Influence of Cross-Inoculation on Groundnut and Bambara Groundnut-Rhizobium Symbiosis: Contribution to Plant Growth and Yield in the Field at Sarh (Chad) and Ngaoundere (Cameroon) ()
1. Introduction
In sub-Saharan Africa, particularly in the Sudano-Guinean savannahs, soils are generally depleted in nitrogen and organic matter by cash crops such as cotton. In these soils, crop production is limited by the lack of nitrogen and other mineral elements available to the plant (Bado [1] ). Population growth considerably reduces the area of arable land (Freeman et al. [2] ). All these phenomena create an imbalance between the quantity of agricultural products available and the food needs of the population on the one hand, and environmental management on the other. This implies that the population does not have enough to eat, therefore does not have a healthy and balanced life (Cooke [3] ).
Under these conditions, growers manage to enhance their crop yields through the use of chemical inputs that are costly, and potentially are polluting human and its environment (Acosta et al. [4] ). In developing countries such as Chad and Cameroon, cereal crops are the basis of diet, although its nutritional quality is deficient in protein (Tien et al. [5] ; Watier [6] ). This lack of protein has stimulated the search for new sources of protein that can supplement or substitute existing proteins (Ahmet and Abdallah [7] ).
To address this issue of food insecurity, soil infertility and environmental pollution, several cropping techniques such as crop rotation and intercropping (Gandebe et al. [8] ; Useni et al. [9] ), and nowdays, the use of organic fertilizers (Ngakou et al. [10] ; Ridine et al. [11] ), microbial biofertilizers such as rhizobia and mycorrhiza (Ngakou et al. [12] [13] [14] ) have been prioritized and applied. No research has yet been carried out to look particularly at the cross-inoculation Rhizobium-legume symbiosis in the Guinea and Sudano-savannahs. It is in this context that this research was designed to evaluate the effect of cross-inoculation with legume-nodulating Rhizobium on productivity of Bambara groundnut, groundnut commonly grown in Cameroon and Chad.
2. Materials and Methods
2.1. Experimental Sites Data
The experimental field sites were located at the University of Sarh in southern Chad (09˚04.51'N, 18˚25.22'E, at 375 m elevation), while in Cameroon, it was established within the University campus (07˚24.61'N, 13˚34.24'E, at 1155.8 m elevation). Trials were carried out during the cropping season extending from October 2015 to Febuary 2016 at Sarh-Chad (temperature: 25˚C - 39˚C, rainfall: 800 - 1200 mm/year), and from March to July 2015 at Ngaoundere-Cameroon (temperature: 22˚C - 28˚C, rainfall: 1500 - 2500 mm/year).
2.2. Biological Materials
Groundnut (Arachis hypogaea L.), a flower variety 11 provided by the Chadian Institute for Agricultural Research and Development (ITRAD) of Bebedja in Chad, has a life cycle of about 90 days. It is a plant with yellow flowers, pinnate leaves that give at maturity two or very rarely three seeds per pod. Some pods may have only a single seed. Bambara groundnut (Vigna subterranea (L.) Verd.), commonly known as voandzou was a local Djar variety, also supplied by ITRAD. It has 90 to 100 days at maturity, with seeds having beige black hile outline. Flowers are yellow in colour, leaves are trifoliolate, while pods give at maturity one, but very rarely two seeds. Rhizobium strains were produced as liquid inoculum from groundnut, Bambara groundnut cowpea, and soybean root nodules at the microbiology laboratory of the Institute for Agricultural Research and Development (IRAD) at Wakwa, Ngaoundere in Cameroon.
2.3. Evaluation of the Physico-Chemical Soil Parameters
Soils were sampled before sowing and after harvest. Physico-chemical analysis of soils was carried out following AFNOR method [15] at the Chadian Institute for Agronomic Research and Development (ITRAD) laboratory of N'Djamena.
2.4. Experimental Field and Treatments
The laboratory-adapted experimental set-up consisted of 2 randomized complete block fields, one field per legume crop species. Each block has 6 plots randomly arranged according to different treatments: control (Ctrl), Groundnut rhizobia (GR), Cowpea (CR), soybean rhizobia (SR), Bambara groundnut rhizobia (BR), and the Mixture of rhizobia (MR) from the 4 legume crop species. These six treatments were replicated uniformly in the three blocks numbered I, II, III, for a total of 18 plots per field trial. The experimental unit was a rectangular plan of (3 × 2) m2. Plots were separated 0.50 m apart, while blocks were separated by a distance of 1 m.
2.5. Sowing and Seed Inoculation
Control plots were sown first. Each plot had 7 lines with 9 sowing holes each, giving a total of 63 plants per plot. The sowing interval between plants was 25 cm, and 30 cm between lines (Ngakou et al. [14] ). The distance between the outer lines and the plot edges was about 25 cm. For each cross-inoculation formula- tion, seeds were coated with 100 ml of liquid inoculum solution, and 25 g of Nido milk serving as adhesive carrier. Different treatments were applied to seeds at sowing time. Weeding was done on the 12, 26 and 45 DAP with hoes and hands, while adding soften soil around each plant to avoid exposure of pods out of the soil surface.
2.6. Evaluation of Plant Growth and Yield Parameters
The plant size was measured from the crown to the last leaf at 30 and 60 DAP on 30 randomly selected plants per treatment (Zakaria [16] ). These measurements were performed early in the morning before 8 a.m. to avoid wind disturbances. Plants were dried at 65˚C in a P-Selecta oven (0˚C to 250˚C) for 48 hours and then, weighed using a Gilbertini brand scale at 0.001 g sensitivity as previously reported (Ngakou et al. [12] ).
2.6.1. Assessment of the Number and Dry Weight of Pods
Pods were harvested from 30 randomly selected plants per treatment. All pods harvested from the same plant were manually counted and stored into a labeled envelope. Groundnut and Bambara groundnut pods were dried at 65˚C for 48 h in the oven and weighed using a Gilbertini scale at 0.001 g sensitivity.
2.6.2. Determination of Seed Yield (Kg∙ha−1)
Seed yield expressed in Kg/ha was evaluated by the following formula:
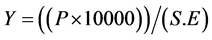
where, Y = dry weight of seeds in Kg/ha; P = dry weight of seeds per unit of experimental area; S.E = Experimental surface area (m²); 1 ha = 10,000 m2.
2.7. Statistical Analysis
All data were subjected to the Analysis of Variance (ANOVA) using the Stat Graphics plus software program. Treatment means were compared using the Duncan multiple Range Test at 5% significance. Correlations between variables were determined using the statistical package for social science (SPSS).
3. Results and Discussion
3.1. Physico-Chemical Characteristics of Soils before Sowing and after Harvest
Table 1 sumarizes the physico-chemical composition of soils prior to sowing of crops and after harvest. The soil of Sarh was sandy clay in texture, and was slightly rich in silt, nitrogen and organic matter compared to that of Ngaoundere, but the latter was rich in phosphorus with the C/N ratio 3 folds higher than that of Sarh soil before sowing. After harvest, the soil organic matter content was reduced by about 50% in Ngaoundere-Cameroon, while at Sarh-Chad, there was a slow increase in soil organic matter content. This result suggests that the cultivation of legumes on Cameroonian soil contributed to a loss of carbon stocks following the mineralization of organic matter. This mineralization would
![]()
Table 1. Variation of the physico-chemical characteristics of experimental soils before sowing and after harvest.
For each granulometric or organic matter parameter in a column and within a study site, values affected by the same letter are not significantly different at the indicated level of probability.
have been enhanced by favorable growth conditions to mineralizing bacteria present in the soil. This indicates that plants have used organic matter from the soil (Ngakou et al. [14] ). The decrease in the soil organic matter content is in agreement with other results obtained in the range of between 20% and 30% (Ngakou et al. [10] ). An increase of 20.66% in soil nitrogen content was observed after harvest at Ngaoundere-Cameroon compared to 3.27% at Sarh- Chad.
The enhanced soil C/N ratio by 40.34% in Chad after harvest justifies the increment of carbon in the soil. The phosphorus contents were 25.88% and 38.09%, respectively in Sarh and Ngaoundere soils, and this could be attributed to the presence of phosphorus solubilizing bacteria in the soil. At Sarh during harvest, soil pH increases towards neutrality, suggesting that free hydronium ions were fixed during the transformation of phosphorus complexes into assimilable phosphorus (Ndeye [17] ; Nwaga et al. [18] ).
3.2. Effect of Cross-Inoculation on Plant Size and Biomass at 30 DAP
At 30 DAP, groundnut plant size was improved by CR, MR and BR respectively by 5.93%, 17.79% and 19.49%, compared to the control at Sarh (Table 2). Plants
![]()
Table 2. Plant sizes and biomasses of groundnut and Bambara groundnut as influenced by treatments at 30 days after sowing (DAS).
For each parameter in a column, values affected by the same letter are not significantly different at the indicated level of probability. GR: Groundnut Rhizobia, CR: Cowpea Rhizobia, SR: Soybean Rhizobia, BR: Bambara groundnut Rhizobia, MR: mélange des quatre Rhizobia.
inoculated with BR, MR were significantly taller (p = 0.002) than those inocu- lated with GR, SR and uninoculated plants. The size of plant treated with cowpea rhizobia was significantly higher (p < 0.0001) than that of plant treated MR, as well as the control plants at 30 DAP in Ngaoundere.
Inoculation of Bambara groundnut plants with rhizobia accelerated their growth height (Table 2) at 30 DAP. This increment in plant size varied from 69.16% to 97.15% compared to that of untreated plants at Sarh. The size of plants treated with SR was more important than that of other treatments. This suggests that these bacteria may have contributed to the production of growth hormones that have accelerated the plant growth in size. Inoculation of cowpea plants with two strains of rhizobia was revealed to greatly increase the stem size (Abed Nour el Houda [19] ). Results obtained from this study line with other findings which reported plant size ranging from between 20 and 38 cm de- pending on the seed varieties used (Ndiang et al. [20] ).
At Ngaoundere, inoculation of Bambara groundnut with nitrogen-fixing bacteria resulted in a considerable improvement in plant size (Table 2) at 30 DAP, from between 25.28% and 88.57% compared to untreated plants. Plants treated with BR significantly have greater size (p = 0.0001) than plants of other treatments. This result is above those pointed out from a research performed in Adamawa (Gomoung [21] ). Inoculation of Bambara groundnut with rhizobia and mycorrhiza was claimed to positively improve the plant biomass (Ngakou et al. [14] ). The positive effect of inoculum on plant size can be explained by the production of plant growth hormones such as auxin and gibberellins (Vargas et al. [22] ). In a related research, soybean inoculated with Bradyrhizobium strain was revealed to exhibit greater biomass than non-inoculated plants through AIA production by bacteria symbiont (Afzal et al. [23] ).
3.3. Effect of Cross-Inoculation on Plant Biomass at 30 DAP
The use of the inoculum significantly improved (p = 0.006) the plant biomass of groundnut plant at Sarh (Table 2). The biomass of inoculated plants was more elevated and varied from 16.62% in CR to 45.45% in MR treated compared to that of uninoculated plants. The enhanced plant biomass was reported to be attributed to soils nutrient improvement by symbiotic bacteria (Ama-Abina et al. [24] ). At Ngaoundere, the plant biomass of untreated groundnut plants was significantly (p = 0.0001) higher than that of inoculated plants, indicating that indigenous rhizobia were more efficients on groundnut than in introduced strains. However, plants inoculated with CR and SR had higher dry biomass than those inoculated with GR and MR.
At Sarh, the most important Bambara groundnut plant biomass at 30 DAP was obtained from SR-inoculated plants. There was a highly significant difference between the biomass of CR, BR, MR, SR treated plants (Table 2). The inoculum improved the Bambara groundnut biomass from 39.33% to 87.86% as compared to that of the control. GR did not affect in anyway the Bambara groundnut biomass. This result lines with another finding which yielded an average biomass of 5 g per plant (Ngakou et al. [14] ).
At Ngaoundere rhizobia inoculum moderately improved the groundnut plants biomass (Table 2). BR treatment contributed to 73.70% improvement of bio- mass as compared to the control. There was a positive and significant correlation (r = 0.63, p = 0.0001) between the size and Bambara groundnut biomass.
3.4. Changes in Number and Pods Weight per Plant in Response to Cross-Inoculation
3.4.1. Pod Number
Results from Table 3 indicate that the number of pods (104 pods) per CR- inoculated groundnut plant was significantly greater (p < 0.0001) than that of other treatments at Sarh. Inocululation improved groundnut pod yield of all plants inoculated with rhizobia. This pods number per plant was superior to the range of between 28 and 37 pods per plant obtained in some uninoculated groundnut varieties during the rainfall cropping seasons (Betdogo et al. [25] ).
At Ngaoundere, the number of pods per individual inoculated groundnut plant (76) was higher (p < 0.0001) than in plants treated with CR, BR, and MR. A significant difference (p = 0.0001) was observed between the number of pods
![]()
Table 3. Number and dry weight of pods per Bambara groundnut and groundnut plants as influence by different rhizobia inoculation receipts at Ngaoundere-Cameroon and Sarh- Chad.
For each parameter in a column, values affected by the same letter are not significantly different at the indicated level of probability. GR: Groundnut Rhizobia, CR: Cowpea Rhizobia, SR: Soybean Rhizobia, BR: Bambara groundnut Rhizobia, MR: mélange des quatre Rhizobia.
of non-inoculated plants and the plants inoculated with CR. This enhanced number of pods could be attributed to efficient wild rhizobia strains that would have interacted rapidly with the host plant, while the introduced inoculants were less competitive. The high content of organic matter in the soil could have also accounted for these poor performances of rhizobial inoculant. These results line with other results reported from which the presence of straw compost attenuated the effect of the inoculum (Ndiaye [26] ). Inoculation enhanced the number of Bambara groundnut pods per plant at Sarh by 58.33% over the control. These results are lower than those reported in Bambara groundnut dual inoculated with rhizobia and mycorhiza in the field (Yao et al. [27] ), and may be attributed to high soil moisture during pods formation as previously pointed out (Ameyaw and Doku [28] ).
At Ngaoundere, the number of Bambara groundnut pods per plant (23) was higher in the CR-inoculated plants than in other treatments. The pods number was improved by 79.74% over the control.
3.4.2. Pods Dry Weight
The pod dry weight of groundnut plants inoculated with CR was higher than that of the control at Sarh. Inoculants improved the pod dry weight by 20.32% to 75.48% relative to untreated pods. The dry weight of pods was twice the one obtained in Senegal after seed inoculation with selected bacteria combined to phosphorus-gypsium intake (Dhery and Dreyfus [29] ).
At Ngaoundere the dry weight of groundnut pods per plant was higher (p = 0.0005) in plants inoculated with GR, SR and untreated plants than in plants treated with CR and MR (Table 3). This high dry weight of pods of untreated plants was due to the fact that some pods were filled with soil, confirming what was previously reported (Gomoung [21] ). The increase in the number of pods per plant positively impacted the dry weight of pods per groundnut plant, implying a positive and significant correlation (r = 0.28, p = 0.006) between the two parameters.
The dry weight of Bambara groundnut pods per plant varied with treatments at Sarh-Chad ranging from 11.1 g to 18.52 g depending on the treatment (Table 3). The decrease pods dry weight of Bambara groundnut in MR inoculated plants compared to that of the control was attributed the fact that some mature pods were not filled with seeds. Pod full containing seeds would depend not only on the instantaneous supply of carbon and nitrogen, but also the remobilization from the vegetative organs. The newly accumulated exogenous nitrogen was thus not sufficient to satisfy the high seeds requirements, and their growth rate then became metabolically associated with nitrogen remissions (Salon et al. [30] ). The pods dry weight of Bambara groundnut per plant was important in the RR treatment (Table 3) with 14.88 g per plant at Ngaoundere, the lowest dry weight accounting for SR-inoculated pods. Increasing the pods dry weight would have involved other factors such as pod size, pod filling in seed, and number of seeds per pod.
3.5. Variation of Seed Yield (kg∙ha−1) of Groundnut and Bambara Groundnut in Response to Cross-Inoculation with Different Rhizobia Strains
Seed yield (kg∙ha−1) of rhizobia inoculated groundnut was improved from 9.83% to 63.73% compared to the control at Sarh (Figure 1(a)). The highest yield was obtained from CR treated plants. This result was favored by the improvement of soil fertility through atmospheric nitrogen fixation (Dey et al. [31] ), and the solubilization of soil phosphorus by specific rhizobia, making it available for the host plant (Andriamananjara [32] ). In pure culture, inoculation of plant with Rhizobium was claimed to be an alternative to improve plant growth and productivity (Diop et al. [33] ).
At Ngaoundere, the enhanced seed yield in groundnut inoculated treatments was attributed either to improvement of soil fertility through atmospheric
![]() (a)
(a) ![]() (b)
(b)
Figure 1. Variation of seed yield of groundnut and Bambara groundnut kg/ha at at Sarh- Chad (a); and Ngaoundere-Cameroon (b). For each of the plant species, bars affected by the same letter are not significantly different at the level of probability indicated.
nitrogen fixation (Badawi et al. [34] ), or the soil phosphorus solubilizing rhizobia (Mekhemar et al. [35] ). Other reported results have indicated seed yield of between 975 kg∙ha−1 and 1.432 t∙ha−1 (Ndiaye [26] ), bellow our findings. Seed yield per hectare of untreated plants (1.57 t∙ha−1) was found to be statistically equal to the average pod yield (1.63 t∙ha−1) of five cultivars of groundnuts introduced into northern Cameroon (Betdogo et al. [25] ).
Results on Figure 1 indicate that Bambara groundnut seed yield was in the range between 491.25 and 910.375 kg∙ha−1 depending on the treatments at Sarh. The performance of the BR treatment was greater (p < 0.0001) than that of the control. BR improved the seed yield of Bambara groundnut by 72.71%. These results are in agreement with recent findings which reported yield values varying between 300 to 800 kg∙ha−1 (Brink [36] ), or between 524.25 kg and 912.1 kg∙ha−1 in Bambara groundnut dually inoculated in the field with rhizobia and mycorhiza (Ngakou et al. [14] ), but are twice those from which seed yield of between 130 to 470 kg∙ha−1 in was obtained (Salon et al. [30] ).
The highest seed yield was that of CR treatment (699.77 kg∙ha−1), whereas the lowest was observed (Figure 1(b)) in SR treatment (327.26 kg∙ha−1) at Ngaoun- dere. Rhizobial inoculation at sowing was reported to increases seed dry weight and yield (Mnasri et al. [37] ). This yield increment was attributed to the improvement in mineral nutrition of plants, as well as seeds content in N and P, following inoculation (Ngakou et al. [12] ). Similar to our findings, grain yield varying from 485 to 1322 kg∙ha−1 depending on Bambara groundnut varieties has been reported (Pungulani et al. [38] ).
3.6. Links between the Yield Parameters
The affinity between yield parameters of each studied crop was express through the Pearson correlation (Table 4). A positive and significant correlation (r = 0.5, p < 0.0001) was detected between the number of seeds and the dry weight of the pods at Ngaoundere. At Sarh, the correlation between the number and dry weight of pods was positive and highly significant (r = 0.55, p < 0.0001). Similarly, the the pod weight highly and significant correlated (r = 0.59, p < 0.0001) with the number seeds per pod on the same site. On the overall, all the yield parameters within each of the studied site positively and significantly correlated each other.
4. Conclusion
This study has indicated that cross-inoculation did contribute to improve soil fertility (nitrogen and phosphorus content) and yield of groundnut and Bambara groundnut in the field. Improved seed yield expressed in kg/ha varied from 9.83 to 63.73% in groundnut, and to 72.71% in Bambara groundnut. These results suggest that groundnut and Bambara groundnut have a symbiotic affinity with rhizobia from other legume crop species. Mixture of Rhizobium from several crop legume species may not be indicated for Bambara groundnut, whereas GR, CR, SR, BR and MR could be used to improve seed yield of groundnuts. For
![]()
Table 4. Correlation between yield parameters of the two crop legumes.
large-scale and sustainable agricultural production, it would be imperative to produce these inoculants in solid form and make them available to farmers.
Acknowledgements
The authors are thankful to all those who, in one way or the other, have contributed to the improvement and proof-reading of this manuscript. The financial support of the University of Sarh-Chad allocated to PhD students within the training of trainers program is particularly appreciated.