Development of Misoprostol Suppositories for Postpartum Hemorrhage ()
1. Introduction
Misoprostol is a synthetic prostaglandin E1 analogue, manufactured as an oral preparation available as 200 µg tablets used to prevent and treat gastroduodenal damage induced by nonsteroidal anti-inflammatory drugs [1]. The most common adverse effects of misoprostol are nausea, vomiting, diarrhea, abdominal pain, chills, shivering, and fever, all of which are dose-dependent [2]. Misoprostol taken by pregnant women increases uterine tone and contractions.
It has also become an important off-label drug in obstetrics and gynecology because of its uterotonic and cervical-ripening actions. It is an alternative to uterotonic drugs such as oxytocin, methylergometrine and prostaglandins, which are unstable at room temperature and require injection [3]. Misoprostol is only commercially available as 200 µg oral tablets in Switzerland. However the tablets are also effective when administered vaginally, rectally, buccally and sublingually. Misoprostol is useful for medical abortion, cervical ripening before surgical abortion, evacuation of the uterus in cases of embryonic or fetal death, and labor induction. The drug is also used in the third stage of labor to prevent and to treat postpartum hemorrhage (PPH). Misoprostol is considered as an “essential drug” by the World Health Organization (WHO) for the management of incomplete abortion and miscarriage, and for prevention of PPH when oxytocin is not available or cannot safely be used [4].
Any woman who gives birth can have PPH which may threaten her life. PPH is one of the leading causes of maternal mortality and an important cause of serious morbidity in the developing and developed world. Even the mild self-limiting cases have consequences for the patient’s puerperium in the form of fatigue, tiredness, failure to breast-feed and possible need for haematinics or blood transfusion [5]. PPH is defined as a blood loss of greater than 500 mL for a vaginal delivery and greater than 1000 mL for a cesarean delivery. The predominant cause of PPH is uterine atony or failure of the uterus to adequately contract after delivery. The incidence of PPH is about 5% in Europe [6]. The majority of patients who develop PPH do so in the absence of well-known risk factors. The first step in reducing morbidity and mortality of PPH is therefore to improve methods of prevention [7]. Active management most commonly comprises uterine massage, controlled traction on the umbilical cord and the use of a medication to favor uterine contractions, e.g. intramuscular injection of oxytocin and/or ergot alkaloids (ergometrine) or misoprostol per os. The treatment consists of improving uterine tone and the exploration of the uterus for any evidence of retained placental tissues. The use of different molecules (oxytocin, sulprostone, misoprostol and/or ergometrine) is common in many countries.
Since 1987, misoprostol has been used to prevent or treat PPH in doses up to 600 µg in oral or sublingual administration [8-10] and up to 1000 µg rectally [11-16]. Misoprostol tablet can also be absorbed by both rectal and vaginal routes [17]. However, the rate of absorption varies considerably between these routes of administration. Rectal administration of misoprostol tablets is associated with a qualitatively similar absorption curve to that of the vaginal route but presents a lower bioavailability. The vaginal route could not be considered in this case because of the blood loss. Oral misoprostol reaches a high peak plasma concentration followed by a rapid fall [18]. Rectal misoprostol absorption in the third stage of labor avoids the first-pass effect and decreases the adverse effects. WHO does not recommend such practice for PPH [19] because its potential benefits and harms are currently unknown. However some organizations, e.g. the Swiss society of gynecology and obstetrics (SSGO), the international federation of gynecology and obstetrics (FIGO) and the international confederation of midwifes (ICM) [20] use it as third-line treatment.
The purpose of this study was to manufacture a 1000 µg misoprostol suppository using commercially available tablets and a suppository base and to determine the in vitro release, the stability and the facility of use.
2. Experimental
2.1. Materials
Hard fat-Adeps solidus Ph. Eur. and Witepsol®H15 were purchased from Hänseler (Herisau, CH). Suppocire® AM and Suppocire® AS2X were purchased from Gattefossé
SAS (Saint Priest, France). Tween® 20, Tween® 80 and sodium laurylsulfate (SLS) were purchased from Hänseler (Herisau, CH). Cytotec® tablets 200 µg misoprostol were purchased from Pfizer (Zurich, CH). Benzophenone was purchase from Sigma Aldrich (Steinheim, Germany). Acetonitrile LC-MS Chromasolv, Fluka analytical was purchased from Sigma Aldrich (Steinheim, Germany). Pure misoprostol was purchased from Sigma Aldrich (Steinheim, Germany). HPCL-filtered water was obtained, in-house, from a Millipore model Synergy 185 (MA, USA).
2.2. Methods
Suppository preparation: Suppositories were manufactured according to the melting method by calculating the displacement value of the suppository bases. The 1000 µg misoprostol suppositories were prepared with different bases (Adeps solidus, Witepsol®H15, Suppocire® AM and Suppocire® AS2X) and different surfactants (Tween® 20, Tween® 80 and sodium laurylsulfate (SLS)) at concentrations of 0.5%, 1%, 2%, 3% or 5%. Suppositories were prepared by crushing five 200 µg misoprostol tablets in a mortar, melting the suppository base with the surfactant in a water bath at 37˚C and adding the crushed misoprostol. The mixture was transferred in suppository molds of 3 g.
In vitro release: A system was specially developed for the set-up of the in vitro release test. Three suppositories were put in a basket immersed in a 500 mL buffer bath at pH 7.2 containing SLS (from 0% to 5%) at 37˚C ± 1˚C with magnet agitation (100 rpm). A 900 µL volume was withdrawn from the medium at 2, 5, 10, 20 and 30 minutes and 100 µL of a 0.3 µg/mL solution of benzophenone (internal standard) was added. The solutions were analysed in triplicate by high-performance liquid chromatography (HPLC).
Stability study: Misoprostol stability in suppositories with the formula allowing the best release was assessed after conservation at 4˚C, 20˚C and 60˚C. Suppositories were tested immediately after preparation and at 7, 14, 30, 60, 90, 120, 150, 180 and 210 days. After the suppository was melted in a water bath at 37˚C ± 1˚C, 7.0 mL of acetonitrile was added and mixed for 2 min. with a Vortex-type mixer to extract the drug. The vortex mixer model SA7 was purchased from Stuard (UK). The mixture was centrifuged at 3000 rpm for 10 min. A 300 µL aliquot was mixed with 100 µL of a 30 µg/mL solution of benzophenone and 600 µL of acetonitrile. This solution was analysed in triplicate by HPLC.
HPLC method: Three HPLC methods were found in the literature for the analysis of misoprostol [21-23]. The method described by Hafirassou [23] was adapted and validated, following the ICH guidelines for the present study. The quantitative analyses were performed on an HPLC pump Serie 1100 with a binair pump model F 1312 A (Agilent, USA). The separation was achieved on a Zorbax Eclipse XDB C18 column (4.6 × 150 mm, 5 µm (Agilent, USA), maintained at 23˚C. The equipment also consisted of an automatic injector model G 1367 A (Agilent, USA) with a 20 µL loop and an equipped with an UV detector model DAD SL G 1315 C (Agilent, USA) set at 202 nm. The mobile phase was prepared by mixing acetonitrile and filtered water in the ratio of 60:40 (v:v). The flow rate was set at 1 mL/min.
No interfering peaks were identified with the lipophilic bases, the surfactants or the degradation products of misoprostol. The retention times for misoprostol and benzophenone were 3.77 and 5.59 minutes, respectively (Figure 1).
Standard solution and standard curve: The linearity, trueness and accuracy of the analytical method were evaluated for both studies (release and stability). A standard solution of misoprostol was prepared by dissolving pure misoprostol in acetonitrile (100 µg/mL = S). This solution was kept at −20˚C. This solution was further diluted in acetonitrile (10 µg/mL=S1). The standard curve was built by plotting the ratio of misoprostol peak area to that of benzophenone according to misoprostol concentration and used for measuring drug concentrations in samples. Three replicate standards at three different days and two replicate injections were used for the standard curve.
Misoprostol standard solutions for the release study were prepared by diluting S1 with acetonitrile to concentrations of 0.3, 3 and 7.2 µg/mL. Each solution contains 0.3 µg/mL of benzophenone (internal standard) diluted in acetonitrile. The standard curve was linear over the concentration range of 0.3 - 7.2 µg/mL. A linear relationship was plotted over the concentration range of 0.3 - 7.2 µg/mL. The intercept was found to be not significantly different from 0 (Student t test, α = 0.05). The intraday and interday relative standard deviations were found to be inferior to 6.2%.
Standard solutions for the stability study were prepared by diluting S solution to 3 different concentrations (28, 40 and 52 µg/mL) with benzophenone at 3 µg/mL in acetonitrile. This curve was also linear over the working range of 28 - 52 µg/mL. The plots intersected the origin at 0.08%. The intraday and interday coefficients of variation were ≤4.3% and 3.5%, respectively.
Data analysis: Misoprostol release was determined by calculating the percentage of misoprostol concentration liberated in the media according to time. Stability was determined by calculating the percentage of the initial concentration remaining after each time interval. Stability was defined as the retention of at least 90% from the initial concentration.
Facility of use: Because of this study the Swiss ethic committee was contacted, and it approved this project. The suppositories were proposed to the gynecology service of a regional hospital. They were used in 6 patients for the treatment of PPH as third-line treatment after oxytocin and methylergometrine instead of the 5 tablets of Cytotec® administrated rectally. The facility of use was evaluated through a questionnaire. These questionnaires were anonymously filled and sent to the pharmacy.
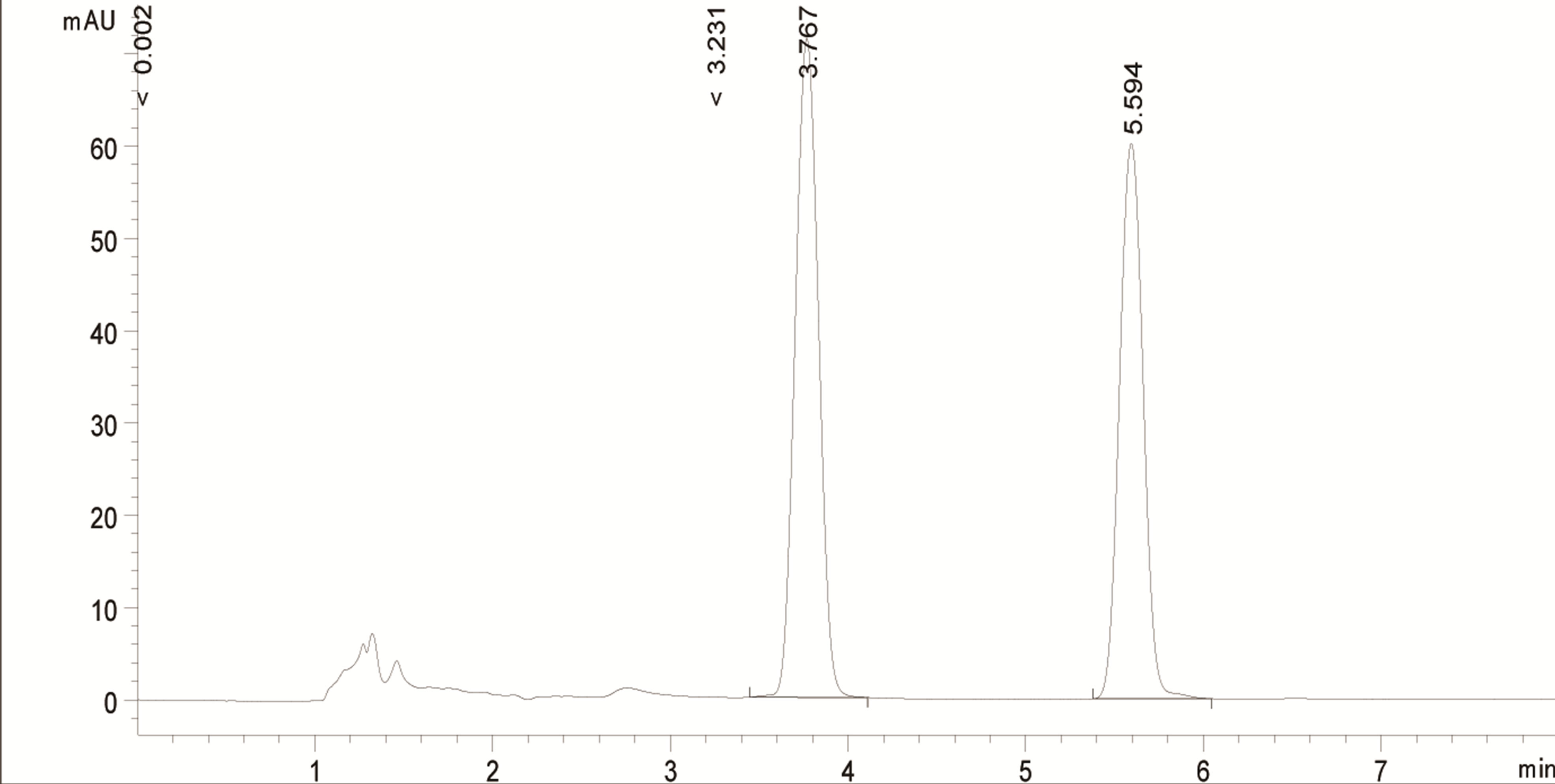
Figure 1. Representative chromatogram of a 40 µg/mL misoprostol and a 3 µg/mL benzophenone solution in acetonitrile. Peak at 3.767 minutes = misoprostol, peak at 5.594 minutes = benzophenone (internal standard).
3. Results and Discussion
The dosage of 1000 µg misoprostol suppository was chosen after discussion with the local gynecologists. Different surfactants were added to the suppository to improve the dispersion and absorption of the drug [24].
There is no standard method to determine the release of an active substance from a suppository and no medium simulating rectal fluid [25]. The European and American Pharmacopoeia propose a continue flow system to control drug dissolution for suppositories in water [26,27]. The Fédération internationale pharmaceutique (FIP) and the American Association of Pharmaceutical Scientists (AAPS) have proposed guidelines on suppository dissolution, using the continue flow system [28]. This system was not available for the present study. A specific system with a medium close to the rectal fluid (pH and temperature) was therefore developed. The addition of SLS (0% to 5%) in the medium is known to improve the dissolution of drug [29,30]. The optimal concentration of the SLS was obtained experimentally and fixed to 0.5%.
Release: Adeps solidus liberated the highest quantity of drug after 30 minutes (19% versus 16% for Witepsol®H15, 11% for Suppocire® AM and 6% for Suppocire® AS2X, Figure 2). The adjunction of Tween® 80 to Adeps solidus and Witepsol® H15 presented a slight influence on misoprostol release from the suppository (data not shown). The adjunction of two others surfactants (SLS and Tween® 20) had more effects. Best results were obtained with the adjunction of 1% SLS (59% ± 1.4% after 30 minutes) and 5% Tween® 20 (57% ± 8.2%) to Adeps solidus (Figure 3). The use of SLS in the release medium enhances misoprostol solubility. SLS has however an irritant property for the mucous membrane [31]. This surfactant was discarded, even if exposure is limited to a single use. Thus, the formula retained contains 5 crushed tablets of Cytotec® in Adeps solidus with 5% Tween 20.
Release tests were conducted over 30 minutes. Indeed, a rapid effect is needed to treat PPH. The release test was performed in a 500 mL phosphate buffer media while the rectum is composed of about 2 mL fluid and the mucus action on the suppository can also influence the absorption. This test allowed us to compare different formula. Nevertheless, only an in vivo study with blood samples could accurately evaluate misoprostol release in the rectum and its bioavailability.
Stability: 1000 µg misoprostol suppositories contained more than 90% of the initial concentration after 210 days of storage at 4˚C (Table 1). At room temperature, the results were not homogeneous and content rapidly decreased at less than 90% (data not shown).
In the study from Hafirassou [23], the Suppocire® suppositories of misoprostol were not stable at more than 120 days at room temperature.
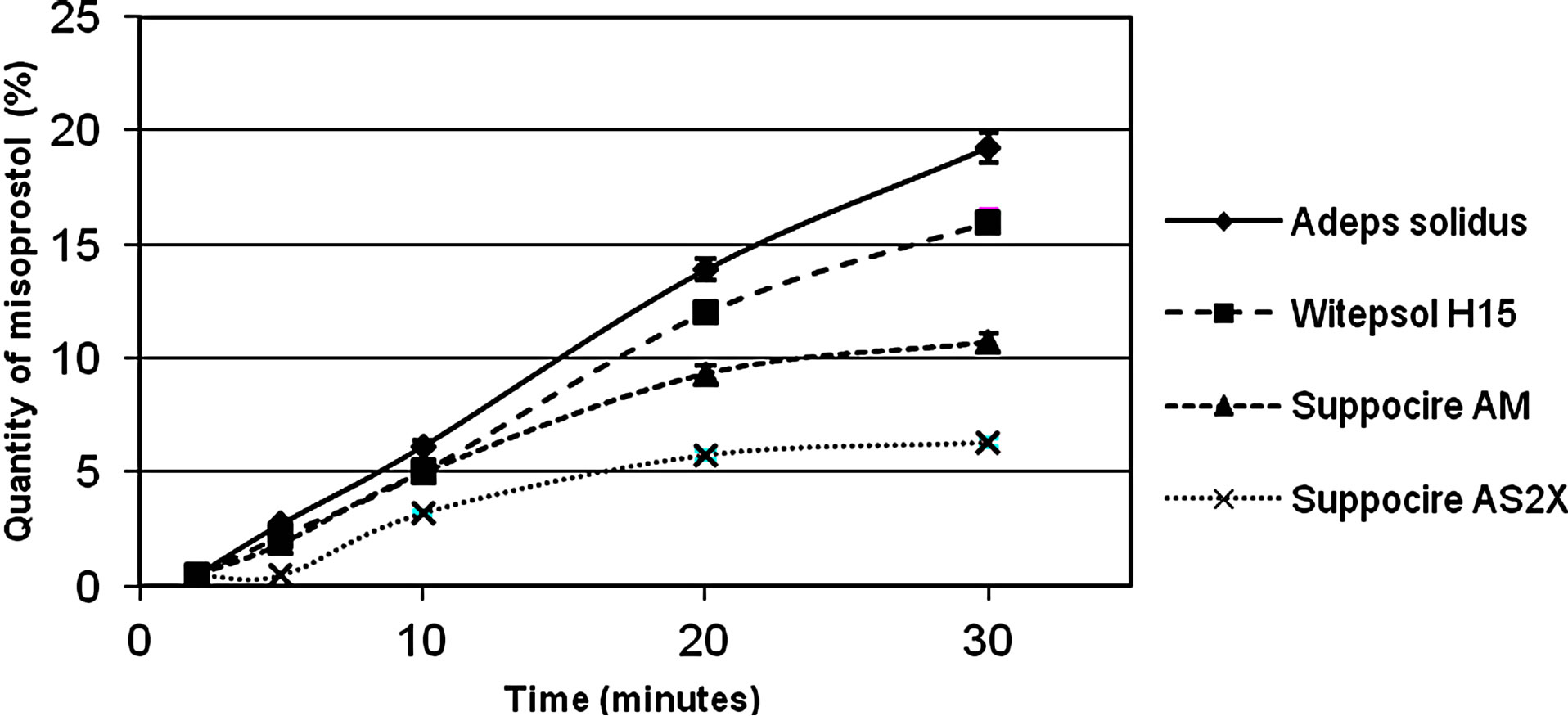
Figure 2. Misoprostol release from different suppository bases.

Figure 3. Misoprostol release from Adeps solidus suppositories alone and with addition of 1% SLS and 5% Tween® 20 in a normal media and with 5% Tween® 20 in a medium containing 0.5% SLS (mean ± SD).
Table 1. Stability of 1000 µg misoprostol suppository conserved at 4˚C.
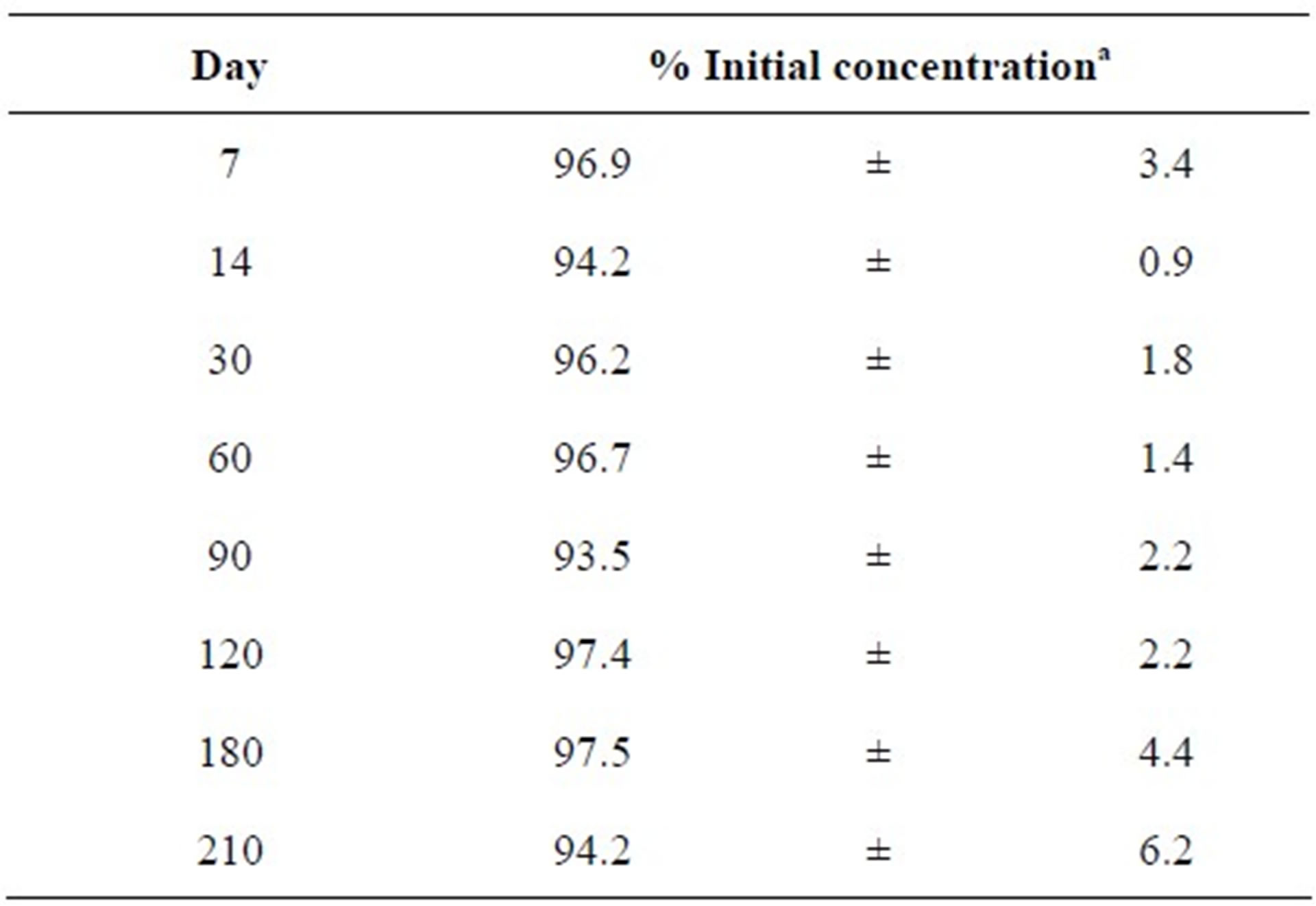
aMean ± S.D. of triplicate determinations for three samples (n = 3).
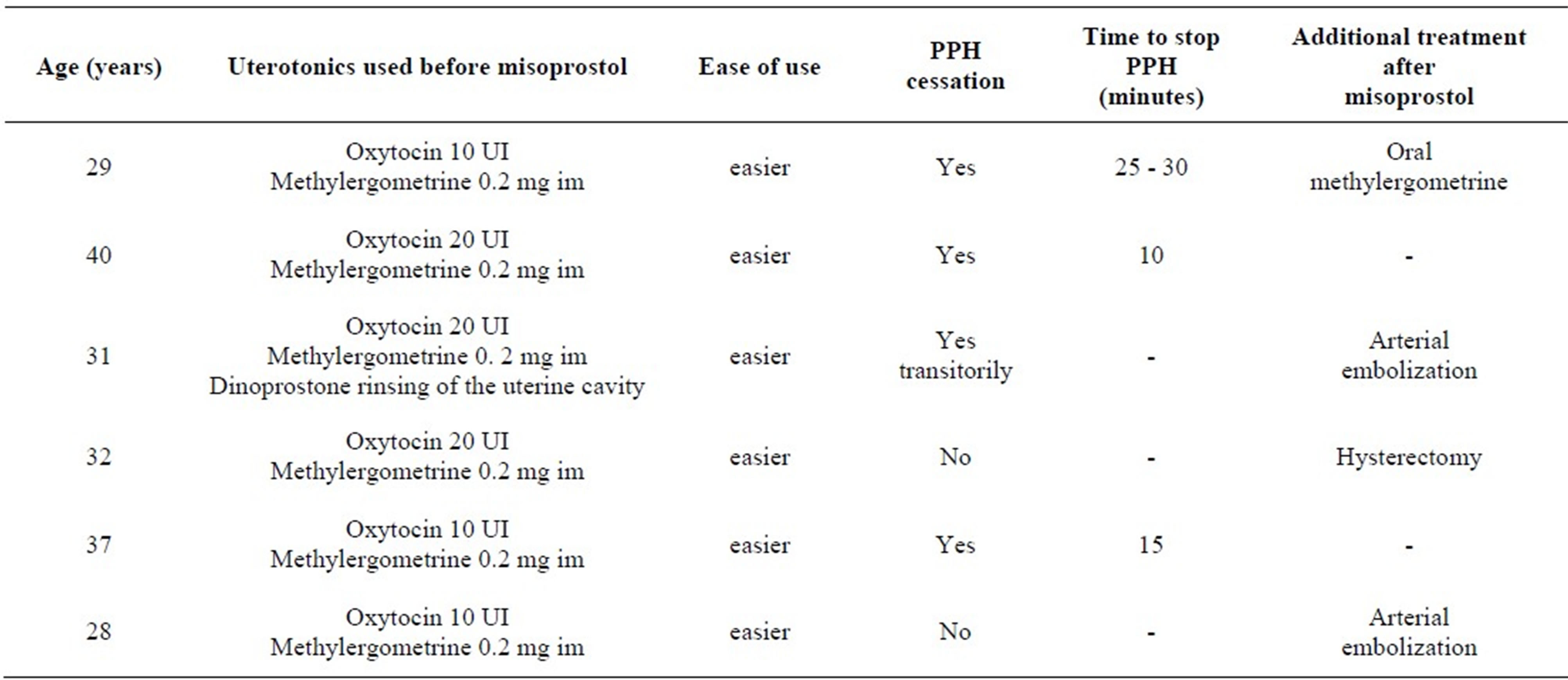
Facility of use: In a pilot study suppositories were used on a gynecology ward. Six questionnaires were filled and sent to the pharmacy. The six patients received oxytocine as prevention and a second dose of oxytocine added to methylergometrine before the use of a misoprostol suppository in case of a declared PPH. All doctors appreciate the facility of use (Table 2). PPH was stopped in 50% of the patients who received misoprostol suppository, which is in agreement with published studies [11-16,32]. The number of patients should be increased in a future study to evaluate to a larger extent the efficacy of this suppository versus tablet in the treatment of PPH.
Table 2. Summary of survey after the use of misoprostol suppository.
4. Conclusion
A 1000 µg misoprostol suppository was elaborated with crushed tablets of misoprostol and hard fat (Adeps solidus Ph. Eur.) +5% Tween® 20. The suppositories liberated in vitro 57% ± 8.2% of misoprostol after 30 minutes. The suppositories are stable for at least 7 months at 2˚C - 8˚C and facilitate the administration of misoprostol compared to tablets in the treatment of PPH.
NOTES