1. Introduction
The resin composite restoration is widely used due to the spread of the concept of Minimal Intervention (MI) advocated by the Federation Dentaire Internationale (FDI) [1] [2] . It is necessary to think about the risk of secondary caries after restoration because dental plaque is formed on the surface of a resin composite [3] . The surface components of a resin composite may play an important role in the adherence of bacteria to the composite. The adhesion of bacteria to a surface represents an important and the initial stage of dental plaque formation [4] . Plaque does not form directly on the surfaces of a tooth or resin composite but requires first the adsorption of proteins to such a surface, resulting in a layer known as the acquired pellicle. The composition of dental pellicles is mainly based on the selective adsorption of salivary proteins [5] [6] .
The surface properties of resin composite material related to bacterial adhesion and plaque formation are affected by the components of the material [7] . A resin composite containing S-PRG filler has unique surface properties compared with other resin composites used in the oral cavity. S-PRG filler particles are produced by pre-reacted glass-ionomer (PRG) technology [8] . With this technology, a glass-ionomer phase is formed on glass particles through the reaction of fluoro-boro-alumino-silicate glass with polycarboxylic acid in the presence of water [9] . An S-PRG filler has fluoride release and recharge ability equivalent to those of glass-ionomer cements [10] . Further, the S-PRG filler was also found to release inorganic elements such as Al3+, Sr2+, Na+, 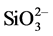 , and
, and 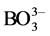 [11] .
[11] .
In this present study we examined the adsorption of various salivary components to S-PRG resins and determined how each component affected the adhesion of S. mutans to the S-PRG resin surface. In the oral environment, all solid surfaces are covered by an acquired salivary pellicle. The pellicle on tooth enamel and restoration materials is formed by the selective adsorption of salivary proteins. In this viewpoint, the physicochemical properties of a material influence microbial adhesion either directly or through adsorption of salivary proteins [12] [13] [14] . Therefore, there is the possibility for the composition of the pellicle film formed on the surface of an S-PRG resin to influence accumulation of bacteria in the plaque, and the occurrence of secondary caries. The working null-hypothesis was that the amounts of salivary protein adsorption and adherence of S. mutans to investigate S-PRG resin composites would not vary between composites containing different amount of S-PRG filler.
2. Materials and Methods
2.1. Specimen Preparation
Materials used in this study are listed in Table 1. Resin blocks containing 0 wt% S-PRG resin (unfilled resin), 30 wt% S-PRG resin (total inorganic filler contained 49.2 wt%) and 60 wt% S-PRG resin (total inorganic filler contained 79.2 wt%), were used in this study. These materials were placed into a metallic mold (4 × 4 × 1 mm) and covered with a micro-slide glass. After irradiation for 60 seconds using a visible light curing unit (Wave length: range 370 to 520 nm with
![]()
Table 1. Resin composite used in this study.
*Containing catalyst.
peak at 470 nm) (Coltolux50, Yoshida Corp., Tokyo, Japan), the specimens were gently polished with 2000 grid silicon carbide abrasive paper. The polished specimens were cleaned by ultrasonic cleansing for 30 minutes at room temperature and stored at 4˚C in sterilized distilled water before any tests were performed.
2.2. Adjustment of Various Saliva Proteins
Five kinds of salivary proteins 1) Mucin 1; 200 mg/ml, Sigma-Aldrich, St. Louis, MO, USA; 2) Lactoferrin; 40 μg/ml, Santa Cruz Biotechnology, Dallas, Texas, USA; 3) IgA; 200 μg/ml, Abcam, Cambridge, UK; 4) Cystatin C; 40 μg/ml, Upstate Biotechnology, New York, USA; 5) Lysozyme; 200 μg/ml, Sigma-Aldrich) were used for the salivary-protein adsorption test. Each salivary component was diluted with sufficient phosphate-buffered saline (PBS; pH 7.4) to approximate its content in human whole saliva.
2.3. Salivary-Protein Adsorption Test
Each resin block was suspended in a test tube with 1 ml of the salivary protein fluid and incubated at 37˚C for 2, 8 or 24 hours. Absorbance (OD280) of the salivary protein fluid was measured by using a spectrophotometer (U-2000, HITACHI, Tokyo, Japan), and the amount of salivary protein that adsorbed to a 1 mm2 area of the resin block was calculated from the amount of the absorbance of the free protein. Absorbance of the salivary protein solution prior to incubation was taken as a control.
2.4. Bacteria Adhesion Test (Quantitative Adherence of Radiolabeled Bacteria)
S. mutans ATCC 25175 was anaerobically inoculated into 50 ml of Trypticase Soy Broth containing 0.5% yeast extract and 74 kBq of [6-3H] thymidine (GE Healthcare, Wauwatosa, Wisconsin, USA) and cultured at 37˚C for 18 hours. The cells were collected by centrifugation at 8000 g for 20 minutes with 0.05 M phosphate-buffered saline (PBS; pH 7.0) at room temperature, and the radiolabeled bacteria were adjusted with PBS to a concentration of 108 CFU/ml.
Each resin block, which had been soaked in one of the five kinds of salivary protein solution for 2 hours, was suspended in a beaker with 260 ml of the labeled bacterial fluid at 37˚C for 1 hour. To remove the non-adhering bacteria, the resin blocks were removed from the beaker and immediately washed 3 times with PBS. Labeled bacteria that had adhered to the resin blocks were quantified by using automatic sample combustion equipment and a liquid scintillation counter (LSC-903, Aloka, Tokyo, Japan). Resin blocks not soaked in the salivary proteins were the control.
2.5. Observation under a Scanning Electron Microscope (SEM)
The resin blocks subjected to the bacteria adhesion test were pre-fixed with 2% glutaraldehyde for 1 hour at 4˚C, washed twice with a buffer (0.1 M sodium cacodylate) at pH 7.4, fixed with 1% osmium tetroxide for 30 minutes at 4˚C, and washed twice with the same buffer. Finally, the specimens were dehydrated with alcohol and isoamyl acetate, dried with CO2 by critical point drying, and observed under the SEM.
2.6. Statistical Analysis
Differences in measured values (n = 4) among all S-PRG resins were tested by one-way analysis of variance (ANOVA) and a post hoc test (Fisher PLSD test) for multiple comparisons. A probability of less than 0.05 for similarity of distribution was considered to indicate a significant difference.
3. Results
3.1. Salivary Protein Adsorption
The absorbance (OD280) of the salivary protein fluid decreased with the time of incubation of each of the 5 kinds of salivary protein. That is, it was suggested that the salivary proteins had adsorbed to the resin block with an increase in incubation time. Moreover, the amount of adsorption of the salivary proteins fluid increased at the rate of the content of S-PRG filler. The adsorption of the salivary proteins to the S-PRG resin block (especially 30%, 60%) soaked in Cystatin C or Lysozyme was significantly (p < 0.05) higher than that of those soaked in the other salivary proteins (Figures 1(a)-(e)). The value for the adsorption of Mucin 1 to the resin block surfaces (μg/mm2) was greater than that for the other salivary proteins (Figure 2).
3.2. Adhesion of S. mutans (Quantitative Adherence of Radiolabeled Bacteria)
The values for disintegrations per minute (dpm) were significantly (p < 0.05) lower for all resin blocks soaked in any of the five kinds of salivary protein than for those for the unsoaked blocks (control). No significant difference in dpm was obtained with respect to filler content rate, though the dpm value for Mucin 1 and Lysozyme tended to decrease with the increasing S-PRG filler content rate. In addition, the dpm value for Mucin 1 was lower than that for the other four kinds of salivary protein, but not significantly so (Figure 3).
![]()
Figure 2. Amount of salivary proteins adsorbed per 1 mm2 area of resin block surface.
![]()
Figure 3. Quantitative adherence of radiolabeled S. mutans (No significant difference was found between specimens having the same characters. MUC: Mucin 1, LAC: Lactoferrin, CYS: Cystatin C, LYZ: Lysozyme).
3.3. SEM Observation
Adhesion of S. mutans was found on the surfaces of all resin blocks. However, reduced adhesion was observed for the resin blocks that had been incubated with any of the salivary proteins (Figures 4(a)-(f)). Figure 4(b) shows the SEM of S. mutans that had adhered to S-PRG resin coated with Mucin 1. Little adhesion found with S. mutans regardless of the content rate of the S-PRG filler. Moreover, the number of S. mutans that had adhered to the Lysozyme-coated resin tended to decrease with an increase in S-PRG filler content rate.
4. Discussion
The current study found that the amount of the salivary protein adsorption varied according to the content of S-PRG filler. Therefore the null hypothesis was rejected. The surface properties of the material used for caries treatment are always influenced by the saliva in the mouth. More than 300 kinds of bacteria exist in the human mouth, and a biofilm is formed on the teeth as a dental plaque [15] . Therefore, it is necessary to know the relationships among the restoration materials, saliva and the plaque in studying about dental materials [16] [17] .
The characteristics of the resin composite containing an S-PRG filler, are that salivary proteins are more readily adsorbed than resin composites without S-PRG filler, and the amount of plaque formation is low [18] [19] [20] . In our past in vitro study, electrophoresis analysis (SDS-PAGE) revealed that a resin composite containing the S-PRG filler has the property of adsorbing salivary proteins of 25 kDa or less, whereas the S-PRG filler alone can adsorb salivary proteins of up to 14 kDa [21] . Moreover, the main proteins detected in vivo on the surface of a resin composite containing the S-PRG filler as determined in an immunity SEM method were Cystatin C and Lysozyme [21] . Therefore, we considered the possibility of plaque formation could be influenced by salivary protein in the mouth that is adsorbed on the surface of a resin composite containing S-PRG filler. In our in vitro study, the amount of adsorption of various salivary proteins was measured by using three kinds of resin composite, each having a different content of the S-PRG filler. We also examined the relationship between bacteria adhesion to the resin composite and the salivary protein covering it.
The absorbance (OD280) of the salivary protein fluid decreased with increasing incubation time for each of the five kinds of salivary protein tested. Regarding the amounts of each salivary protein that adsorbed to the resin block (μg/mm2) regardless of the content of the S-PRG filler, we calculated that Mucin 1 gave the greatest adsorption, followed by Lysozyme. That is, there was almost a correlation between the amounts for various salivary proteins and simulated human whole saliva in this study. Moreover, the amount of adsorption of the Lysozyme increased as the content of the S-PRG filler was increased. The means of interaction between Lysozyme and hydroxyapatite in the mouth is thought to be an electrostatic attraction; because the tooth surface becomes negatively charged when Ca2+ is discharged and Lysozyme, which is positively charged, is then attracted to the teeth [22] . On the other hand, the resin composite containing
S-PRG filler discharges various inorganic ions (Al3+, Sr2+, Na+, 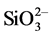 ,
,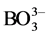 ) in the solution such as saliva, and the amount of discharge is proportional to the content of the S-PRG filler. Therefore, as the ionization potential is high in the case of the resin composite with a high content of the S-PRG filler, the positively charged Lysozyme may unite with the part of resin composite containing the negatively charged S-PRG filler.
) in the solution such as saliva, and the amount of discharge is proportional to the content of the S-PRG filler. Therefore, as the ionization potential is high in the case of the resin composite with a high content of the S-PRG filler, the positively charged Lysozyme may unite with the part of resin composite containing the negatively charged S-PRG filler.
We examined bacterial adhesion by using radiolabeled S. mutans and by SEM observation. Though the bacteria adhered to all the resin blocks coated with any of the salivary proteins, their adherence was less than that in the control group. Moreover, the resin composites coated with Mucin 1 showed less adhesion than those coated with the other salivary proteins. Adhesion of the radiolabeled bacteria to the Lysozyme-coated composite increased with an increase in the S-PRG filler content, and the number of adherent S. mutans seen by SEM observation tended to decrease with the increase in this content. These two experimental results approximated one another, indicating that the amount of adsorption and the kind of the salivary protein influenced the adhesion by S. mutans.
The initial conditioning salivary coat plays an important role in bacterial adhesion to a restorative surface [23] . However, it must be pointed out that although the acquired pellicle itself is free of bacteria [24] , it is the starting point for microbial colonization on oral hard surfaces, whereby the salivary pellicle acts as a receptor for the initial adhesion of bacteria. Indeed, the formation of oral biofilms on hard surfaces is a complex process that begins with salivary pellicle formation and pellicle adsorption to the surface, and then progresses to passive transport of bacteria to the pellicle surface, followed by irreversible adhesion and multiplication of the attached organisms [25] .
Among the five kinds of salivary proteins tested in this study, S. mutans adhesion to resin composites coated with any of these salivary proteins was low. In addition, in the case of Lysozyme there was somewhat of a correlation between the increases in bacterial adhesion with increasing S-PRG filler content. Many anti-bacterial proteins are found in human whole saliva, and Lysozyme is classified as a non-immunoglobulin protein [26] . Moreover, adsorbed Lysozyme is effective in preventing Streptococcus from adhering to the hydroxyapatite covered by saliva [27] . Our finding suggest that the S-PRG filler used in a dental restoration material exposed to endogenous Lysozyme, which is an anti-fungoid salivary protein that is selectively adsorbed, is very profitable to prevent secondary caries.
5. Conclusion
The adherence of S. mutans was low when the resin blocks were soaked in any of the five kinds of salivary proteins tested regardless of the content of the S-PRG filler. In addition, the amount of adsorption of Lysozyme to the composite increased with increasing content of the S-PRG filler. Moreover, the adhesion of S. mutans to the surface of a resin block coated with Lysozyme seemed to become lower with an increase in the content of the S-PRG filler, though no significant difference was found.
Acknowledgements
The authors would like to thank Shofu Inc. for providing the materials needed.