1. Introduction
Many types of injury result in damage to the skin. The body is typically capable of closing these wounds spontaneously to restore the original functions of its protective covering as quickly as possible. This process involves various repair mechanisms in the individual layers of skin as well as the growth and differentiation of numerous cells. Understanding these processes provides valuable information for designing wound dressings and cultured skin substitutes. We have developed various wound dressings and cultured skin substitutes. The wound dressing is composed of hyaluronic acid (HA) and collagen (Col) spongy sheet containing epidermal growth factor (EGF) [1] [2] . The cultured dermal substitute (CDS) is composed of HA and Col spongy sheet containing human fibroblasts [3] [4] . Fibroblasts in CDS are capable of releasing various types of cytokines and extracellular matrix, which are necessary for wound healing. Clinical results using autologous and allogeneic CDS have been reported [5] -[10] .
In our clinical study, the allogeneic CDS is covered with conventional ointment-coated gauze and secured with a tie-over dressing. To improve the conventional covering procedure, we reported the combined use of an allogeneic CDS and EGF-incorporating sponge-type wound dressing [11] . A sponge-type wound dressing is advantageous as a top dressing for CDS in the treatment of full-thickness burn and intractable skin ulcer having a high risk of infection, because it is able to absorb a large amount of exudate and, if necessary, drain excess amount of exudate from the wound. On the other hand, a membrane-type wound dressing is advantageous as a top dressing for CDS in the treatment of skin defects caused by removal of burn scar contracture and pigmented nevus having a low risk of infection, because of no excess amount of exudate from the wound. The present study was designed to investigate fibroblast cytokine production when CDS was elevated at the interface between air and culture medium (referred to as a wound surface model), on the top of which EGF-containing membrane was placed. We aimed to determine the appropriate concentration of EGF in a membrane.
2. Materials and Methods
2.1. Materials
HA powder (Bio Sodium Hyaluronate HA 20; molecular weight, 2000 kDa; Shiseido, Tokyo, Japan), Col powder (NMP Collagen PS; Nippon Meat Packers, Osaka, Japan), and freeze-dried EGF (rh-HGF; Shanghai Haohai Biological Technology Co., Ltd., Shanghai, China) were used to prepare the matrices for CDS and EGFincorporating membrane. This recombinant human (rh-) EGF has been commercially available as a medical drug for the treatment of intractable skin ulcer. Ethylene glycol diglycidyl ether (EX810; Nagasekasei Co. Ltd., Osaka, Japan) was used as a cross-linking agent. Dulbecco’s modified Eagle’s medium (DMEM; Sigma, St.Louis, USA) and fetal bovine serum (FBS; Gibco, California, USA) were used for fibroblast culture.
2.2. Preparation of Membrane with or without EGF
HA powder (4 g) was dissolved in distilled water (400 mL) in order to prepare a high molecular weight (HMW)- HA solution. Another amount of HA powder (4 g) was dissolved in distilled water (400 mL), and was then placed in an autoclave at 120˚C for 1 h to obtain a partially hydrolyzed low molecular weight (LMW)-HA solution (molecular weight, 150 kDa). Col powder (4 g) was dissolved in distilled water (400 mL), and was then warmed at 50˚C for 10 min to yield heat-denatured Col solution. These three solutions were mixed and adjusted to pH 7.0 using 1 N-NaOH, and then divided into 4 portions (each 300 mL), in which a given amount of EGF dissolved in distilled water (15 mL) or only distilled water (15 ml) was added. Each mixture (21 mL) was poured into a plastic tray (5 cm × 8 cm), and was then placed in a clean bench for 5 days in order to prepare a membrane containing EGF. The EGF concentration in each membrane is 0, 0.1, 0.2, or 0.5 µg/cm2. The EGF concentration was expressed for convenience as a unit of µg/cm2, because the thickness of membrane was very thin (10 μm).
2.3. Preparation of HA Spongy Matrix for CDS
HA powder (30 g) was dissolved in distilled water (2000 mL). This HA solution was adjusted to pH 3.5 with 10% diluted HCl. EX810 (6 g) was mixed with distilled water (14 mL). The aqueous solution of EX810 was then added drop-wise to the HA solution under vigorous stirring. The weight ratio of HA to EX810 was adjusted to 5:1. This mixed solution (40 mL) was poured into a tray (5 cm × 8 cm) and kept at 50˚C for 5 h in order to promote cross-linking. During this procedure, the volume of HA solution was reduced by about 50%. The tray was then kept at 4˚C for 2 h, and was quickly frozen in a freezer at −85˚C, followed by freeze-drying to obtain the HA spongy sheet. The spongy sheet was rinsed with distilled water in order to remove free cross-linking agent. After rinsing, the hydrated HA spongy sheet was again placed in a tray and kept at 4˚C for 2 h, followed by quick freezing at −85˚C and freeze-drying to yield purified HA spongy sheets. These HA spongy sheets were punched to produce numerous holes (diameter 0.5 mm) separated by a distance of 4 mm. Each spongy sheet was packed into a bag and kept in a dry sterilizer at 110˚C for 1 h.
2.4. Preparation of CDS
Col powder was sterilized in a vacuum drying oven at 121˚C for 2 h and dissolved in sterilized distilled water at a concentration of 1%. The DMEM used for mixing with Col solution was prepared at twice the standard concentration. Fibroblasts were suspended in double concentrated DMEM supplemented with 20% FBS. Cell suspension (10 mL) and Col solution (10 mL) were mixed at less than 10˚C, and were then poured into a dish measuring 5 cm × 8 cm, in which a sterilized HA spongy sheet of the same size was immersed, followed by overnight incubation under 5% CO2 at 37˚C to allow jellification. The resulting product was cultured for 5 days in a dish containing additional 30 ml of culture medium (DMEM supplemented with 10% FBS) in order to give cultured dermal substitute (CDS). Cell seeding density was 2 × 105 cells/cm2 (Group I) and 4 × 105 cells/cm2 (Group II).
2.5. Cultivation on the Wound Surface Model
Cultivation on the wound surface model was conducted according to the method described in our previous article [11] . The potential of the CDS to release angiogenic cytokine such as vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF) was evaluated under the conditions acceptable in the clinical use. To make a wound surface model, the CDS was cut to a size of 5 cm ´ 4 cm and was elevated at the interface between air and culture medium in the 100 cm2 dish containing 60 ml of culture medium. A membrane with or without EGF (5 cm ´ 4 cm) was placed on top of the CDS, and then the CDS was cultured for 7 days (Figure 1).
2.6. MTT Assay of Fibroblast in the CDS
The procedure was described previously [11] . Briefly, cell metabolic activity was measured using 3-(4.5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay (Research Organics, Ohaio, USA). CDS after 7 days of cultivation on a wound surface model was cut into a size of 3 cm ´ 3 cm and transferred into another dish, into which 6 mL of culture medium containing 0.6 mL of MTT had been added, followed by incubation at 37˚C for 3.5 h. This sample was immersed in 5 mL of dimethylsulfoxide in order to obtain a blue-violet solution. Absorbance of this solution was measured using a microplate reader (Microplate Reader Model 550; Bio-Rad

Figure 1. Wound surface model: a CDS (5 cm ´ 4 cm) was elevated at the interface between air and culture medium in the 100 cm2 dish containing 60 ml of culture medium. A membrane with or without EGF (5 cm ´ 4 cm) was placed on top of the CDS, and then the CDS was cultured for 7 days. The amount of cytokine (VEGF and HGF) released from CDS was measured by ELISA.
Laboratories Inc., California, USA) at 570 nm.
2.7. Quantitative Analysis of VEGF and HGF Released from CDS
The procedure was described previously [11] . Briefly, the amount of cytokines (VEGF and HGF) released from CDS was measured by ELISA using a Quantikine® VEGF ELISA kit and a Quantiline® HGF ELISA kit (R&D Systems, Inc., Minneapolis, USA). Culture medium after 7 days of cultivation on a wound surface model was collected and stored at −30˚C. Samples of culture media and standards for each cytokine were added in duplicate to the wells of a 96-well microplate, and ELISA was performed according to the manufacturer’s instructions.
2.8. Statistical Analysis
Data are expressed as mean ± standard error. Statistical analysis was performed using Tukey Kramer test.
3. Results
3.1. Preparation of Membrane with or without EGF
This membrane was composed of HMW-HA, LMW-HA, and heat-denatured Col. The heat-denatured Col aqueous solution was not precipitated even at a neutral pH condition. Therefore, it is possible to prepare a clear mixed solution at neutral pH condition. The resulting clear mixture had an appropriate viscosity, and thereby could prepare a membrane with thickness as thin as 10 μm.
3.2. Preparation of HA Spongy Matrix for CDS
Scanning electron microphotograph of a cross-section of the cross-linked HMW-HA spongy matrix is shown in Figure 2. The spongy matrix showed a unique cross-sectional view displaying vertical holes and a porous structure.
3.3. Preparation of CDS
In general, the size of collagen gel sheet including fibroblasts decreases easily, because fibroblasts contract the collagen microfibers in gel. To improve the size contraction, a unique structure was designed. The CDS is composed of a fibroblasts-incorporating collagen gel combined with a cross-linked HMW-HA spongy matrix. This HA spongy sheet was punched to produce numerous holes (diameter 0.5 mm), thereby a fibroblasts-incorporating collagen gel was found within a HA spongy structure (Figure 2). No contraction of CDS was observed, even at the high cell seeding density in Group II (4 × 105 cells/cm2). CDS had good handling property.
3.4. Cell Metabolic Activity in CDS in Wound Surface Model
The optical density (OD) value on MTT assay corresponds to the metabolic activity of fibroblasts in CDS under each culture condition. The data after 7 days of cultivation at the interface between air and culture medium are shown in Figure 3. The OD values in Group II were higher than those in Group I. This finding corresponds to the cell number in CDS. In Group I (2 × 105 cells/cm2), cell metabolic activity increased when CDS was laminated with a membrane containing EGF at 0.1 µg/cm2, but decreased when CDS was laminated with membranes containing EGF at 0.2 and 0.5 µg/cm2. In Group II (4 × 105 cells/cm2), cell metabolic activity was not affected when CDS was laminate with membranes containing EGF at 0.1 µg/cm2, but decreased slightly when CDS was laminated with a membrane containing EGF at 0.2 and 0.5 µg/cm2. EGF affected cell metabolic activity in a dose-dependent manner. This dose-dependence appeared more slightly in Group II compared with Group I, because of double cell number in Group II.
3.5. VEGF Production by Fibroblasts in CDS
Figure 4 shows the VEGF production by fibroblasts in CDS after 7 days of cultivation in Group I (2 × 105 cells/cm2) and Group II (4 × 105 cells/cm2). The amount of VEGF in Group II was double of those in Group I, corresponding to the double cell number. The amount of VEGF in Group I increased sufficiently when CDS was laminated with membranes containing EGF at 0.1 and 0.2 µg/cm2, in spite of the dose-dependence in OD values,

Figure 2. CDS structure: (a) Scanning electron microphotograph of spongy matrix, (b) Light microphotograph of hematoxylin-eosin stained specimen, and (c) Macroscopic appearance of CDS. A scale bar is 1 mm. Arrowheads show a fibroblasts-incorporating collagen gel within HA spongy frame.

Figure 3. Optical density at 570 nm on MTT assay of fibroblasts in CDS laminated with each membrane after cultivation for 7 days on wound surface model. *P < 0.01 vs. 0.5 μg/cm2.
but slightly decreased at EGF concentration of 0.5 µg/cm2. A membranes containing EGF at 0.2 µg/cm2 stimulated fibroblasts in CDS to release 2.3 times more VEGF after 7 days of cultivation, as compared with a control. The amount of VEGF in Group II increased sufficiently when CDS was laminated with membranes containing EGF ranging from 0.1 to 0.5 µg/cm2. A membrane containing EGF at 0.5 µg/cm2 stimulated fibroblasts in CDS to release 2.7 times more VEGF after 7 days of cultivation, as compared with a control.
3.6. HGF Production by Fibroblasts in CDS
Figure 5 shows the HGF production by fibroblasts in the CDS after 7 days of cultivation in Group I (2 × 105

Figure 4. VEGF production by CDS laminated with each membrane after cultivation for 7 days on wound surface model. Group I: *P < 0.01 vs. control, †P < 0.01 vs. 0.1 μg/cm2, Group II: *P < 0.01 vs. control, †P < 0.01 vs. 0.1 μg/cm2.
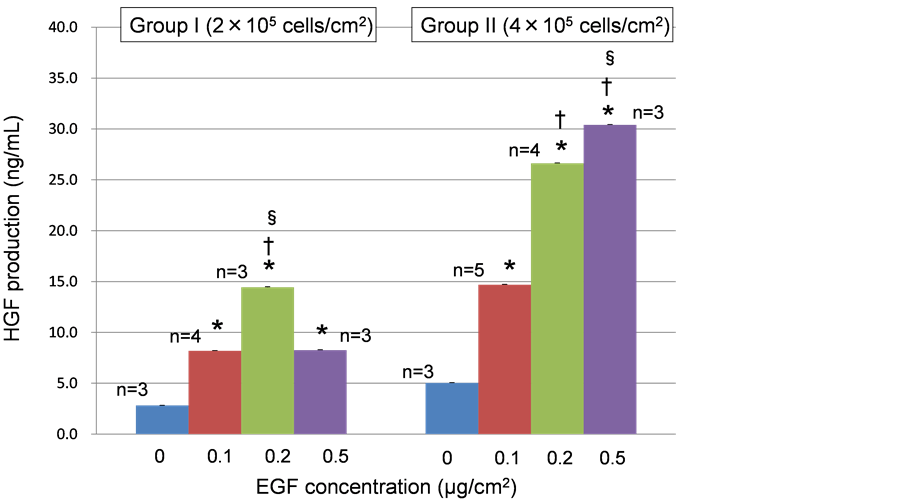
Figure 5. HGF production by CDS laminated with each membrane after cultivation for 7 days on wound surface model. Group I: *P < 0.01 vs. control, †P < 0.01 vs. 0.1 μg/cm2, §P < 0.01 vs. 0.5 μg/cm2. Group II: *P < 0.01 vs. control, †P < 0.01 vs. 0.1 μg/cm2, §P < 0.01 vs. 0.2 μg/cm2.
cells/cm2) and Group II (4 × 105 cells/cm2). The amount of HGF in Group II was double of those in Group I, corresponding to the double cell number. The amount of HGF in Group I increased sufficiently when CDS was laminated with membranes containing EGF ranging from 0.1 to 0.2 µg/cm2, in spite of the dose-dependence in OD values, but decreased at EGF concentration of 0.5 µg/cm2. A membranes containing EGF at 0.2 µg/cm2 stimulated fibroblasts in CDS to release 5.8 times more HGF after 7 days of cultivation, as compared with a control. The amount of HGF in Group II increased sufficiently when CDS was laminated with membranes containing EGF ranging from 0.1 to 0.5 µg/cm2. A membrane containing EGF at 0.5 µg/cm2 stimulated fibroblasts in CDS to release 6.0 times more HGF after 7 days of cultivation, as compared with a control.
4. Discussion
HA is a particularly promising biomaterial [12] [13] , as it stimulates cell migration [14] [15] and angiogenesis [16] -[18] . Low molecular weight HA promotes angiogenesis [19] , while HA fragments, particularly oligomers, are capable of stimulating endothelial cell proliferation [20] . Col and Col-derived peptides act as chemoattractants for fibroblasts in vitro and may have similar activity in vivo [21] . Moreover, Col-derived dipeptides and proline-hydroxyproline stimulate cell proliferation and HA synthesis in cultured human dermal fibroblasts [22] .
CDS was designed to combine the wound healing effect via growth factors released from fibroblasts and the wound healing effect via biomaterials, i.e., HA and Col used as matrix. This type of CDS was employed as an autologous cultured gingival dermal substitute in clinical study. The excellent clinical result was reported in our previous article [23] . In addition, EGF-incorporating membrane was designed to enhance the cytokine production by fibroblast in CDS and to promote wound healing via combined effects with EGF and biomaterials, i.e., HA and Col.
It is known that growth factors accelerate wound healing by regulating various cell functions such as proliferation, differentiation, migration and morphogenesis [24] [25] . The potential benefits of exogenous growth factors in the treatment of skin wounds have long been of interest, and have been fueled by observations that endogenous levels of growth factors are reduced in some chronic wounds [26] . EGF is known to be a potent stimulator of cell proliferation of keratinocytes, fibroblasts and endothelial cells, as well as a potent stimulator of fibroblasts cytokine production, thus facilitating the formation of granulation tissue and re-epithelialization [27] .
In the present study, we aimed to determine the appropriate concentration of EGF in a membrane by altering the EGF concentration in the membrane, and by altering the seeding density of fibroblast in the matrix. The practical issue is to find out the appropriate EGF concentration in the membrane under the practical condition in which EGF-incorporating membrane was placed on the CDS for one week.
The effects of growth factor on cell proliferation are generally dependent on its concentration. In a previous study [28] , we reported that fibroblast proliferation was facilitated in a flask containing culture medium with bFGF ranging from 2 to 10 µg/15mL, but failed to facilitate proliferation at 50 µg/15mL. On the other hand, fibroblast proliferation was facilitated in a flask containing culture medium with EGF ranging from 2 to 50 µg/15mL. In the present study, we designed a membrane to contain EGF ranging from 0.1 to 0.5 µg/cm2, in which EGF concentration was expressed for a convenience as a unit of µg/cm2. When a membrane containing EGF at 0.5 µg/cm2 (5 cm × 4 cm) was immersed directly into 60 ml of culture medium, and if all of the EGF could be dissolved into this culture medium, the maximum concentration of EGF would be 2.5 µg/15mL. The EGF can facilitate fibroblast proliferation in the flask at a concentration of 5.0 µg/15mL. However, the EGF released from the membrane showed dose-dependent effects on the metabolic activity of fibroblast in CDS. In Group I (2 × 105 cells/cm2), fibroblasts in CDS showed lower OD values when laminated with a membrane containing EGF at 0.5 µg/cm2, as compared with control. This suggests that EGF-incorporating membranes provide fibroblasts with a microenvironment of excess EGF concentration to suppress fibroblast function. EGF dose dependency appeared to more sensitive in Group I than in Group II, as the sell seeding density in Group I was half that in Group II. In the experiment using ELISA, fibroblasts in CDS in Group I released increased amounts of VEGF and HGF when laminated with membranes containing EGF at 0.1 and 0.2 µg/cm2, but failed to increase at 0.5 µg/cm2. On the other hand, fibroblasts in CDS in Group II released increased amounts of VEGF and HGF when laminated with a membrane containing EGF ranging from 0.1 to 0.5 µg/cm2. EGF dose dependency appeared to more sensitive in Group I than in Group II, as the sell seeding density in Group I was half that in Group II. Thus, the appropriate concentration of EGF in the membrane was found to be between 0.1 and 0.2 µg/cm2.
5. Conclusion
The EGF-incorporating membrane is capable of enhancing angiogenic cytokine production by fibroblasts in CDS, as well as serving as a top covering to protect CDS. In practice, the appropriate concentration of EGF in the membrane was found to be between 0.1 and 0.2 µg/cm2. EGF released from the membrane is able to stimulate fibroblasts in CDS to release an increased amount of VEGF and HGF that are essential for angiogenesis.
NOTES
*Corresponding author.