Flow Cytometer Performance Characterization, Standardization and Calibration against CD4 on T Lymphocytes Enables Quantification of Biomarker Expressions for Immunological Applications ()
1. Introduction
Flow cytometry allows simultaneous measurement of the light scatter properties of different cell types as well as the fluorescence intensities of distinct fluorophores attached to these cell types. It is a powerful analytical tool that has been widely used to perform immunophenotyping, cell counting and numerous cell function assessments, such as intracellular cytokine production, protein phosphorylation, cell proliferation and apoptosis.
Development of quantitative flow cytometry (QFC) has been urged for clinical biomarker standardization because accurate and reproducible results will greatly aid our understanding of diseases, enable timely diagnosis and allow proactive monitoring of therapeutic and off-target drug side effects. Standardization of flow cytometryin hematological malignancies has been a great challenge. A few published studies describe standardization of data at the analytical stage by exchanging the list mode files to harmonize and standardize the analysis of minimal residual disease in multiple study groups [1] -[4] . Letestuand co-workerse valuated ZAP-70 expression in Chronic Lymphocytic Leukemia (CLL) in a multicenter international harmonization process [5] . Kraan et al. [6] presented general rules for cytometer setup using up to four colors in clinical settings using analog flow-cytometric systems, and Shankey et al. [7] reported complete standardization of 4-color ZAP-70 investigation among three institutions. Additionally, the Euro Flow Consortium demonstrated successful standardization of flow cytometer settings and measurement protocols for immunophenotyping in a multicenter study [8] .
Quantitative flow cytometry has been demonstrated in a single laboratory study [9] -[11] . However, multicenter quantitative studies including various flow cytometer platforms have not yet been reported, suggesting the enormous difficulty of the task. This type of study requires both multi-platform instrument standardization and the use of a biological reference marker with a known expression level for ultimate biomarker quantification. The instrument standardization includes multiple steps, e.g. characterization of each instrument performance, target instrument identification, optimization of the target instrument fluorescence detector voltages as well as the application of the median fluorescence intensity (MFI) values from the target instrument to other instrument platforms. Once all flow cytometers are standardized, cell biomarker expression levels can be quantified with a reference biomarker, e.g. CD4 on normal human T lymphocytes with a known number of the antibody bound per cell (ABC) value [12] -[14] . In the present study, we have developed an instrument standardization procedure across three different flow cytometer platforms from the same vendor with somewhat different optical configurations and in two different locations. After the standardization of these instruments, CD19 quantification is carried out to validate the ABC quantification approach and demonstrate that consistent and reliable results can be accomplished between instruments using the developed procedure.
2. Material & Method
Four flow cytometry instruments from the same manufacturers in two different laboratories were used in this study. Instrument 1 (FACSCanto II), Instrument 2 (LSRFortessa), Instrument 3 (FACS Aria III) and Instrument 4 (FACSAria II) are all from BD Biosciences (San Jose, CA). The first three Instruments are located at FDA and the fourth instrument is located at NIST. For this study all four instruments were configured with three lasers: blue (488 nm), red (633 or 640 nm) and violet (405 or 407 nm) lasers (Table 1(a)). These instruments have flexible and compatible optical configurations with the exceptions described in Table 1(b), which could potentially be used in diagnostic laboratories. Cytometer Setup & Tracking (CS & T) beads were from BD Biosciences; they are composed of equal concentrations of dim, mid, and bright dye embedded polystryrene beads [2]
 (a)
(a)  (b)
(b)
Table 1. (a) Laser excitation wavelength for instruments used in the study; (b) Optical configuration and filter specifications for instruments used in this study.
μm (dim bead), 3 μm (mid and bright beads)]. The same master lot of CS & T beads was used throughout the study to define the base line for each instrument and daily QC/QA for the instrument. In addition, the bright bead population wasutilized to determine the target MFI values that were transferred to the other flow cytometers. Ultra Rainbow bead kit (Catalog #: URCP-38-2K) containing a blank and five different fluorescence intensity populations was acquired from Spherotech Inc (Lake Forest, IL) and used to calibrate the linearity of the fluorescence channels used for biomarker quantification in the unit of equivalent reference fluorophore fluorescence (ERF) [10] .
Anonymous heparinized normal donor samples were obtained from NIH’s Department of Transfusion Medicine according to the institutional review boards (IRB) guidelines. The unstained normal donor blood was used to adjust the Photomultiplier tube (PMT) voltage for each fluorescence detector to adequately segregate the auto-fluorescent negative signal from instrument electronic noise in such a way that dim fluorescent events can be discriminated from the true negative. Singly stained normal donor blood was used to ensure that the brightest populations are within the dynamic scale of each detector and to derive the compensation matrix. Lastly, multicolor stained normal donor blood was employed to compare blood cell subset typing results across the selected platforms and determine CD19 expression levels on B lymphocytes.
3. Results & Discussion
3.1. Selection of a Target Flow Cytometer
A two-step approach was used for the selection of a target cytometer [15] : 1) Forall cytometers to be standardized, a baseline performance was carried out for instrument performance characterization. The robust standard deviation of electronic noise (rSDEN), linearity maximum (Lin Max), and linearity minimum (Lin Min) values were determined for each detector as provided from the baseline performance report; 2) A target cytometer was determined across multiple platforms based on the highest rSDEN and lowest Lin Max for each detector (Table 2). On the basis of these two criteria, Cytometer (1) was identified as the primary target cytometer for this study.
3.2. Photomultiplier (PMT) Voltage Adjustment
An unstained normal donor sample was first used to adjust PMT voltage for each fluorescence detector. Briefly, 10 ml of heparinized whole blood was washed twice with phosphate buffer saline (PBS; pH 7.4) containing 5% Fetal Bovine Serum (FBS) (Sigma-Aldrich, St Louis, MO). The sample was then lysed using 1X FACS Lysing

Table 2 . Cytometer characterization based on electronic noise robust standard deviation (rSDEN) and linearity maximum (Lin Max).
Solution (BD Biosciences) according to the manufacturer’s recommendations. After centrifugation, the cell pellet was washed twice with 5% FBS/PBS and re-suspended in 2 ml of the same buffer. On the flow cytometers, the FSC/SSC gate for lymphocytes was used and the PMT voltages of the primary target instrument were adjusted to established voltages in such a way that the robust standard deviations of the autofluorescence signals from unstained cells are within 2.5 to 3 times the rSDEN of each detector to be used in the analysis. These optimized PMT voltages were further verified using singly stained blood control stained with either CD45 or CD3for each channel to ensure that the brightest populations are within the dynamic scale of each detector. In summary, the PMT voltages were adjusted so that negatively stained cell populations were located above the electronic noise on the low end, and the positively stained cell populations were within the linear range of the detector for each fluorochrome used.
3.3. Target Values Determination and Application for Multiplatform Standardization
Using the optimized PMT values obtained from the previous step, the bright CS & T bead population was gated and its fluorescence intensities were recorded in all channels of the primary target flow cytometer. These intensity values were sed as preliminary “Target MFIs” (See Supplement Figure S1) and transferred to all other companion instruments utilized in this study. After each companion cytometer passed its routine CS & T performance check, the PMT voltages in these instruments were adjusted in such way that the same bright bead population has equal or nearly equal “Target MFI” in each detector. In other words, the bright bead MFI values were copied from the primary instrument to other cytometers to be standardized by adjusting PMT voltages to bring the same bright bead peak to the target MFI of each detector. The adjusted PMT voltages of all instruments were recorded and followed on a daily basis to monitor instrument stability. With the recorded PMT voltages applied, the MFI of the bright CS & T bead population in each fluorescence channel of any cytometer used in the study was monitored on the day of use with the corresponding target MFIs for that instrument after the instrument standardization process was completed. Variations of only a few volts were acceptable. Whenever instrument performance failed on any instrument, measures such as thorough cleaning, de-gassing flow cell and laser delay verification were taken. When the performance was not restored to pass the monitoring criteria, a service visit was requested. Note that if the target MFI setting resulted in suboptimal PMT setting on any instrument, the target MFI values were adjusted accordingly on the target flow cytometer until consensus target MFI values were reached under optimal PMT settings for all instruments.
3.4. Verification of Instrument Standardization by Single Tube Analysis of Blood Cell Subsets
Sample Staining Procedure: To validate the cytometer standardization procedure and ensure data comparability across all four cytometers, a one ml of heparinized whole blood samples was used at each site within 6 - 8 hours after venipuncture. Prior to staining, the whole blood was washed twice with phosphate buffer saline (PBS; pH 7.4) containing 5% FBS. The sample was then stained in one location with the following seven reagents: CD20 eFluor 450 (eBiosciences, San Diego, CA), CD45 V500, CD3 APC-Cy7, CD5 PE-Cy7, CD4 PE, CD8 APC, CD19 PerCP-Cy5.5 (BD Biosciences) for 30 min in the dark. After staining, the cells were washed twice and lysed using 1X FACS Lysing Solution. After one wash, cells were fixed in 1% paraformaldehyde and acquired within 1 - 2 hours of cell fixation at both locations. Unstained control cells and singly stained tubes for the eight fluorochromes (FITC, PE, PerCP-Cy5.5, PE-Cy7, APC, APC-Cy7, eFluor 450 and V500) were also prepared and used to set the flow cytometer compensation. The number of events collected in each sample of these experiments varied from 500,000 to 1 million events.
Blood Cell Subset Typing: The stained normal donor sample was acquired in the four optimized and standardized flow cytometers at a “low” flow rate mode within the first 2 hour after sample preparation. Data were subsequently analyzed using the FACSDiva V6software. A doublet exclusion gate based upon (FSC-A vs. FSC-H) was utilized to gate on singlet cells, and within this singlet cell gate, a lymphocyte gate was drawn in a dot plot of FSC-A vs. SSC-A (Supplement Figure S2). Using a dot plot of CD45 vs. SSC-A under the lymphocyte gate, aR1 gate was drawn around the CD45 positive cells and used for all subsequent gating. Figure 1 shows comparative dot plots of CD3 vs. CD20 and CD20 vs. CD5 and Figure 2 displays dot plots of CD45 vs. CD3 and CD45 vs. CD4, both under R1 gate. The percentage of CD3, CD4, CD8, CD19, and CD20 were obtained and provided in Table3
Under the CD45+ cell gate, the percentage of CD3 positive cells was 53.8% for instrument 1, 53.1% for instrument 2, 53.7% for instrument 3, and 53.3% for instrument 4, respectively. Under the same CD45+ main gate, the percentage of CD4 positive cells was 33% for instrument 1, 32.7% for instrument 2, 32.1% for instrument 3, and 32.6% for instrument 4, respectively. For CD8 expression, the percentage obtained was 22.2%, 19.4%, 20%, and 20.6% for instruments 1 to 4, respectively. The percentage of CD19 positive cells was 29.6%, 31.7%, 32.5%, and 30% for instruments 1 to 4, respectively. Lastly, for cells expressing CD20 the percentage obtained was 28.2%, 31.8%, 28.9%, and 31.5% for instruments 1 to 4, respectively.
When gated on CD3 positive cells, the percentage of CD4+ cells was 53.3%, 58%, 58.8%, and 57.6% for instruments 1 to 4, respectively; for cells with CD 8 expression the percentage obtained was 35.5%, 34.9%, 35%, and 36.7% for instruments 1 to 4, respectively. The results on the percentages of the blood cell subsets are reasonably consistent among the four instruments demonstrate that these instruments are standardized with the procedure implemented. The slightly larger coefficient variations of the mean percentages derived for CD8+, CD19+ and CD20+ cells under the CD45+ cell gate (Table 3) might be related to difference in band path pass filters equipped, e.g. in PerCP Cy5.5 and APC channels (Table 1(b)), and wider expression of CD45 on many different blood cell types.
3.5. Biomarker Quantification
Quantification Scheme: Three different flow cytometer platforms (instrument 1 to 3) at one location were used for CD19 quantification, a B lymphocyte marker after the three instruments were standardized. The quantification scheme using CD19 APC-Cy7 as an example is illustrated in Figure 3. The linearity of APC-Cy7 channel is assured by using Ultra Rainbow beads for linearity calibration in the unit of the equivalent APC fluorescence (EAF) [10] (square symbol). Using CD4 APC-Cy7 as a reference biomarker with a known antibody bound per cell (ABC) value of 45,000 for fixed whole blood [14] , the linear fluorescence calibration scale (left Y-axis in Figure 3) obtained by using Rainbow beads is transformed to the ABC scale (right Y-axis). The quantification of CD19 APC-Cy7 is then carried out using the converted ABC calibration on the right Y-axis. With this relative quantification scheme, it is assumed that different antibodies against different antigens have very similar average fluorescence per antibody values because both CD4 APC-Cy7 and CD19 APC-Cy7 are from the same vendor such that the same conjugation chemistry and process control for APC-Cy7 labeling were applied. It is critical that the labeled antibodies are free of unconjugated APC-Cy7 fluorophores due to likely higher binding affinity for unconjugated antibodies than fluorophore conjugated antibodies. This assumption is further tested in this study using different fluorophore labeled antibodies, CD4 PerCP-Cy5.5 and CD19 PerCP-Cy5.5. Note that monoclonal antibody affinity issue on both CD4 reference marker and the marker of interest has not been taken into account. However, high affinity monoclonal antibodies against specific antigen epitopes are generally used for quantitative flow cytometry measurements in terms of ABC values.
Quantification of CD19 Expression: Four tubes were stained with either CD19 PerCP-Cy5.5, CD4 PerCPCy5.5, CD19 APC-Cy7 or CD4 APC-Cy7 together with CD45 V500. A doublet exclusion gate based upon (FSC-A vs. FSC-H) was utilized to gate on only singlet cells (Supplement Figure S3). Within this singlet cell gate, a lymphocyte gate was drawn in a dot plot of FSC-A vs. SSC-A. Under the lymphocyte gate a dot plot of CD45 vs. SSC-A were used to gate on CD45 positive cells and used for all subsequent gating. Dot plot of
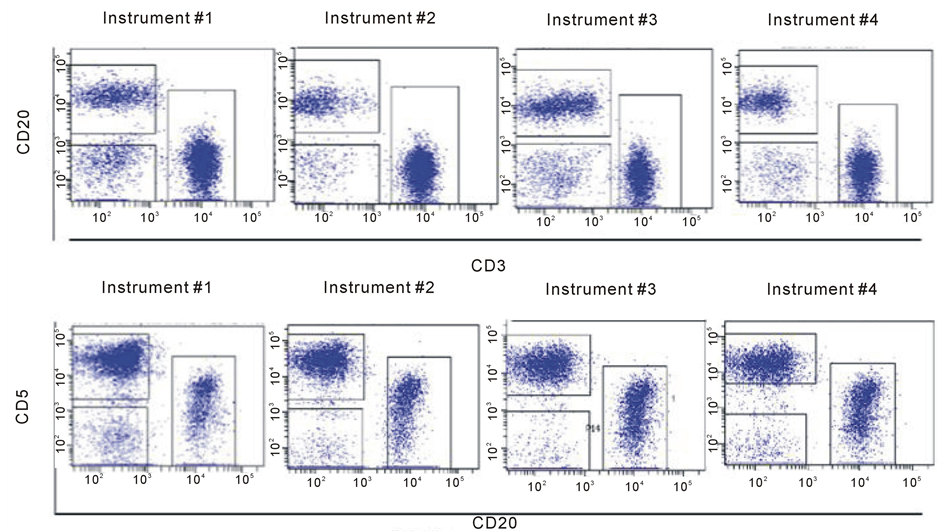
Figure 1. Comparative Dot plots of CD3 on X-axis vs. CD20 on Y-axis upper row and CD20 on X-axis vs. CD5 on Y-axis lower row for one normal donor. Data was obtained from the same sample acquired on four different flow cytometersin two different locations. The instruments were set using one set of target values as described in “Method”. Percentages of positive cells were determined after gating on CD45 positive cells following the doublet exclusion.
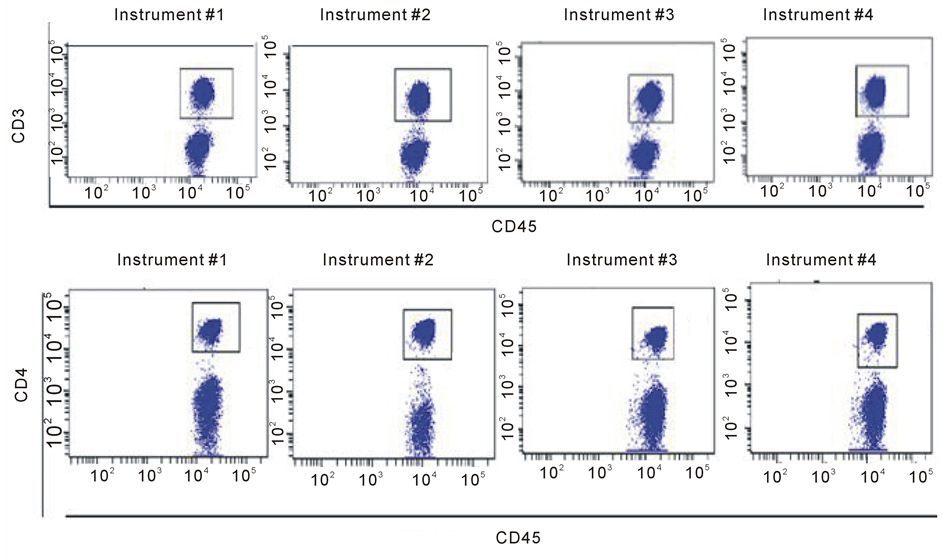
Figure 2. Comparative Dot plots of CD45 on X-axis vs. CD3 on Y-axis upper row and CD45 on X-axis vs. CD4 on Y-axis lower row for one normal donor. Data was obtained from the same sample acquired on four different flow cytometersin two different locations. The instruments were set using one set of target values as described. Percentages of positive cells were determined after gating on CD45 positive cells following doublet exclusion.

Figure 3. Schematic of CD19 quantification on APC-Cy7 channel. The linearity of APC-Cy7 channel is assured by using Ultra Rainbow beads for the calibration in the unit of the equivalent APC fluorescence (EAF) [10] (square symbol). The adjusted R-squared for the linear fit to the log transformed data as shown is 0.994. Using CD4 APC-Cy7 as a reference biomarker with a known ABC value of 45,000 for fixed whole blood, the CD19 expression level in ABC unit is determined.

Table 3. Percentage of CD3, CD4, CD8, CD19, and CD20 cells gated on CD45 positive cells obtained from individual instruments, and mean percentage and associated CV of each cell subset averaged from all four instruments.
CD45 vs. CD 19 or CD45 vs. CD 4 was used to gate on either CD19+ cells or CD4+ cells, respectively, using either APC-Cy7 or PerCP-Cy5.5 fluorochrome.
The median fluorescence intensity (MFI) values for CD19 receptor protein in both APC-Cy7 and PerCPCy5.5 channels on two fixed normal whole blood samples are provided in Table4 Although these values were obtained on the same day with assured linear responses from both fluorescence channels of the standardized instruments, they show large variations with at least 20% of CV for the same sample among different instruments. Upon the application of the ABC calibration with CD4 as the reference biomarker, an ABC value for CD19 APC-Cy7 for the sample, ND-1 and instrument 2 is calculated to be
 with the assumption that the effective fluorescence per antibody for anti-CD4 and anti-CD19 made by the same manufacturer is the same [16] . The ABC values obtained for CD19 as summarized in Table 5 are highly comparable for the same donor sample, independent of fluorochrome label (APC-Cy7 or PerCP-Cy5.5) and cytometer plat form used. The mean ABC value and associated CV summarized over three instruments and two fluorophores are 22,153 and 2.8% for ND-1 and 25,519 and 4.6% for ND-2, respectively. The consistent results from two different fluorophore labels verify the validity of the assumption that different antibodies against different antigens with the same fluorophore label likely have very similar average fluorescence per antibody values as long as they are produced by the same vendor under rigorous good manufacturing practice (GMP). The study results demonstrate comparable and consistent biomarker quantification across different fluorescence channels
with the assumption that the effective fluorescence per antibody for anti-CD4 and anti-CD19 made by the same manufacturer is the same [16] . The ABC values obtained for CD19 as summarized in Table 5 are highly comparable for the same donor sample, independent of fluorochrome label (APC-Cy7 or PerCP-Cy5.5) and cytometer plat form used. The mean ABC value and associated CV summarized over three instruments and two fluorophores are 22,153 and 2.8% for ND-1 and 25,519 and 4.6% for ND-2, respectively. The consistent results from two different fluorophore labels verify the validity of the assumption that different antibodies against different antigens with the same fluorophore label likely have very similar average fluorescence per antibody values as long as they are produced by the same vendor under rigorous good manufacturing practice (GMP). The study results demonstrate comparable and consistent biomarker quantification across different fluorescence channels

Table 4 . Median fluorescence intensity (MFI) values for CD19 in APC-Cy7 and PerCP-Cy5.5 channels obtained from two normal donors (ND). The mean MFI value and CV (column 5 and 6) are obtained for each donor from three different instruments.

Table 5. The antibody bound per cell (ABC) values obtained for CD19 on two fixed normal whole blood samples using two different fluorophore labels, APC-Cy7 and PerCP-Cy5.5 and three different cytometer platforms. The mean ABC value and CV (column 5 and 6) are obtained for each donor from three different instrument platforms. The quantification of CD19 is carried out relative to CD4 with a known expression level of 45,000 for fixed whole blood.
and cytometer platforms can be only achieved with the use of the ABC calibration scheme.
4. Summary
Quantitative flow cytometer assays are routinely used in clinical laboratories to estimate the number of specific receptor/antigen sites on the surface or inside of cells taken from blood, lymph nodes, and bone marrow. It is also appropriate for other body fluids such as a bronchial lavage, pleural and peritoneal fluids and even bladder washings. Specific receptors/antigens can serve as biomarkers for pathological conditions at various stages of a disease. Additionally, flow cytometry measurements are used to support many clinical, pharmacologic, and calibration and measurement capability claims used for drug/vaccine, device, and biologics product development and approval. However, it is still a great challenge to obtain comparable results from different laboratories using different instrument platforms due to the lack of the optimized and standardized procedures and biological reference materials. This study was designed in a small scale to develop a standardized procedure for quantifying CD19 expression level relative to CD4 reference marker. The comparable results of CD19 expression from two normal individuals on three different instruments and two different fluorophore labels support the biomarker quantification approach that relies on instrument standardization and the use of CD4 reference biomarker. To expand the scope of the investigation, flow cytometers from different manufacturers are included in an on-going study to quantify several disease markers of CLL. In addition, efforts on the production of lyophilized peripheral blood mononuclear cells (PBMC) pre-stained with fluorescently labeled anti-CD4 antibodies would simplify the developed quantification scheme without the use of fresh human whole blood.
Acknowledgements
Certain commercial equipment, instruments, and materials are identified in this paper to specify adequately the experimental procedure. In no case does such identification imply recommendation or endorsement by the National Institute of Standards and Technology, nor does it imply that the materials or equipment are necessarily the best available for the purpose.