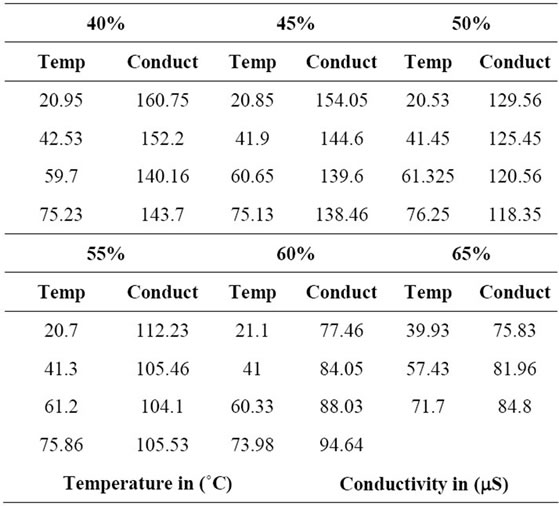
Table 1. Electrolyte Conductivity for different concentrations.
given by Equation (2).
 (2)
(2)
The values of the constants ai and bi are given in Table 2 for different concentration Xi%.
An equation is then constructed using the above results and equation to produce a relationship giving the concentration of the lithium bromide solution {X%} as a function of temperature {T, ˚C} and electrolyte conductivity {C, mS}. This is presented in Equations (3)-(6).
 (3)
(3)
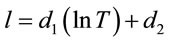 (4)
(4)
 (5)
(5)
 (6)
(6)
where d1 = 409.66, d2 = −1677.2, d3 = −297.03, d4 = 942.01, d5 = 37.673, d6 = 103.56.
Figure 1 presents the experimental data of electrolyte conductivity of the aqueous lithium bromide versus the temperature. These experimental data agree with the trends presented by Hamann et al., 1998 [5]. However, at higher concentrations, the conductivity rises less rapidly than expected from the extrapolation of the results; this is due to the increase of the inter-ionic interactions as the mean distance between ions decreases.
4. Experimental Test Rig
The general arrangement of the experimental apparatus used in the present study is shown in Figure 2.
The measurements of various parameters were taken in different positions of the test rig. The variables measured and controlled during the operation of the system were the temperature, pressure and concentration. Concentration was measured using Electrolyte Conductivity-Temperature technique.
4.1. Working Fluids
4.1.1. Absorbent Solution
An aqueous solution of lithium bromide containing about 61% lithium bromide is used initially as an absorbent. The working solution was prepared from pure dry lithium bromide powder 99.9% “Sigma-Aldrich product” and pure deionised water.
A digital mass balance was used to measure the masses of the lithium bromide and the deionised water before mixing. As the reaction of the water and the powder is exothermic, a binary solution of 61% (LiBr-Water) was prepared and left to cool to the ambient temperature.
4.1.2. 1-Octanol
1-Octanol is used in the experimental investigation for this work. The product was bought from “Sigma Aldrich Co. Ltd.”. The Molecular formula for the 1-Octanol is: C8H18O. This product is also known as Capryl alcohol, n-Octyl alcohol, Heptyl carbinol or 1-Hydroxyoctane. It has the following identifications and properties:
Molecular weight: 130.23 Appearance: Clear, colourless liquid.
Solubility: Slightly soluble, 540 mg/L soluble in water Boiling point: 195˚C.
Melting point: −16˚C

Table 2. Constants ai and bi for Equation (2).

Figure 1. Water/Lithium bromide temperature vs. conductivity for different concentrations.

Figure 2. Measurement and control diagram of the test rig.
Specific gravity: 4.5 g/L Flash point: 81˚C.
4.1.3. Refrigerant
Pure de-ionised water was used as the refrigerant in the evaporator vessel.
5. Purpose of the Experiment
The processes in a LiBr absorber are coupled heat and mass transfer processes. As the water vapour is absorbed into the liquid, the latent heat associated with the phase change is released along with the heat mixing. This energy release occurs at the vapour-liquid interface and causes the interface to be the location of highest temperature in the system. To allow the process to continue, the energy must diffuse into the bulk of the liquid. These two diffusion processes are coupled in the sense that stopping either process causes system variables to adjust in such a way as to stop the other process. For the overall transfer to take place, both processes have to proceed in tandem.
In the case of the absorber, the mass transfer process controls the coupled transfer. Thus, design efforts to improve overall absorber performance is focused on the mass transfer process [2]. Several additives have been tested in the literature. Cul et al., (1991) tested the impact of several additives in the absorption. 2-ethyl-hexanol, n-Octanol such as 1-Octanol and 2-Octanol, decanol, 1- heptanol, soluble alcohol as 2-methyl-2propanol, fluorinated surfactant such as FC-430, and Ion extraction such as 15-crown 5 have been tested. The results showed that the 1-Octanol has the best mass of water vapour absorbed during a 15 minutes run by Lithium bromide solution of initial concentration of 60%. 1-Octanol has been found to improve the absorber performance.
Experiments were performed by changing the number of droplets of 1-Octanol, consequently the distance between the droplets. This in turn leads to the change of the aspect ratio given by the ration of absorbent solution depth to the distance between the additive droplets.
Figure 3 shows the different additives positions of the additive droplets. The additive droplets were located using specially designed disc (180 mm diameter) with number of holes to allow the additives to be in the desired positions as shown in the figure. The 1-Octanol droplets were positioned on the surface of the aqueous lithium bromide solution using a very fine pipette.