1. INTRODUCTION
Recent estimates suggest that aquaculture industry uses about 2.5 million metric tones of fish meal (FM), representing 40% of the global production which was stabilized at 6 to 7 million tones since 1987 [1]. As aquaculture production continues to increase, there is a subsequently increasing demand for FM. Consequently, the prices has rising, driving research in the feed industry towards finding substitutes [1]. Then, research on the use of non-conventional sources of protein as ingredients to repldcace FM in fish feeds is receiving increasing attention by fish nutritionists around the world. Several vegetable sources have shown to be valuable to replace FM in fish diets, either partially or in total substitution. The use of Sesbania seeds (Sesbania aculeata L.), Moringa leaf (Moringa oleifera Lamarck), Mucuna seeds (Mucuna pruriens L.), duckweed (Spirodela polyrrhiza L. Schleiden), Azolla Lamarck, etc. in diets for O. niloticus had yielded encouraging results [2-6]. Particularly, the fern Azolla has been successfully used recently as a main component in Nile tilapia-diets to replace FM, thus promoting the aquaculture of this fish for poors [7-9]. However, increasing interests were focused nowadays on the fatty acid composition of fish, especially when evaluating the suitability of vegetal sources to replace FM in fish diets. This importance in fatty acid profile of fish stems from their importance in human health, mainly in reducing the occurrence of heart diseases, strokes and various inflammatory injuries, and improving neurological function in newborn infants [10,11]. In that area, findings have confirmed that body fatty acid profile to a large extent mirrors the dietary fatty acid composition [11,12]. Hence, differences could appear between tank and pond rearing systems, due to the natural food present in the latter, when feeding fish with same artificial diets. Indeed, many phytoplankton families [13,14], copepods and rotifers [15,16], and chironomids and oligochaetes [17] are rich sources of fatty acid, particularly linolenic acid (18:3n-3), eicosapentaneoic acid (EPA) and docosahexaneoic acid (DHA), as well as amino acids and vitamins, which could influence growth and nutrient retention in fish. According to Schroeder [18], natural foods contribute for about 50% - 80% in Nile tilapia growth, both in fertilizer and in semi-intensive system. Although an abundant literature is available on fish production in either pond or tank culture systems, very few authors [19,20] have tried to compare fish growth between both systems. Generally, pond conditions are simulated in controlled systems, and comparisons between tank experiments and those in fields like in stagnant earthen ponds are scarcely available.
In the present study, we attempted to investigate the effects of two rearing systems (tank or pond) on growth and fatty acid composition of Nile tilapia when fed diets containing different levels of Azolla, as substitutes for FM.
2. MATERIAL AND METHODS
2.1. Experimental Design, Diets and Feeding
The experiments were conducted during the same period in 18 square concrete tanks (600 L, each) and 18 stagnant earthen ponds (10 m × 3 m × 1 m, each). Tanks were subjected to a recirculating system with a water flow rate of 2 L∙min−1 whereas experimental ponds were filled continuously from the water table, both culture systems being exposed to natural conditions (32˚C - 35˚C; 12-h:12-h dark diurnal photoperiod). Culture units were stocked with male fingerlings of Nile tilapia (initial mean weight: 16.3 g) from the same cohort. Sixty fish (60) per tank and per pond were stocked. Six isonitrogenous (29.2% CP) and isoenergetic (16.9 kJ∙g−1) diets were formulated containing 0% (A0, control diet), 10% (A10), 20% (A20), 30% (A30), 40% (A40) and 50% (A50) using locally available fish meal (locally made of grounded Sardinella aurita fingerlings), maize bran, cottonseed cake, brewery draff and the aquatic fern Azolla (Table 1). The Azolla strain used is Azolla filiculoides Lam., which is the most suitable strain for fish and the more productive and easiest to grow under local conditions. Proximate composition of ingredients, diets processing and biochemical analysis, and methods for conservation before feeding have been detailed in Abou et al. [7]. They were fed to experimental fish in both culture systems, each administered to randomly-assigned groups of three replicate ponds/tanks (6 × 3 ponds/tanks). The daily ration was calculated according to Melard [21] and was adjusted fortnightly based on the actual fish biomass. Daily rations were divided into two parts, each handdistributed at 8:00 h and 16:00 h, respectively.
2.2. Fatty Acid Analysis
At the beginning of the experiment, thirty fish from the initial batch and six fish per pond and per tank at the end were randomly taken and mashed using a Robot coupe food processor. Subsamples were taken and stored

Table 1. Formulation and proximate composition (on dry matter basis) of experimental.
at −20˚C for carcass analysis. Body crude lipid was determined according to Folch et al. [22]. Fatty methyl esters were prepared after transesterification with anhydrous methanol containing 1% sulphuric acid and 0.01% (w/v) butylated hydroxytoluene (BHT) for 16 h at 50˚C [23]. Fatty acids were separated and quantified by gas chromatography (GC Trace, 2000, Thermo Quest, Carlo Erba, Italy), using a 30 m × 0.32 mm capillary column (FAME-WaxTM, USA). Helium was used as carrier gas. The temperature in the oven was programmed to rise from 50˚C to 170˚C at 25˚C∙min−1, then raise the temperature to 190˚C at 2˚C∙min−1 and hold this temperature for 4 min, then increase to 210˚C at 3˚C∙min−1 and to 235˚C at a rate of 5˚C∙min−1 and hold for 5 min. The total running time was 40 min. Fatty acids peaks were integrated and quantified by Chrom-Card for Trace computing integrator. Identification was made by comparison with a well known and characterized standard PUFA-3, from Menhaden oil (Supelco, Bellefonte, PA).
2.3. Water Quality
During both experiments, water quality parameters were monitored fortnightly in the cultured systems. Water temperature, dissolved oxygen (DO) and pH were measured at 10 cm depth at the following times: 08:00, 11:00, 14:00 and 17:00 hours, using an oxythermometer (WTW Oxi 197i, WTW, Weilheim, Germany, precision: ±0.01˚C and ±0.01 mg∙L−1) and a pH meter (WTWpH 330i, precision: ±0.01) respectively. Nitrite levels were measured using spectrophotometric methods according to APHA [26]. Estimation of chlorophyll a concentration was carried out according to standard methods described in APHA [26]. To estimate zooplankton abundance in experimental ponds, 20 L of water were collected in three points of each pond and filtered through a 55 µm plankton net to obtain a concentrated sample. The samples were preserved immediately with 5% formalin. The absolute abundance of zooplankton was estimated by counting samples in a Dolfus cell under a binocular magnifying glass (×40).
2.4. Statistical Analysis
Variations in growth, fatty acid composition of fish, and water quality parameters were analyzed in each culture system using one-way Anova, after verifying the homogeneity of their variances [27]. Two-way Anovas were used to compare performances in both culture systems. Values for percentage data and ratios were logtransformed prior to analyses. When the effect was significant, comparisons between means were run using Duncan’s multiple range test [28] at P = 0.05. A multivariate procedure, principal component analysis (PCA) was used to analyze the extent to which the fatty acid profile in fish differed among culture system and how they are comparable to the fatty acid profile in experimental diets. This analysis was carried out on standardized variables of fatty acid composition as original data. The software Canoco for Windows version 4.5 was used for the analyses and drawing diagrams.
3. RESULTS
3.1. Water Quality
Water quality parameters were statistically similar in each experiment (Table 2). Ranges for temperature, pH, dissolved oxygen and nitrite levels were respectively 30.0˚C - 30.2˚C, 6.33 - 6.62, 4.65 - 5.49 mg∙l−1 and 0.01 - 0.02 mg∙l−1 in ponds and 29.5˚C - 30.3˚C, 6.80 - 7.02, 6.55 - 7.68 mg∙l−1 and 0.0 - 0.02 mg∙l−1 in tanks. Dissolved oxygen and pH were significantly higher in tanks than in ponds (P < 0.05).
Evaluation of natural food in experimental ponds, mainly chlorophyll a biomass and zooplankton abundance, showed similar values between dietary treatments, ranging from

Table 2. Pond and (tank) values of water quality parameters. Data are mean ± S.D. of three replicates from fortnight measures. A0 to A50: diet with 0% to 50% Azolla.
16.8 mg∙l−1 to 18.7 mg∙l−1 and from 759 l−1 to 862 l−1, respectively.
3.2. Fish Growth
In pond and in tank experiments, survival rate did not show any significant differences, and values were higher in both culture systems, ranging from 89.4% to 93.3% in tanks and from 85.0% to 90.0% in ponds. Final mean weight decreased significantly in tanks and in ponds when AM level exceeded 10% and 20% in diets (P < 0.05), ranging from 88.3 g to 51.0 g and from 135.9 g to 88.8 g respectively. Two-way anovas show that for each diet, the values obtained in ponds were higher than those in tanks (Figure 1, P < 0.001). The improvement of fish growth in ponds compared to that in tanks for the same diet is important in fish fed high AM level in diets, representing 54% to 75% from fish fed A0 to those fed A50.
3.3. Variation in Fatty Acid Composition of Fish
Data spread across up in the PCA axes showed that fatty acid composition in fish varied according to culture system (pond or tank) and experimental diets (Figure 2). The first axis, which accounts for 51% of the total variability, separated fatty acid as a function of experimental diets. Diet formulations with a higher contribution of AM contained lower percentages of 14:0, 16:1n-7, 18:1n-9 and 18:3n-3 fatty acids, and higher percentages of 18:0 and 20:4n-6 fatty acids. These differences in dietary fatty acid compositions were well observed in fish fatty acids, especially for fish fed in tanks.
The second axis, which accounts for 19.3% of the total variability, separated the fatty acid composition of fish in function of culture system (pond or tank), especially for

Figure 1. Pond and tank values of final mean weight of Nile tilapia fed gradual Azolla meal. a,bFor each Azolla meal level, bars with different letters are significantly different (P < 0.05).

Figure 2. Principal Component Analysis ordination of fatty acid composition of fish fed in ponds (grey circles) or in tanks (white circles). Profiles of experimental diets (black circles) were added as passive variables. Circle size is proportional to the amount of AM in diets. The principales fatty acids are: Myristic acid, 14:0; palmitic acid, 16:0; stearic acid, 18:0; palmitoleic acid, 16:1n-7; vaccenic acid, 18:1n-7; Oleic acid, 18:1n-9; linolenic acid, 18:3n-3; linoleic acid, 18:2n-6; arachidonic acid, 20:4n-6; eicosapentaenoic acid, 20:5n-3; docosahexaenoic acid, 22:6n-3; clupanodonic acid, 22:5n-3.
fish fed on diets A30, A40 and A50. Globally, fish in ponds had higher levels of high unsaturated fatty acid (HUFA) namely clupanodonic acid (22:5n-3), EPA (20:5n-3) and DHA (22:6n-3). The Euclidean distance between each fish fatty acid profile and the one of its diet (Figure 3) showed that the distance between both profiles increased with increasing AM in diets (P < 0.05). Within each AM level, values were statistically different for fish fed in both systems with A20, A30 and A40 (P < 0.05).
Values of (n-3) fatty acid to (n-6) fatty acids ratios (n-3/n-6) in fish were plotted with AM level in diets (Figure 4). Values tend to increase in ponds whereas a decreasing trend is found in tanks (P < 0.05) when AM level increased in diets. Pond values were significantly higher than tank values (P < 0.001).
4. DISCUSSION
In this study, fish from the same cohort were fed the same diets at the same period. The growth of O. niloticus revealed that final mean weight in fish fed in ponds was significantly higher than in fish fed in tanks, using the same diets. These results are in accordance with those reported in the literature. Higher weight gain of koi carp (Cyprinus carpio var. koi) was obtained in earthen ponds compared to that in tanks [20]. Significantly lower growth rates in Catfish, Ictalurus punctatus Rafinesque, had been reported in concrete pools compared to earthen ponds [19]. It is well established that fish size, water temperature and composition of diet are the main factors generally affecting growth rate in fish [29-31]. As ex-
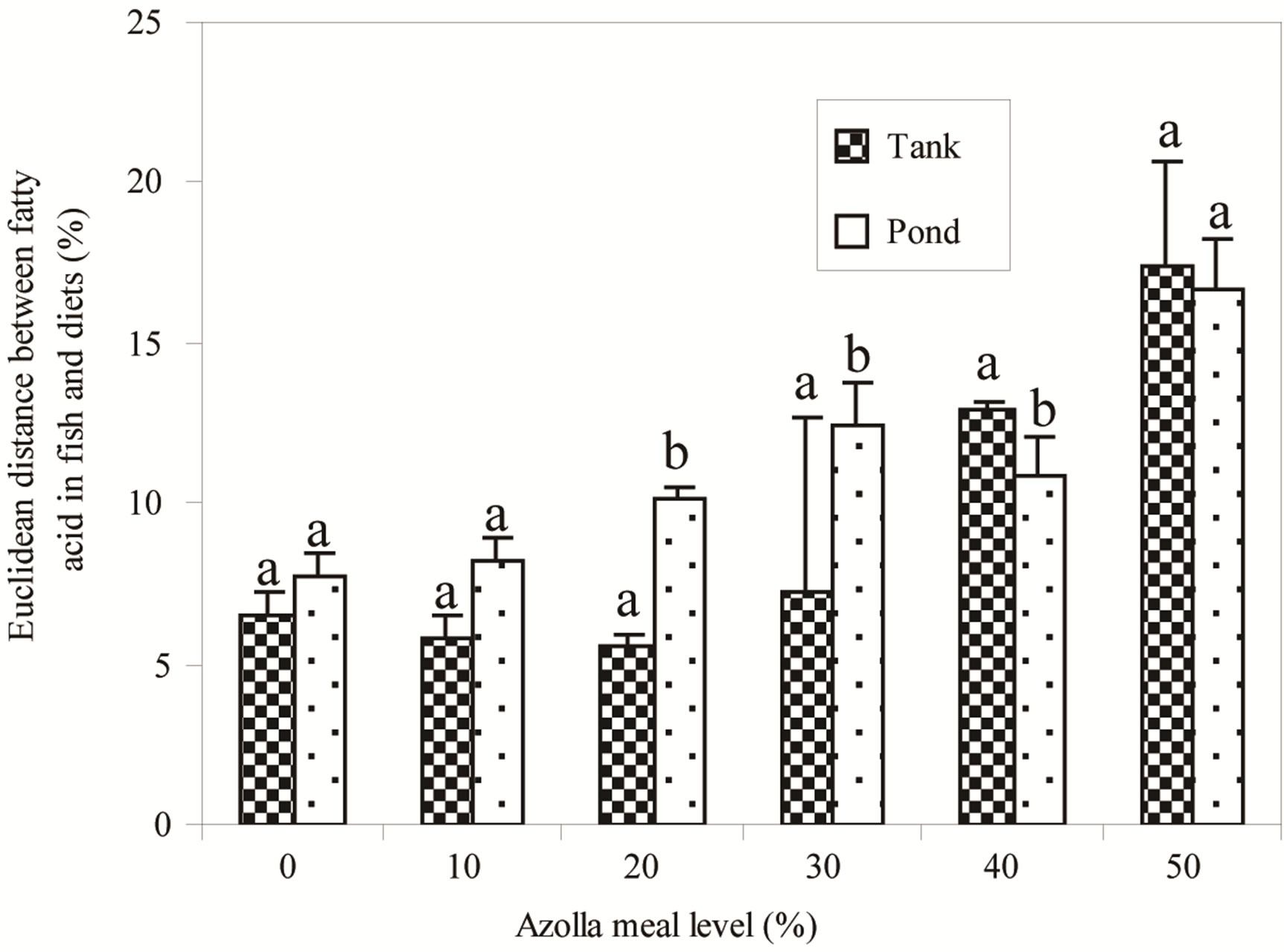
Figure 3. Euclidean distances between fish fatty acid profiles and fatty acid profile of their food. (a) and (b) For each Azolla meal level, bars with different letters are significantly different (P < 0.05).

Figure 4. n-3/n-6 ratios in fatty acid of fish fed in tanks and in ponds with gradual Azolla meal level in diets. (a) and (b) For each Azolla meal level, bars with different letters are significantly different (P < 0.001).
plained above, fish size and water temperature were closely similar in both culture systems, and could not influence the variation in growth rates. Identical artificial feed was provided, based on fish biomass after each fortnight. Alternatively, the natural food available in ponds, mainly phytoplankton, zooplankton and other invertebrates, should be considered. Like other tilapias species, the Nile tilapia O. niloticus feeds permanently on plankton and indiscriminately on whatever is available in water bodies [32]. High filtration rate of phytoplankton by Nile tilapia was obtained in laboratory studies [33] while phytoplankton, zooplankton and other insects larvae constitute the main food resources found in the stomach of this fish under natural conditions [34]. Phytoplankton is reported to contain protein levels ranging from 12% to 35%, lipids ranging from 7.2% to 23%, and carbohydrates ranging from 8.2% to 8.7% on a dry matter basis [35]. Moreover, Albrecht & Breitsprecher [36] have reported that the mean protein, carbohydrate and lipid contents of fish food organisms could reach 511, 273 and 77 g∙kg−1 respectively, the energy value ranging from 6.7 to 23.8 kJ∙g−1. Many phytoplankton, zooplankton and invertebrates families are rich sources of amino acids, fatty acids, minerals [13-17] and vitamins. This high nutritional value of natural foods in ponds could partly balance any deficiency observed in artificial diets through additional proteins, amino acids, fatty acids, minerals and vitamins, and therefore improve growth of fish in ponds compared to those receiving only artificial food in tanks. Natural foods generally account for a large share of total fish growth. The growth in ponds was 54% to 75% higher than that in tanks, consistent with values of 50% to 80% reported by Schroeder et al. [18] in tilapia reared in a semi-intensive system, as the contribution of natural food. The contribution of natural foods seems to be higher in fish fed high AM levels in diets. Experimental ponds were newly constructed for the purpose of the study, and any management practices were applied.
Also, no differences were recorded in the quantity of natural foods, mainly chlorophyll a and zooplankton abundances, during the experiment. It had been proved that small tilapia filtered significantly more plankton than larger individuals [37-39]. And according to Schroeder [40], fish feed at lower trophic levels if their usual food becomes limited. Because of the lower growth in fish fed A30 to A50 diets in ponds, due to methionine and (n-3) fatty acids deficiencies, and relative lower digestibility of AM, the consumption of natural foods by fish fed in ponds receiving diets A30-A50 should be higher than those fed in ponds with diets A0-A20. In fact, the contribution of natural foods to growth of fish fed A30-A50 should therefore be higher.
As shown in Figure 2, fish fatty acid composition indicates significant variations. In fish fed in tanks, an influence of dietary fatty acids was shown, in agreement with several studies reporting on fatty acid composition of fish reared in controlled conditions [11,12,41]. Fatty acid profiles of fish reared in ponds were clearly different to those of fish fed in tanks, and seem less influenced by the diets. In tanks as in ponds, Euclidean distances between fish fatty acid profiles and the composition of their diets increased with AM in diets. It means that the more Azolla meal in diets, the less fish fatty acid profiles are similar to the one in the diets. The fatty acid mainly concerned in fish fed in tanks is arachidonic acid (20:4n-6) in fish fed A40 and A50 (Figure 2). This fatty acid is a product of an elongation and desaturation of linoleic acid (18:2n-6). Dietary fatty acid has no influence in fish fed high AM in ponds. Interestingly, high amounts of EPA (20:5n-3), clupanodonic acid (22:5n-3) and DHA (22:6n- 3) were found in fish fed A30, A40 and A50. Such composition of fish mirrors the fatty acid profiles of phytoplankton and zooplankton. Indeed, phytoplankton is known to contain high amount of EPA [14,42] whereas copepods and rotifers are rich sources of EPA and DHA [16,43,44]. It is worth noting that this fatty acid content in fish is consistent with our hypothesis on the reliance of fish fed more AM on natural foods, compared to those fed low AM. Our findings are in agreement with several reports that fish consuming natural foods are generally richer in eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), which exert positive health effects for humans [45,46]. The proof was given in this study (Figure 4) by the higher values of the n-3/n-6 ratios in ponds compared to those in tanks for the same diets, showing that quality of fish produced in ponds is better than that produced in tanks.
5. CONCLUSION
From these experimental results, earthen ponds appeared to be better alternative to concrete tanks for production of Nile tilapia with Azolla-diets, through the presence of natural foods. Hence, taking into account the aforementioned aspects of the study, it can be recommended to raise Nile tilapia with Azolla-diets in ponds to improve growth and to enhance fatty acid composition of fish produced, for the well-being of consumers.