Global Dynamic Analysis of a Vector-Borne Plant Disease Model with Discontinuous Treatment ()
1. Introduction
The plants play an important role in our lives, as most of our daily food, clothing and building materials come from plants. With the change of environment, there are outbreaks of plant diseases, which seriously affect the health of plants and people’s life, such as huanglongbing [1] , Blackleg [2] . We know that plant diseases have been responsible for the death and suffering of millions of people and countless animals [3] . Controlling the outbreak and spread of plant diseases has become the common goal of scientists. Available control measures include biological, cultural, and chemical methods [4] . Chemical control is a quite effective method, but the residues of chemical drugs have a direct negative impact on environment and thus are not encouraged. To effectively control plant disease and to reduce the harm to environment, it is crucial to understand disease transmission dynamics.
The prevention and control of plant infectious diseases is of vital importance in agricultural production [5] . To work with the plant disease, we first understand how they spread. There are many ways that plant viruses interact with the vectors; this transmission works in the following way. The vectors consume sap from an infected host through their stylet. When the infected vector contacts a healthy plant, some virus particles leave the vector and invade the plant [6] . So the vector-borne is a very important part of the transmission of plant diseases.
Treatment plays a very important role to control the spread of diseases. In recent years, many researchers [7] [8] [9] [10] have studied some mathematical models incorporating treatment. For example, in [11] , Wang and Ruan studied an epidemic model, and provide the limited resources for the treatment of patients. In [12] , Wang proposed constant treatment, which simulates a limited capacity for treatment. In practice, when the number of infectives is large, the constant treatment is suitable for hypothesis of model. Recently, discontinuous treatment strategies are proposed by [13] [14] . The results show that discontinuous treatment strategies would be accord with real condition. Applying this discontinuous treatment strategy makes the mathematical model a discontinuous system. At the same time, some non-smooth analysis techniques [15] are used for this system.
In [16] , Shi and Zhao presented a vector-borne plant disease model, but they do not studied treatment to the infected plant host. Treating infected plant is a quite effective method which to control the outbreak of the plant disease. Although continuous treatment is an effective method, the outbreak of the plant disease is periodic, and continuing treatment can be a huge waste of resources. In order to be realistic, we built a vector-borne plant disease model with discontinuous treatment.
The paper is organized as follows. In the next section, we will construct the model and introduce the rational assumptions for model. In Section 3, positivity of the solution for the model will be clearly discussed. We obtain the existence of possible equilibria, the basic reproductive number, and the stability of equilibria in Section 4. In Section 5 and Section 6, we summarize our main results and main results are numerically simulated.
2. Model and Preliminaries
To construct the model, the following assumptions are being made by Shi et al. in [16] .
(A1) The total of the insect vector population is divided into X and Y, which denotes the densities of the susceptible vector and infective vector at time t, respectively. The total of the plant host population is divided into S, I, and R, which represents the numbers of the susceptible, infective, and recovered host plant population at time t, respectively. At the same time, we assume that the number of plants in one area is fixed. The total number of plants
is a positive constant. In fact, when a plant has died, it would be replaced by a new plant to keep the total number of plants. Further, we assume that those new plants are susceptible, i.e., we chose the birth rate of susceptible plant host as
.
(A2) The susceptible plants can be infected not only by the infected insect vectors but also by the infected plants.
(A3) A susceptible vector can be infected only by an infected plant host, and after it is infected, it will hold the virus for the rest of its life. Further, there is no vertical infection being considered.
(A4) The replenishment rate of insect vectors is a positive constant, and all of the new born vectors are susceptible.
According to the principle of the compartmental model, consider the following model with discontinuous treatment:
(2.1)
Here the dimensionless variables and parameters (with parameter values) are given in Table 1.
The function
represents the treatment rate.
satisfies the following assumptions. Obviously, the treatment rate should be nondecreasing as the number of infectious individuals is increasing. The following assumption will be needed throughout the paper.
(H1)
is nondecreasing and has at most a finite number of jump discontinuities in every compact interval. No loss of generality, we always assume that
is continuous at
, otherwise we define
to be
. Here
denotes the right limit of
as
.
By adding the fourth and fifth equations of system (2.1), we get
(2.2)
where
. From Equation (2.2), we easily get
as
Note that
. Since the variable R and X does not appear in the first two equations of model (2.1), meanwhile, let’s substitute X for
in
the fifth equation. We only need to study the first two equations and the fifth
![]()
Table 1. Dimensionless variables and parameters (with illustrative parameter values) in system (2.1).
equation of model (2.1), thereby lowering the order of the system to be studied, i.e.
(2.3)
where
. Obviously,
(2.4)
is the positively invariant set for system (2.3).
According to the definition of solutions for differential equations with discontinuous right-hand sides in [15] [17] ,
is called a solution with initial condition
(2.5)
of model (2.3) on
, if it is absolutely continuous on any compact subinterval of
, and almost everywhere on
(abbreviated to a.e. on
) satisfies the following differential inclusion:
(2.6)
where
. Here,
and
denote the left limit and the right limit of the function
at I, respectively.
From (H1), it is clear that the set map
(2.7)
is an upper semi-continuous set-valued map with non-empty compact convex values. By the measurable selection theorem [15] , if
is a solution of model (2.3) on
, then there is a measurable function
such that
(2.8)
3. Positivity
In this section, we will prove the positive of the solution to the initial condition of the model (2.3) with positive initial value. First, we will prove the following theorem.
Theorem 3.1. Suppose that assumption (H1) holds and let
be the solution with initial condition (2.5) of model (2.3) on
. Then
is nonnegative on
.
Proof: By the definition of a solution of (2.3) in the sense of Filippov,
must be a solution to differential inclusion (2.6). From the first equation of (2.6), we have
(3.1)
for all
.
Based on the previous hypothesis of (H1), we have
and
is continuous at
. Combining the continuity of
at
, it may be concluded that there exists a positive constant
such that
is continuous as
. On this account, when
the differential inclusion (2.6) becomes the following system of differential equations:
(3.2)
We divide this into four cases to discuss the positivity of the solutions for (2.6).
1)
.
From (3.2), we see that
for all
.
2)
.
By the continuity of
at
and
, we conclude
and
for all
. If it is not true, then we can set
(3.3)
If
, then from
for
, we have
. This is a contradiction.
If
, then there is a
such that
and
on
. Therefore, the second equation of (3.2) implies
(3.4)
We have
(3.5)
This is also a contradiction. Hence,
and
are positive for all
. The same conclusion can be reached for the following two cases.
3)
.
4)
. This completes the proof.
4. The Equilibria and Their Stability
In this section, we will discuss the existence of equilibria of system (2.3). First, we prove the existence of endemic equilibrium.
Let
is a constant solution of (2.3), where
satisfies the following system:
(4.1)
Since
, there always exists a disease-free equilibrium
of the model (2.3), where
. Next, we consider that the existence of an endemic equilibrium of the model (2.3).
It follows from the first and third equations of (4.1), we conclude that
(4.2)
Substituting (4.2) into the second inclusion of (4.1), we have the follows
(4.3)
where
Denote
(4.4)
and let
(4.5)
We next claim that
is the basic reproductive number for the model (2.3) which will determine the existence of an endemic equilibrium.
Theorem 4.1. Suppose that assumption (H1) holds. If
, then there only exists a disease-free equilibrium
. If
, then there exists a unique positive endemic equilibrium
except
.
Proof: By
, we get
. Since
is nonincreasing on I and
is nondecreasing on I. For this reason, the inclusion (4.3) is only valid at
. Hence, the model (2.3) has a unique disease-free equilibrium as long as
.
From (4.4), we have the following
(4.6)
where
If
, then
, and the Equation (4.6) has a unique positive root I, where
(4.7)
If
, then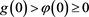 . Meanwhile, the inequality
. Meanwhile, the inequality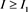 , it implies
, it implies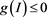 . Therefore, the set
. Therefore, the set
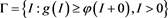 (4.8)
(4.8)
is bounded and non-empty. We can write
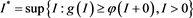
It follows easily that
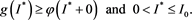 (4.9)
(4.9)
We claim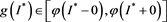 . Assumption, contrary to our claim, that
. Assumption, contrary to our claim, that
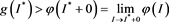
From (H1), there exists a 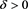 such that
such that
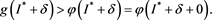 (4.10)
(4.10)
This contradicts the definition of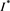 . Thus, we have
. Thus, we have 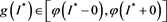 . That is to say,
. That is to say, 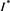 is a positive solution of the inclusion (4.3). We proceed to show that
is a positive solution of the inclusion (4.3). We proceed to show that 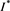 is the only one positive solution of the inclusion (4.3). If the inclusion (4.3) has another positive solution
is the only one positive solution of the inclusion (4.3). If the inclusion (4.3) has another positive solution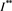 , then there must exist two numbers
, then there must exist two numbers
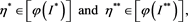 (4.11)
(4.11)
which satisfy
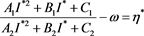 (4.12)
(4.12)
and
![]() (4.13)
(4.13)
Subtracting (4.13) from (4.12) gives
![]() (4.14)
(4.14)
which implies
![]() (4.15)
(4.15)
This is a contradiction. Hence, ![]() is the unique positive solution of the inclusion (4.3). Combining it with (4.2), we conclude that the
is the unique positive solution of the inclusion (4.3). Combining it with (4.2), we conclude that the ![]() is the unique endemic equilibrium of (2.3). The proof is completed.
is the unique endemic equilibrium of (2.3). The proof is completed.
Next, we prove the global stability of the disease-free equilibrium and the endemic equilibrium. We do this in several steps. We first investigate the local properties of the equilibria of system (2.3).
Theorem 4.2. Assume (H1) holds. The disease-free equilibrium ![]() is locally asymptotically stable if
is locally asymptotically stable if![]() , and is unstable if
, and is unstable if![]() .
.
Proof: We analyze the stability of the disease-free equilibrium by investigating the eigenvalues of the Jacobian matrix of model (2.3) at![]() . The matrix is
. The matrix is
![]() (4.16)
(4.16)
Thus, the characteristic equation at the disease-free equilibrium ![]() is
is
![]() (4.17)
(4.17)
It is easy to see that one of the roots with respect to ![]() of (4.14) is
of (4.14) is![]() . the other two roots are determined by the following characteristic equation of
. the other two roots are determined by the following characteristic equation of![]() :
:
![]() (4.18)
(4.18)
From (4.18) and Routh-Hurwitz criteria [18] , it is easily seen that both the real parts of ![]() and of
and of ![]() are negative when
are negative when![]() . When
. When![]() , one of
, one of ![]() and
and ![]() is a number with a positive real part. Thus the disease-free equilibrium is locally asymptotically stable if
is a number with a positive real part. Thus the disease-free equilibrium is locally asymptotically stable if ![]() and unstable if
and unstable if![]() .,
.,
We have shown that there exists a positive endemic equilibrium if and only if ![]() in Theorem 4.1. Here, we will establish its local stability.
in Theorem 4.1. Here, we will establish its local stability.
Theorem 4.3. Suppose that assumption (H1) holds. If![]() , the endemic equilibrium
, the endemic equilibrium ![]() of the system (2.3) is locally asymptotically stable.
of the system (2.3) is locally asymptotically stable.
Proof: The Jacobian matrix of (2.3) at the endemic equilibrium ![]() is
is
![]() (4.19)
(4.19)
Replacing![]() ,
, ![]() by
by![]() ,
, ![]() , respectively. So we have
, respectively. So we have
![]() (4.20)
(4.20)
The characteristic equation of ![]() is
is
![]() (4.21)
(4.21)
where
![]()
![]()
![]()
Since ![]() is nondecreasing,
is nondecreasing,![]() . This implies
. This implies![]() .
.
Then
![]()
![]()
Hence, all of the Routh-Hurwitz criteria are satisfied. Thus it follows that the endemic equilibrium ![]() of (2.3), which exists if
of (2.3), which exists if![]() , is always locally asymptotically stable. The proof is completed.,
, is always locally asymptotically stable. The proof is completed.,
Next, we will prove global stability of the disease-free equilibrium and endemic equilibrium of (2.3). We need to use the LaSalle-type invariance principle for the differential inclusion (Theorem 3 in [19] ) to prove their global stability.
Let![]() . We obtain the following system analogous to (2.5)
. We obtain the following system analogous to (2.5)
![]() (4.22)
(4.22)
Set
![]() (4.23)
(4.23)
and
![]()
For any![]() , there exists an
, there exists an ![]() such that
such that
![]() (4.24)
(4.24)
Hence,
![]() (4.25)
(4.25)
When![]() , the nondecreasing of
, the nondecreasing of ![]() implies
implies
![]() (4.26)
(4.26)
It shows that ![]() is a Lyapunov function of (4.23).
is a Lyapunov function of (4.23).
Furthermore, when![]() , we have
, we have
![]() (4.27)
(4.27)
When![]() , we have
, we have![]() , which implies
, which implies![]() . For any
. For any![]() , we set
, we set
![]() (4.28)
(4.28)
Hence, the largest weakly invariant subset of ![]() is the singleton
is the singleton![]() .
.
When![]() , we have
, we have
![]() (4.29)
(4.29)
From the first equation of (4.23) and x = 0, it may be concluded that![]() . Therefore, we see that the largest weakly invariant subset of
. Therefore, we see that the largest weakly invariant subset of ![]() is also the singleton
is also the singleton![]() . By the LaSalle-type invariance principle, the equilibrium
. By the LaSalle-type invariance principle, the equilibrium ![]() of (4.23) is globally asymptotically stable as
of (4.23) is globally asymptotically stable as![]() . Summarizing the above analysis, we obtain the following theorem.
. Summarizing the above analysis, we obtain the following theorem.
Next, we demonstrate the global stability of the endemic equilibrium ![]() of (2.3). So, we have the following theorem.
of (2.3). So, we have the following theorem.
Theorem 4.4. Suppose that assumption (H1) holds. If![]() , the disease-free equilibrium
, the disease-free equilibrium ![]() of the system (2.3) is globally asymptotically stable.
of the system (2.3) is globally asymptotically stable.
Proof: Let
![]() (4.30)
(4.30)
Write
![]()
and
![]() (4.31)
(4.31)
For any![]() , there exists an
, there exists an ![]() such that
such that
![]() (4.32)
(4.32)
Hence
![]() (4.33)
(4.33)
The monotonicity of ![]() implies
implies![]() . Thus
. Thus![]() . This shows that V is a Lyapunov function of (2.24). Define
. This shows that V is a Lyapunov function of (2.24). Define
![]() (4.34)
(4.34)
If![]() , then the first equation of (2.6) implies
, then the first equation of (2.6) implies![]() . Consequently, for any
. Consequently, for any![]() , the largest weakly invariant subset of
, the largest weakly invariant subset of ![]() of (2.6) is the singleton overline
of (2.6) is the singleton overline![]() . Here
. Here
![]()
Therefore, ![]() is globally asymptotically stable if
is globally asymptotically stable if![]() . This completes the proof.,
. This completes the proof.,
5. Numerical Simulation
To make our analysis more intuitive, some numerical simulations of solutions of the model (2.6) is provided which to illustrate the influence of insect vector and discontinuous treatment on the spread of plant disease. We apparent a treatment function satisfying (H1) as follows:
![]() (5.1)
(5.1)
where![]() . The treatment function
. The treatment function ![]() is applied at the following case: when the infective individuals I attain some threshold
is applied at the following case: when the infective individuals I attain some threshold![]() , the treatment rate should be strengthened to control the spread of the plant disease.
, the treatment rate should be strengthened to control the spread of the plant disease.
To better illustrate the effects of non-continuous healing on the spread of plant disease, the following parameters are derived from [16] . Let![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() , then we easily calculate
, then we easily calculate ![]() by using (4.5). Figure 1 shows that the infective individuals I and the infective insect vectors Y tend to 0, and it means that the disease goes to extinction. In addition, we find that the peak values of the infective is affected by the different values of
by using (4.5). Figure 1 shows that the infective individuals I and the infective insect vectors Y tend to 0, and it means that the disease goes to extinction. In addition, we find that the peak values of the infective is affected by the different values of![]() . Figure 1 shows that larger values of
. Figure 1 shows that larger values of ![]() can reduce the peak values of the infective. Therefore, we can increase the treatment rate to prevent the spread of disease after the number infective individuals reaching some high level. From Figure 1, the infective individuals reach some level and strengthening the treatment rate is also effective for disease control, even though we do not take any treatment measures at the initial time of the diseases outbreak.
can reduce the peak values of the infective. Therefore, we can increase the treatment rate to prevent the spread of disease after the number infective individuals reaching some high level. From Figure 1, the infective individuals reach some level and strengthening the treatment rate is also effective for disease control, even though we do not take any treatment measures at the initial time of the diseases outbreak.
If we fixed all parameter values as follows:![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() one could easily see that
one could easily see that![]() , by using (4.5). Figure 2 shows that the endemic equilibrium is globally asymptotically stable. By (4.5), it’s obvious find that the basic reproductive number
, by using (4.5). Figure 2 shows that the endemic equilibrium is globally asymptotically stable. By (4.5), it’s obvious find that the basic reproductive number ![]() is independent of
is independent of![]() , and the different values of
, and the different values of ![]() can affect the stability level of the infective. That is to say, larger values of
can affect the stability level of the infective. That is to say, larger values of
![]()
Figure 1. Stability of disease-free equilibrium. The parameters are fixed as follows:![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() , and the initial values are
, and the initial values are![]() .The time series charts for
.The time series charts for ![]() and the phase diagram are given in (a), (b), (c), and (d), respectively.
and the phase diagram are given in (a), (b), (c), and (d), respectively.
![]() can impact level of the infective. It implies that the strengthening of the treatment rate can effectively control the spread of plant disease after the number of infective individuals has increased to some high level.
can impact level of the infective. It implies that the strengthening of the treatment rate can effectively control the spread of plant disease after the number of infective individuals has increased to some high level.
6. Discussion
As for the plant infectious disease model, our main object is to investigate the effect of the insect vector and discontinuous treatment function on the dynamics of spreading the plant disease. We calculated the basic reproduction number![]() , which is derived under some reasonable assumptions on the discontinuous treatment function. It is an important threshold parameter which plays an important role in determining the global dynamics of the model (2.6) and whether it persists or dies out of the disease. When
, which is derived under some reasonable assumptions on the discontinuous treatment function. It is an important threshold parameter which plays an important role in determining the global dynamics of the model (2.6) and whether it persists or dies out of the disease. When![]() , the disease-free equilibrium is globally stable, which means that the disease always dies out, and when
, the disease-free equilibrium is globally stable, which means that the disease always dies out, and when![]() , the plant disease will be permanent which means that after some period of time the plant disease will become endemic and it is global stable.
, the plant disease will be permanent which means that after some period of time the plant disease will become endemic and it is global stable.
In this paper, we studied the existence, local stability and global stability of the disease-free equilibrium and endemic equilibrium of the system (2.3) in detail. By building a suitable Lyapunov function, and the Jacobian matrix method,
![]()
Figure 2. Stability of endemic equilibrium. The parameters are fixed as follows:![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
, ![]() ,
,![]() . The time series charts for S(t), I(t), Y(t) and the phase diagram are given in (a), (b), (c), and (d), respectively.
. The time series charts for S(t), I(t), Y(t) and the phase diagram are given in (a), (b), (c), and (d), respectively.
employing Routh-Hurwitz criteria and LaSalle-type invariance principle, the main results as shown in Theorems 4.2, 4.3 and 4.4 have been derived. Our main results indicate that if![]() , then the disease-free equilibrium is globally asymptotically stable, if
, then the disease-free equilibrium is globally asymptotically stable, if![]() , the unique endemic equilibrium is globally asymptotically stable of the system (2.3). From above results, it is easy to find that the basic reproduction number
, the unique endemic equilibrium is globally asymptotically stable of the system (2.3). From above results, it is easy to find that the basic reproduction number ![]() plays an important role in determining the persistence or dying out of the disease.
plays an important role in determining the persistence or dying out of the disease.
Acknowledgements
The research have been supported by The Natural Science Foundation of China (11561004), the Science and Technology research project of Jiangxi Provincial Education Department (171373, 171374, GJJ170815), The bidding project of Gannan Normal University (16zb02).