1. INTRODUCTION
Clopidogrel, a second-generation thienopyridine, is a prodrug that requires enzymatic biotransformation into its active thiol metabolite before interacting with the P2Y12 receptor on platelets. Pharmacokinetic, pharmacodynamic, and genetic studies have provided abundant evidence that in vivo bioactivation of clopidogrel is a twostep process involving the cytochrome P450 (CYP) system. The isoenzyme CYP2C19 was found to play a major role, because it contributes to both clopidogrel bioactivation steps [1].
A common genetic variant within the CYP2C19 gene, the CYP2C19*2 loss-of-function polymorphism, was found to be associated with an attenuated response to clopidogrel [2-5], which resulted in worse clinical outcomes for patients undergoing coronary stenting [6,7] or with acute myocardial infarction (AMI) [8]. Poor prognosis was also demonstrated in a genome-wide association study [9] and in randomized clinical trials [10]. Although a 2012 meta-analysis questioned the relevance of the CYP2C19 loss-of-function alleles in the prediction of major cardiovascular events beyond stent thrombosis [11] a previous 2010 collaborative meta-analysis demonstrated significant association between carriage of the CYP2C19*2 allele and ischemic events [12].
The recently discovered allelic variant CYP2C19*17 results in increased CYP2C19enzyme activity because of the mutation (−806 C > T) in the 5’ flanking region of the gene, which causes increased transcription of the enzyme [13]. Carriage of CYP2C19*17 was significantly associated with enhanced response to clopidogrel and an increased risk of bleeding in the context of percutaneous coronary intervention [14], but was also found to confer protection against ischemic events in patients after AMIs [15]. Moreover, it has also been reported that combinations of the alleles CYP2C19*2 and CYP2C19*17 have significant effects on platelet aggregation [3], although there have been no reports on prognosis regarding ischemic events after an AMI. We therefore undertook a study of patients carrying combinationsof CYP2C19 *2 and CYP2C19*17 variant alleles, to determine mediumterm outcomes after an acute coronary syndrome (ACS).
2. MATERIAL AND METHODS
2.1. Study Design and Eligibility
This was a prospective longitudinal selection of 220 consecutive patients admitted for an ACS between April and October 2009, who survived hospitalization. Eligible patients were younger than 75 years of age and were prescribed clopidogrel (75 mg/day) and acetylsalicylic acid (100 mg/day) at discharge.
Patients were excluded if they were immediately referred to surgery, or if they were enrolled in 2 other clinical trials investigating antiplatelet therapy: The TRA•CER (Trial to Assess the Effects of SCH 530348 in Preventing Heart Attack and Stroke in Patients With Acute Coronary Syndrome—ClinicalTrials.gov identifier: NCT00527943) [16] and the TRILOGY ACS (A Comparison of Prasugrel and Clopidogrel in Acute Coronary Syndrome Subjects—ClinicalTrials.gov identifier: NCT006- 99998) [17].
The initial study population included 220 patients. In-hospital mortality was 3.6% (8 patients), and 78 patients were excluded because they were older than 75 years. During the study period, 24 patients were randomized to TRA•CER and 5 to TRILOGY ACS. In addition, 10 patients were immediately referred to surgery. The final study population included 95 patients. The institutional Ethics Committee approved the research protocol, and informed consent was obtained from the study participants.
For analysis, the patients were divided into 4 groups according to theCYP2C19*2 and CYP2C19*17 genotyped combination:
1) Group A ([+]*2/[+]*17): Intermediateplus ultrarapid metabolizers;
2) Group B ([+]*2/[−]*17): Intermediate plus non-ultrarapid metabolizers;
3) Group C ([−]*2/[+]*17): Non-intermediateplusultrarapid metabolizers;
4) Group D ([−]*2/[−]*17): Non-intermediate plus non-ultrarapid metabolizers.
The phenotypic assignment presented was in accordance to the clinical pharmacogenetics guideline for cytochrome P450-2C19 genotype and clopidogrel therapy [18].
Because there was a low number of homozygous patients, 1 patient with CYP2C19*2 and 3 patients with CYP2C19*17, they were included in the heterozygous Groups B and C, respectively, for analysis.
AMI was diagnosed based on the universal definition of myocardial infarction as follows: 1) Positive for cardiac biomarker (troponin I) with symptoms of ischemia or 2) ECG changes indicating new ischemia (ST segment and T wave changes and new bundle brunch block) [19]. ST-elevation AMI was defined by a new onset ST elevation ≥0.2 mV for men and ≥0.15 mV for women in V2-V3, and ≥0.1 mV in other leads. Non-ST elevation AMI, in addition to the previously specified laboratory and clinical criteria, may be associated with or without ECG-associated ischemic changes, such as ST depresssion or T wave inversion [19].
Unstable angina was defined either by new-onset angina (at least class III Canadian Cardiovascular Society [CCS]); progressive angina; or angina at rest, with or without ECG ischemic changes; plus a negative cardiac biomarker assay [20].
The decision to allocate patients to an invasive or conservative management was performed by the cardiologists at the coronary care unit. A stress test was performed in a third of the patients.
A 300 mg clopidogrel loading dose was administered in the emergency department to patients with non-STelevation ACS, and a 600 mg dose was administered to patients with ST-elevation ACS. The subsequent daily and discharge dose of clopidogrel was 75 mg administered at 8 AM. Acetylsalicylic acid was administered at a loading dose of 300 mg, and the subsequent daily dose was 100 mg every day 4 hours after clopidogrel.
Platelet function testing was performed at discharge using multiple electrode aggregometry (MEA) (Multiplate Analyzer®, Dynabyte, Munich, Germany). MEA was assessed using whole blood, according to the princeples of aggregometry; ADP (6.4 μmol/L) was used as the agonist and results were expressed in arbitrary units (U).
2.2. Genotyping
Genomic DNA was extracted by standard methods at hospital discharge from peripheral blood leucocytes. Patients were genotyped retrospectively. CYP2C19*1 (wild type) CYP2C19*2(c.681G>A; rs4244285) and CYP2C19 *3 (c.636G>A; rs4986893) alleles were identified using a dual-priming ologonucleotide polymerase chain reaction (PCR) assay from a commercially available kit (Seegene®). A real-time PCR assay was used to identify theCYP2C19*17 allele (c.-806C>T; rs12248560), using a commercially available kit (Applied Bio systems®).
2.3. Baseline Data and Patient Follow Up
Admission data from patient records that included demographic, clinical, and laboratory information were analyzed. Information on medical therapy, catheterization, and discharge medication were also registered and analyzed.
Clinical follow up was performed over median of 136.0 (79.0 - 188.0) days after hospital discharge. Information was collected by phone calls to patients, from hospital records, or at the outpatient clinic. The primary endpoint used for analysis was a composite of cardiovascular death or readmission for ACSwhichever came first. Clopidogrel compliance was confirmed during follow up.
2.4. Statistics
The Kolmogorov-Smirnov test was used to confirm that all continuous variables were normally distributed. Continuous data are presented as mean and standard deviation and groups were compared usinganalysis of variance (ANOVA) or the Student t test, when appropriate. Categorical variables are reported as frequencies and percentages, and the χ2 or Fisher exact tests were used when appropriate. Chi square tables were used to compare the observed number of CYP2C19 genotypes with that expected for a population in Hardy-Weinberg equilibrium.
Cumulative survival curves were constructed using the Kaplan-Meier method, and patient groups were compared using the log-rank test. The period of observation started at hospital discharge.
A multivariate Cox regression analysis was performed for the primary endpoint. Variables that were significant at the bivariate level (with a p-value< 0.05) and that had clinical relevance were included in the model.
The study had a calculated post-hoc power of 46.3% to identify differences between the groups regarding the primary endpoint on the follow up.
All statistical tests were two-tailed, and a p-value less than 0.05 was considered significant. The analysis was performed using SPSS 15 (Statistical Package for Social Sciences) from SPSS Inc®, Chicago, IL. The post-hoc power calculation was performed with G*Power 3.1.
3. RESULTS
The mean age of the study patients was 59.9 ± 10.7 years with male gender predominance (83.2%). All were Caucasian. The frequencies of the CYP2C19*2 and CYP2C19 *17 alleles were 14.2% and 20.0%, respectively. No deviations from the expected proportions of genotypes in the population as predicted by the Hardy-Weinberg equilibrium equation were noted (p = 0.44 for CYP2C19*2 and p = 0.61 for CYP2C19*17). The CYP2C19*3 variant alleles was not detected.
There were no significant differences between diplotype groups regarding patient demographics, previous cardiovascular history, and risk factors. Group B had the lowest rate of non-ST elevation AMI as the admission diagnosis (respective rates for Groups A, B, C, D were 37.5% vs 11.1% vs 25.9% vs 50.0%; p = 0.02, Table 1).
Almost 75% of both sub-populations underwent invasive treatment. Patient groups had similar coronary anatomy, but Group B patients had the highestrate of stent placement (respective rates for Groups A, B, C, D were 42.9% vs 81.81% vs 81.0% vs 44.1%; p = 0.02). The groups were also similar with respect to ischemic and hemorrhagic risk assessments according to the Global Registry of Acute Coronary Events (GRACE), and the Can Rapid risk stratification of unstable angina patients Suppress adverse outcomes with early implementation of the ACC/AHA guidelines (CRUSADE) scores. Other variables such as left ventricular ejection fraction, peak troponin I levels, change in hemoglobin levels, and glomerular filtration rate were also similar for all groups. There were no significant differences in platelet function between patient groups, but Group A had a lower ADP platelet aggregation than Group B patients (respective values for Group A and B were 14.2 ± 4.2 vs 19.3 ± 6.5U, p = 0.03, Table 2)
All patient groups received similar medical therapy during the first 24 hours of hospitalization and upon discharge (Table 3). Almost all of the patients were administered clopidogrel and acetylsalicylic acid during the first 24 hours of hospitalization. The most frequently used anticoagulant was enoxaparin. Statins, beta-blockers and ACE inhibitors were also administered at high ratesduring the first 24 hours and upon discharge.
Prognosis and Results of Multivariate Analysis
Follow-up data were available for 91 patients (4.2% lost on follow-up). During follow up, there were 10 primary endpoints. One patient died, 6 had new nonfatal myocardial infarctions, and 3 were readmitted for unstable angina. Group B had the lowest event-free-survival rate (respective rates for Groups A, B, C, D were87.5% vs 68.8% vs 96.3% vs 92.5%; log rank p < 0.02, Figures 1-2). After adjusting for covariates, the CYP2C19(+)*2/(−)*17 diplotype was an independent predictor of outcome. It conferred a 5.2-fold higher adjusted risk for occurrence of the composite endpoint compared to the other diplotypes (Table 4).
4. DISCUSSION
The results of our study suggest that the diplotype combination CYP2C19*2 (intermediate clopidogrel metabolizer) and CYP2C19*17 (ultrarapid clopidogrel metabolizer) has prognostic significance over the medium term after an ACS. The patients with the diplotype CYP2C19 (+)*2/(−)*17 (intermediate and non-ultrarapid metabolizers) had higher risk for ischemic events following discharge compared to patients with the other diplotypesCYP2C19(+)*2/(+)*17 (intermediate and ultrarapid metabolizers), CYP2C19(−)*2/(−)*17 (non-intermediate and non-ultrarapid metabolizers), and CYP2C19(−)*2/(+)*17
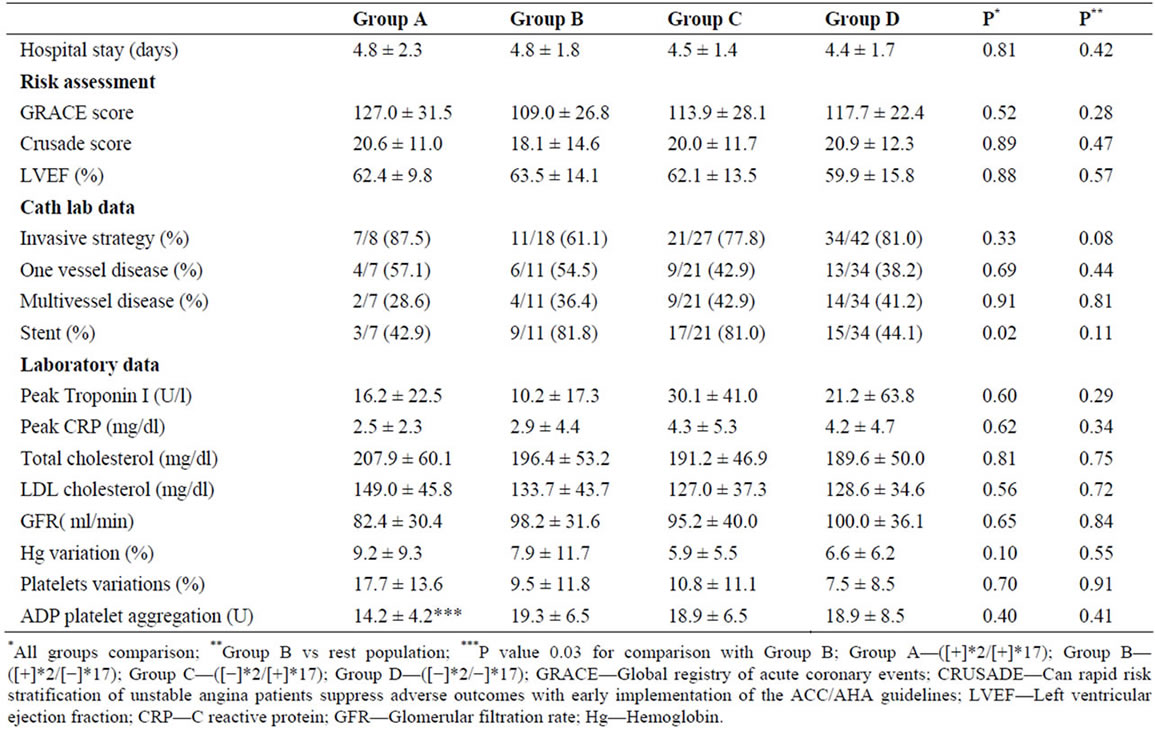
Table 2. Risk assessment, management and laboratory data.
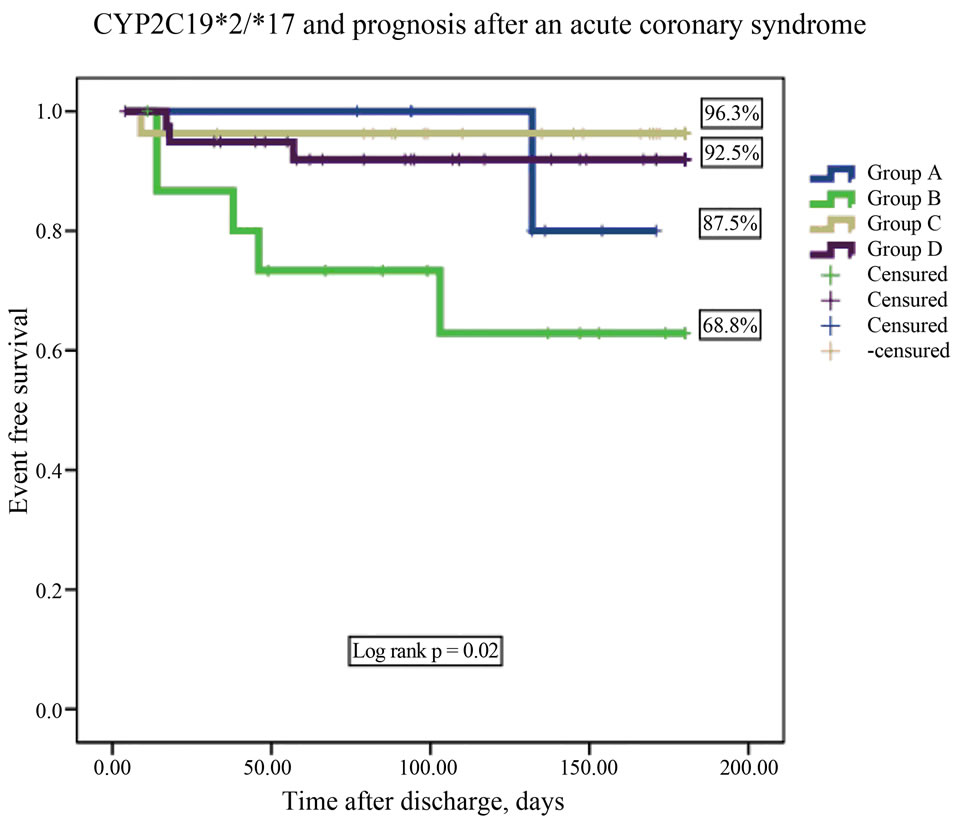
Figure 1. CYP2C19*2 and CYP2C19*17 diplotype combination and prognosis after an acute coronary syndrome.

Figure 2. Intermediate and non ultrarapid metabolizers and prognosis after an acute coronary syndrome.
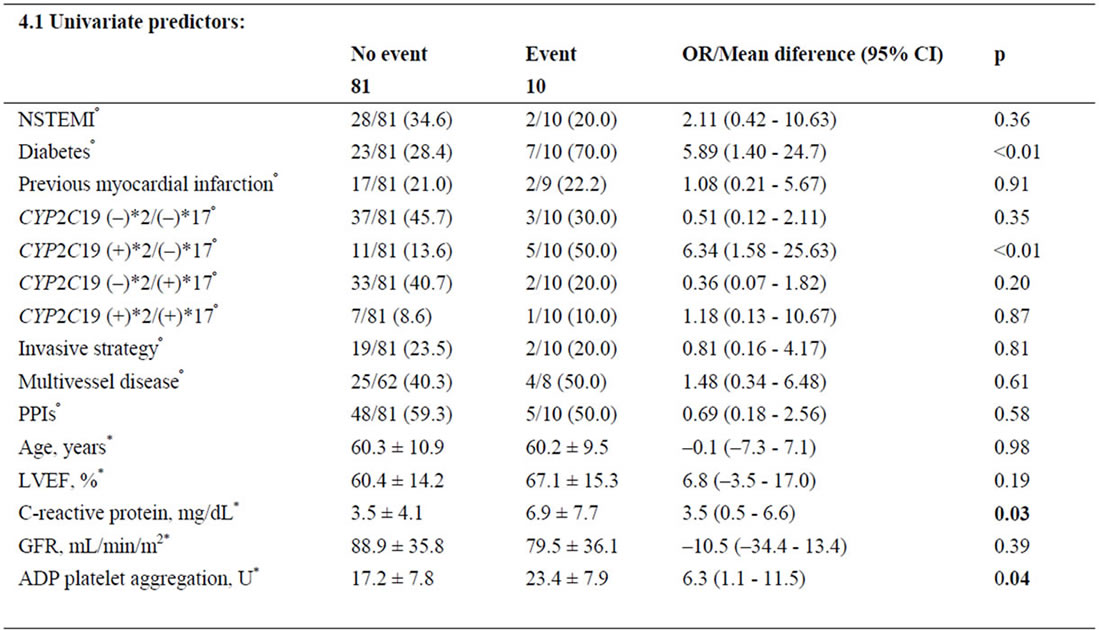

Table 4. Predictors of the combined event.
(non-intermediate and ultrarapid metabolizers).
Using a candidate gene approach, Hulot et al were the first to report an association between the CYP2C19*2 genotype and ADP-stimulated platelet aggregation in response to clopidogrel [2]. Different genes have been implicated in clinically significant clopidogrel response; however, a recent genome-wide association study clarified the importance of the CYP2C19*2 genotype regardingthe metabolism of clopidogrel [9]. A major locus on chromosome 10q24 that influences clopidogrel response extends across the CYP2C18-CYP2C19-CYP2C9-CYP 2C8 gene cluster. Follow-up genotyping indicated that the common loss-of-function CYP2C19*2 variant was significantly associated with clopidogrel response and could account for most of the adverseevents in the initial genome-wide association study. The CYP2C19*2 genotype accounted for approximately 12% of the variation in clopidogrel response and was associated with a worse ischemic outcome, while clinical variables such as age, body mass index, and lipid levels were responsible for approximately 10% of the variation in clopidogrel response [9]. An important conclusion from this analysis was that not only stent thrombosis, but also other events such as myocardial infarction, ischemic stroke, unplanned revascularization, and hospitalization for coronary ischemia without revascularization should be assessed with relation tothe CYP2C19*2 genotype [21].
By contrast, carriage of the CYP2C19*17 allele was previously shown to be associated with increased activetion of the CYP2C19 enzyme, leading to an increased rate of conversion of the pro-drug clopidogrel to an active compound, which was directly associated with a higher degree of platelet inhibition,as analyzed by platelet aggregometry [14]. Assessment of a cohort of postAMI patients who had undergone percutaneous intervention showed that carriage of CYP2C19*17 was associated with better clinical outcome regarding ischemic complications [15].
Previous authors have reported that the CYP2C19*2 and CYP2C19*17 variants, although 19959 base pairs apart are in linkage disequilibrium, and therefore are not independent of one another. Therefore, individuals heterozygous or homozygous for the *17 allele are less likely to carry the *2 allele, whereas those with no copies of the *17 allele (“wild type” at the *17 locus) are more likely to carry the *2 allele, supporting the concept that the *2 variant could account for most or all of the association with clopidogrel response at the CYP2C19 locus [22]. According to Gurbel et al., a hypothesis to overcome this disequilibrium would be to study the phenotypic and prognostic influence of CYP2C19*2/ CYP2C19 *17 diplotypes [22].
Sibbing et al. first reported on the phenotypic association ofclopidogrel-induced platelet aggregation with the diplotype CYP2C19*2 plus CYP2C19*17. Using platelet aggregometry in patients chronically medicated with clopidogrel, the authors reported that a gradual increase in platelet aggregation was seen, starting with the (–)*2/ (+)*17patients who had the lowest ADP-induced platelet aggregation, to the (–)*2/(–)*17 and (+)*2/(+)*17 patients, and ending with the (+)*2/(–)*17 patients, who exhibited the highest value. It was concluded that the increased platelet aggregation seen with CYP2C19*2 was in part reduced by the concomitant presence of CYP2C19*17, although no ischemic or bleeding events were reported for this gene interaction among the 986 stable post-PCI patients with coronary artery disease [3]. A recent paper by Harmsze et al confirmed the phenoltypic changes seen for this diplotype. They measured platelet aggregation using the Verify Now® and the light transmission aggregation assays, and the results were consistent with the hypothesis that the effect of CYP2C19 *2 effect was partly reduced by the concomitant presence of the CYP2C19*17 allele.However, their in vitro findings were not corroborated withdecreased risk of bleeding events or increased ischemic events. The authors proposed that CYP2C19*2 leads to a complete loss of enzyme function, whereas CYP2C19*17 only enhances existing enzyme capacity [23].
None of the studies we have discussed included findings on the risk of ischemic events in patients with the diplotype CYP2C19*2 plus CYP2C19*17. Although the association between this diplotype and platelet aggregation has been previously reported, in our study, the results of platelet aggregation were not statistically significant for all groups comparison because of the low statistical power of our study with respect to platelet aggregation. Nevertheless the (+)*2/(+)*17 patients had a significant higher platelet reactivity than the (–)*2/(+) *17 patients.
Our study patients represent a real-world ACS cohort, with low risk for bleeding, and an intermediate-to-high risk for ischemic events, probably related to the exclusion of patients older than 75 years of age. Although previous studies have identified significant differences between the allelic and genotypic frequencies of polymorphisms (CYP2C19*2 and CYP2C19*17) associated to clopidogrel according to ethnicity [24], our patient population only included Caucasians. We note that the patients with the intermediateplusnon-ultrarapid clopidogrel-metabolizing genotype (Group B) had the highest rate of percutaneous coronary intervention for stent placement. Therefore increased platelet reactivity may have been critical for that group of patients, influencing their adverse outcomes with regard to ischemic events. Our data indirectly supports the concept that the loss of functionCYP2C19*2 allele is partly compensated by the concomitant presence of CYP2C19*17, resulting in clinically significant outcomes after ACS.
5. CONCLUSION
The CYP2C19*2 plus CYP2C19*17 diplotype combination had a significant medium-term effect on the occurrence of ischemic events in patients taking clopidogrel following an acute coronary syndrome.
6. ACKNOWLEDGEMENTS
The authors thank João Pêgo, Gilberto Marques, Carolina Ribeiro, and Alda Reboredo for their work on genotyping.
NOTES