 Journal of Biomaterials and Nanobiotechnology, 2011, 2, 461-471 doi:10.4236/jbnb.2011.24056 Published Online October 2011 (http://www.SciRP.org/journal/jbnb) Copyright © 2011 SciRes. JBNB 461 A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles Yuri Mikhailovich Yevdokimov1*, Sergey Gennadevich Skuridin1, Victor Ivanovich Salyanov1, Vladimir Ivanovich Popenko1, Victor Moiseevich Rudoy2, Olga Vadimovna Dement’eva2, Eleonora Vladimirovna Shtykova3 1Engelhardt Institute of Molecular Biology of the Russian Academy of Sciences, Moscow, Russia; 2Frumkin Institute of Physical Chemistry and Electrochemistry of the Russian Academy of Sciences, Moscow, Russia; 3Shubnikov Institute of Crystallography of the Russian Academy of Sciences, Moscow, Russia. Email: *yevdokim@eimb.ru Received June 21st, 2011; revised July 26th, 2011; accepted August 15th, 2011. ABSTRACT Au-nanoparticles (size about 2 nm, but not 5 or 15 nm) are capable of effectively incorporating into quasinematic lay- ers of particles of cholesteric liquid-crystalline dispersion formed by double-strand ed nucleic ac id molecu les of various families (DNA and poly(I)xpoly(C)). This Au-size-dependent process is accompanied by a decrease in amplitudes of abnormal bands in the CD spectra specific to in itial cholesteric liquid-crystallin e dispersions and simultaneously by an appearance of plasmon resonance band in visible absorption spectrum. The study of properties of particles of choles- teric liquid-crystalline disp ersion treated with Au-nan oparticles by means of va rious physico -chemica l methods demon - strates that incorporation of Au-nanop articles into quasinematic layers of these particles results in two effects: i) it fa- cilitates reorganiza tion of the spatial ch olesteric structure o f particles, and ii) it induces the forma tion of Au-clusters in the content of particles. It is not excluded that these effects represent a possible reason for genotoxicity of Au-nanopar- ticles. Keywords: Double -Str anded Nucleic Acids, Gold Nanoparticles, Nucleic Acid Liquid-Crystalline Dispersio ns, Circular Dichroism, Structure of Lyot r opi c Li q ui d Cr yst als of (Bio)Polymers, Genotoxity of Nanoparticles 1. Introduction Understanding how nanomaterials affect live-cells func- tions is the principal aim of nanobiotechnology [1]. Al- though applications of various nanomaterials (nanoparti- cles or quantum dots) have shown promising potential for medicine, the fundamental interactions and effects of nanomaterials in living systems for the most part remain unknown. This depends mainly on the solution of two problems: 1) there are many nanomaterials with strongly different properties; 2) an adequate, “coordinated” sys- tem to test nanomaterials is not elaborated. For instance, it was suggested that biological effect of Au-nanopar- ticles on spatially organized double-stranded (ds) DNA structures in vitro and in vivo is similar to the effect of mutagenic molecules [2,3]. One can stress that the inter- action of metal nanoparticles with native single- or dou- ble-stranded nucleic acid molecules is a well-known phenomenon [4]. Although, the cytotoxicity of Au-nano- particles in vitro and in vivo has been examined by se- veral research groups, no general conclusion can be drawn at present [5-8]. It is connected (maybe) to the fact that the functioning of the nanomaterials in living cells was tested in quite different biological test-systems and the estimation of the mechanism (or mechanisms) of this process is a complicated problem [9,10]. This means that great importance is attached to creation of simple model systems to test the effects of nanomaterials under rea- sonable conditions. Despite the fact that in vivo condi- tions are different from in vitro, one of these model sys- tem is the so-called “cholesteric liquid crystals” and “cholesteric liquid-crystalline dispersions” of ds DNA molecules [11]. Indeed, the physico-chemical properties of ds DNA cholesteric liquid-crystalline dispersions (CLCDs) reflect some properties of these macromole- cules in biological objects such as chromosomes of pri- mitive organisms (for instance, chromosomes of the Di- noflagellate, etc.) and DNA containing viruses [12]. Hence, this test-system is not only of nanotechnological,  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles 462 but of biotechnological interest. Here one can add, that due to the high local concentration of the ds DNA mole- cules and their ordered arrangement in particles of CLCD, the low-molecular-mass chemical and biologi- cally active compounds quickly diffuse into the content of particles of CLCD and interact with ds DNA mole- cules forming different complexes [11]. This process in- duces an appearance peculiarities (specific for these compounds) in the circular (CD) spectra [13]. This means that every particles of the CLCD was considered not only as an effective “adsorber” but as a sensing unit [14] for the low-molecular-mass compounds as well. However, to the best of our knowledge, the interaction of nanoparticles added to solutions containing particles of CLCDs (PCLCD) formed by ds DNA molecules has not been investigated so far. The goal of this work is the study of the effects in- duced by Au-nanoparticles in the DNA PCLCDs using various physico-chemical methods. 2. Materials and Methods Three colloidal gold solutions (hydrosols) with spherical nanoparticles of different sizes were used in our study. The spherical Au-nanoparticles were synthesized ac- cording to previously reported methods [13-15]. The first hydrosol was prepared by Turkevich et al. [15] method and contained particles with average diameter of about 15 nm. The second one, with Au-nanoparticles of 5.5 nm in size, was synthesized according to Natan et al. [16]. Finally, hydrosol containing quasi-metallic Au-nanopar- ticles with sizes of 2 - 3 nm was synthesized as described in [17]. The average size of Au nanoparticles in stock preparations was estimated by means of dynamic light scattering and by electron microscopy. The particle num- ber concentration was equal to 1012; 1013; and 1015 cm−3, respectively. Stock Au-nanoparticle solutions were stored in dark in refrigerator at 4˚C and used 2.5 month after their synthesis. Calf thymus ds DNA preparation (Sigma, USA) was used after additional purification and depolymerization (molecular mass of 0.3 - 0.7) × 106 Da. Ds poly(I)xpoly(C) preparation (Sigma, USA, lot 023K4032) was used without additional purification. Poly (ethylene glycol) (PEG; Serva, Germany; mole- cular mass 4,000 Da) and cyanine dye SYBR Green I (SG; Sigma, USA) were used without extra purification. SG concentration in solutions was measured spectropho- tometrically [18]. SG stock solution was stored at 4˚C in dark. Water-salt solutions of DNA, PEG and NaCl were made in phosphate buffer (pH ~7.0) and then filtered through membrane filters (Millipore, USA) with pore dia- meter 0.8 μm to remove possible mechanical impurities. Schemes of control tests (1 and 2) and performed ex- periments are shown below: The method of preparation of control cholesteric liquid- crystalline dispersions (CLCDs) of nucleic acids (NA) (control 1) in PEG-containing water-salt solutions is de- scribed in [11]. This method was used for preparation of dispersions and phases of ds NA CLCD treated with Au- nanoparticles (see “Experiments” above). Absorbance spectra were taken by spectrophotometer Cary 100 Scan (Varian, USA); and the CD spectra, using portable dichrometer CDS-2 (produced by Institute of spectroscopy of the RAS, Troizk, Moscow region) [19]. The fluorescence “images” of particles of the ds DNA CLCD treated with Au nanoparticles were obtained by the confocal microscope “Leica TCS SP5”. The morphology of the ds DNA CLCD particles formed in PEG-containing solutions and treated with Au- nanoparticles was examined by AFM. These solutions were filtered through a poly(ethylenetherephtalate) (PETP) nuclear membrane filter with size of pores of 150 nm C opyright © 2011 SciRes. JBNB  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles463 (produced by the Institute of Crystallography of the RAS). This procedure allows one to immobilize particles on the surface of the filter. Then the filters, containing obtained samples, were dried in air for no less than 1 h. The samples were measured on a scanning probe micro- scope SmartSPM (produced by AIST-NT Co, Zeleno- grad, Moscow region, Russia) in semicontact mode. The fpN10 silicon probe with resonance frequency around 250 kHz was used. The initial amplitude of the probe was fixed at 100 nm. The scanning frequency was varied from 2 to 10 Hz; the number of pixels was 256 × 256 and 512 × 512. Pellets (~3 mg) obtained after low speed centrifuga- tion of the DNA CLCD treated with Au nanoparticles, were investigated by SAXS. SAXS measurements were performed on the diffractometer AMUR-K (developed at the Institute of Crystallography of the RAS, Moscow) [20]. Technical details of SAXS experiments are de- scribed in [20,21]. The analysis of characteristic Bragg peaks on the small-angle scattering curves was made by the program PEAK [22]. 3. Results and Discussion The CD spectra of ds DNA and ds poly(I)xpoly(C) CLCDs treated with Au-nanoparticles (diameter ~ 2 nm) are compared in Figure 1. First of all, one can see that the formation of the liquid- crystalline ds NA dispersions (compare curve 1 and 1I) induced by the high concentration of PEG in water-salt solution is accompanied by an appearance of intense (abnormal) bands in the CD spectra located in the region of absorption of NA nitrogen bases( λ ~270 nm). Ac- cording to the theory [11,13], the appearance of these abnormal (negative or positive) bands unequivocally tes- tify the macroscopic, cholesteric, twist of neighboring NA molecules in particles of dispersion formed as a re- sult of their phase exclusion from PEG-containing water- salt solutions. The negative sign of the band in the CD spectrum proves the left-handed cholesteric twist of the right-handed DNA molecules (B-form, curve 1), whereas the positive sign corresponds the right-handed choles- teric twist of the right-handed poly(I)xpoly(C) molecules (A-form, curve 1I) in the formed particles. Hence, the abnormal bands in the CD spectrum (Figure 1) located in the absorption region of the nitrogen bases of the NA molecules is the direct evidence for the formation of the particles characterized by helically twisted spatial struc- ture [18-20], or a so-called cholesteric structure of parti- cles and the term CLCD (cholesteric liquid-crystalline dispersion) was used to signify these particles. In the physicochemical sense, the systems under in- vestigation are PCLCD of the ds NA molecules that are distributed isotropically in the water-salt solution of PEG. Figure 1. The CD spectra of ds DNA CLCD (curves 1-4) and ds poly(I)xpoly(C) CLCD (curves 1I-5I) treated with Au-nanoparticles (2 nm). 1: Cnano-Au = 0; 2: Cnano-Au = 0.07 × 1014 particle/ml; 3: Cnano-Au = 0.26 × 1014 particle/ml; 4: Cnano-Au = 0.82 x 1014 particle/ml; CDNA = 9 g/ml; CPEG = 150 mg/ml; 0.27 M NaCl + 1.78 × 10-3 M Na-phosphate buffer. 1I : Cnano-Au = 0; 2I : Cnano-Au = 0.16 × 1014 particle/ml; 3I : Cnano-Au = 0.33 × 1014 particle/ml; 4I : Cnano-Au = 0.66 × 1014 particle/ml; 5I : Cnano-Au = 0.82 × 1014 particle/ml, Cpoly(I)xpoly(C) = 9 g/ml, CPEG = 170 mg/ml; 0.27 M NaCl + 1.78 × 10-3 M Na-phosphate buffer. A = (AL – AR) × 10–6 optical units; L = 1 cm; T = 22˚C. In all cases, the CD spec - tra were taken after 3 h of treatment. These particles do not exist in the absence of high os- motic pressure (π, dyne/cm2) of the solvent (for instance, in the water-salt solution, where lgπ value is about 6) [11]. Note that the particles of the low-molecular-mass ds NA dispersions are “microscopic droplets of concen- trated NA solutions”, and due to “liquid” mode of pack- ing these particles cannot be “taken in hand” or “directly seen”. (The estimations have shown that in the case of ds DNA molecules every PCLCD has a mean diameter about 500 nm; it contains about 104 ds DNA molecules fixed on distances within 2.5 - 5.0 nm (depending on osmotic pressure of the solvent [11,12]). Figure 1 shows that in both cases the amplitudes of abnormal bands in the CD spectra drop. The higher the concentration of Au-nanoparticles in solution, the greater is decrease in abnormal band in CD spectra of CLCDs of both types of nucleic acids. Copyright © 2011 SciRes. JBNB  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles 464 Figure 1 needs a few comments. First of all, the am- plitude of the abnormal band in the CD spectrum de- pends mainly on helical twisting of quasinematic layers of NA in cholesteric structure of PCLCD [11,13] Secondly, the theory [23] predicts that the change in the efficiency of interaction between ds NA molecules in quasinematic layers must be accompanied by changes in the parameters of the spatial helical structure of CLCD particles and in the value of amplitude of an abnormal band in the CD spectra. This means that the decrease in the amplitudes of abnormal bands in the CD spectra (curves 2 - 4 and 2I - 5I) is connected with the change in the extent of helical twisting of quasinematic layers in structure of PCLCD formed by any type of ds NA mole- cules, i.e., B-family for DNA and A-family for poly(I) xpoly(C). This effect reflects only one process, i.e., an incorporation of Au-nanoparticles into PCLCD formed by ds NA molecules (Figure 1). Thirdly, any possible aggregation of independent Au- nanoparticles outside of PCLCD can not induce the change in the real value of the abnormal band in the CD spectra in the region of absorption of NA molecules. Finally, the changes in amplitudes of abnormal bands in the CD spectra represent a unique criterion for incor- poration of Au-nanoparticles in quasinemtic layers for- med by ds NA molecules in PCLCD. The efficiency of decrease in the amplitudes of ab- normal bands for the CLCDs formed by ds DNA or poly (I)xpoly(C) molecules (Figure 1) depends on the size of Au-nanoparticles and the time of treatment with Au- nanoparticles. For instance, if the diameter of the Au- nanoparticles is 2 nm, abnormal band amplitude de- creases by 75% after 4 hours of treatment, but in the case of 15 nm Au-nanoparticles, it decreases only by 20% [21]. This fact allows one to suggest that the incorpora- tion of Au-nanoparticles in the content of PCLCD is de- termined by the distance between ds NA molecules packed in the quasinematic layers under used conditions (150 mg/ml of PEG) [11] (compare Control 2 and Ex- periments above). Au-nanoparticles with the size about 2 nm are capable of diffusing between ds NA molecules, because this size is close enough to the distance between ds NA molecules under used conditions (concentration of PEG, etc.) [21]. However, Au-nanoparticles with the sizes equal to 5 nm and 15 nm are too “big” in compari- son to the distance between ds NA molecules in quasi- nematic layers, hence these particles do not diffuse ef- fectively into content of ds NA PCLCD. Explaining the results shown in Figure 1 one can take into account the following. The first experiments [24,25] demonstrated that Au- nanoparticles can be assembled near DNA molecules forming supramolecular structures. Later it was shown that an assembly of the Au-nanoparticles is accompanied by formation of planar superstructure consisting of re- petitive neighboring linear DNA molecules and Au- nanopartices. These results unequivocally show that lin- ear, rigid, ds DNA molecules after interaction with Au- nanoparticles form planar superstructures of type (…- Au-DNA-Au-DNA-Au-DNA-Au-…), despite the aniso- tropic properties of initial ds DNA molecules [4, 26-29]. Hence, on one hand, Au-nanoparticles enforce ds DNA molecules to be organized into planar superstructures, in which neighboring DNA molecules are not only closely packed, but сollaterally located. On the other hand, it is well known that neighboring ds DNA molecules packed in quasinematic layers of PC- LCD are helically twisted due to the anisotropic pro- perties of these molecules [11]. Comparison of these two circumstances means that immobilization (incorporation) of Au-nanoparticles in ds NA quasinematic layers will, indeed, results in untwist- ing of neighboring ds NA layers. This explains the changes in the CD spectra in Figure 1. Indeed, the steric limitations resulting from both dense packing of ds NA molecules in quasinematic layers and fixed PEG concen- tration in solution prevent the spatial separation (denatu- ration) of the two strands of the neighboring ds NA molecules [11]. In this case both the incorporation of Au- nanoparticles into quasinematic layers of PCLCD and their ordering near ds NA surfaces must be “trans- formed” by this or that way into the changes in the en- ergy of interaction between neighboring NA molecules in quasinematic layers. Hence, Au-nanoparticles can in- duce a transition from cholesteric structure of ds NA PCLCD that has high abnormal optical activity to a cer- tain structure, having very low (if any!) abnormal optical activity. For this process the differences in the secondary structure of various ds NA molecules, are not important. This means that the changes in the CD spectra (Figure 1) clearly demonstrate that Au-nanoparticles are incor- porated in the structure of PCLCD. These changes in the CD spectra depend only on presence of Au-nanoparticles in the content of quasinematic layers of the PCLCD. The concrete peculiarities of the mechanism of interaction of Au-nanoparticles with ds NA molecules are not impor- tant for this effect. Additional study of visible absorption spectra of Au- nanoparticles opens a way to evaluate the size of the as- semblies formed by these particles under various con- ditions [30-37]. Figure 2 shows that the pronounced changes in the visible optical spectrum were observed at the treatment of the ds DNA CLCD with Au-nanoparticles. (The simi- lar results for ds poly(I)xpoly(C) are not shown). First of all, this treatment is accompanied by an appea- C opyright © 2011 SciRes. JBNB  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles Copyright © 2011 SciRes. JBNB 465 Figure 2. (a) The change in the absorption spectrum of Au-nanoparticles (2 nm) at their interaction with ds DNA CLCD. 1: 5 min; 2: 12 min; 3: 43 min; 4: 109 min; 5: 230 min; 6: 1200 min after the addition of Au-nanoparticles. CDNA = 9 g/ml; CPEG = 150 mg/ml; 0.27 M NaCl + 1.78 × 10-3 M Na-phosphate buffer; Cnano-Au = 0.82 × 1014 particle/ml; T = 22˚C; (b) The correlation between the position of SPR band and size of Au-nanoparticles. Curve 1-open squares-experimental data from Figure 2(a); curve 2: filled squares [32]; curve 3: filled circles [36]; curve 4: triangles [37]. rance of absorption at 505 nm (surface plasmon resonance (SPR) absorption band) [30,32]. The plasmon resonance is responsible for the pile-violet color of solu- tion containing ds DNA CLCD treated with Au-nano- particles. This resonance is definitely absent in the initial DNA solution and only slightly defined at ~ 505 nm in colloid solution containing Au-nanoparticles with the size about 2 nm. This means that the immobilization of Au-nanoparticles in the content of ds DNA CLCD par- ticles (Figure 2) brings Au-nanoparticles close together. This results in modifying their local environment and changing the position of the SPR band, because the SPR absorbance is sensitive to local environment [35]. Se- condly, the intensity of the SPR band is gradually in- creased and the maximum of this band is shifted from ~ 505 nm to ~ 550 nm with time. Comparison of our data (Figure 2(b), curve 1) to a- vailable numerical data [30-34,36,37], concerning the correlation between position of the maximum of the SPR band and the size of Au-nanoparticles shows that in our case the change in the SPR band reflects the increase in the size of Au-nanoassemblies from 2 to about 50 nm. Of course, this is a very simple estimation, that does not take into account such factors as real shape of Au-assem- blies, local dielectric constant or local refractive index [38]. The comparison of the SPR band typical for PEG con- taining water-salt solutions added with 5 nm Au-nano- particles to analogous solution containing PCLCD for- med by ds DNA molecules showed no changes in the SPR band amplitude. The absence of changes in the SPR band was detected in the case of PCLCD treated with 15 nm Au-nanoparticles as well (data are not shown). These results confirm once again that “big” Au-nanoparticles do not incorporate into the structure of quasinematic la- yers of PCLCD. Here one can add that due to the spherical symmetry of individual Au-nanoparticles used by us the formation of assemblies from these particles is a significant prob- lem. But, Figure 2 shows that the ds DNA molecules, ordered in the quasinematic layers, provide, as templates, the necessary symmetry-breaking mechanism to form extended linear structure from Au-nanoparticle fixed between neighboring ds DNA molecules. Finally, after 20 hours one can detect the formation of dark-violet pre- cipitate in the tested solution. The intensive stirring of the solution containing the pellet permits to restore its optical properties and to measure its optical spectrum. Curve 6 in Figure 2 demonstrates the presence of plas- mon resonance band located at ~ 545 - 550 nm. Close similarities between the shapes and position of the SPR bands (curves 6 and 5) show that the dark-violet precipi- tate consists of isolated ds DNA PCLCD, which, in its turn, contain extended structures (about 50 nm) formed by Au-nanoparticles. This result shows that the spectral changes in the visible optical spectrum are induced by the processes that take place, mainly, within isolated ds NA PCLCD treated with Au-nanoparticles, but not be- tween neighboring PCLCD. (The curves of the diminishing in the amplitudes of abnormal bands in the CD-spectra and the increase in the SPR bands for ds DNA and poly(I)xpoly(C) CLCDs treated with Au-nanoparticles showed that there are two different processes: i) quick drop in the amplitudes of the CD bands due to incorporation of Au-nanoparticles into quasinematic layers of ds NA PCLCD, and ii) relatively 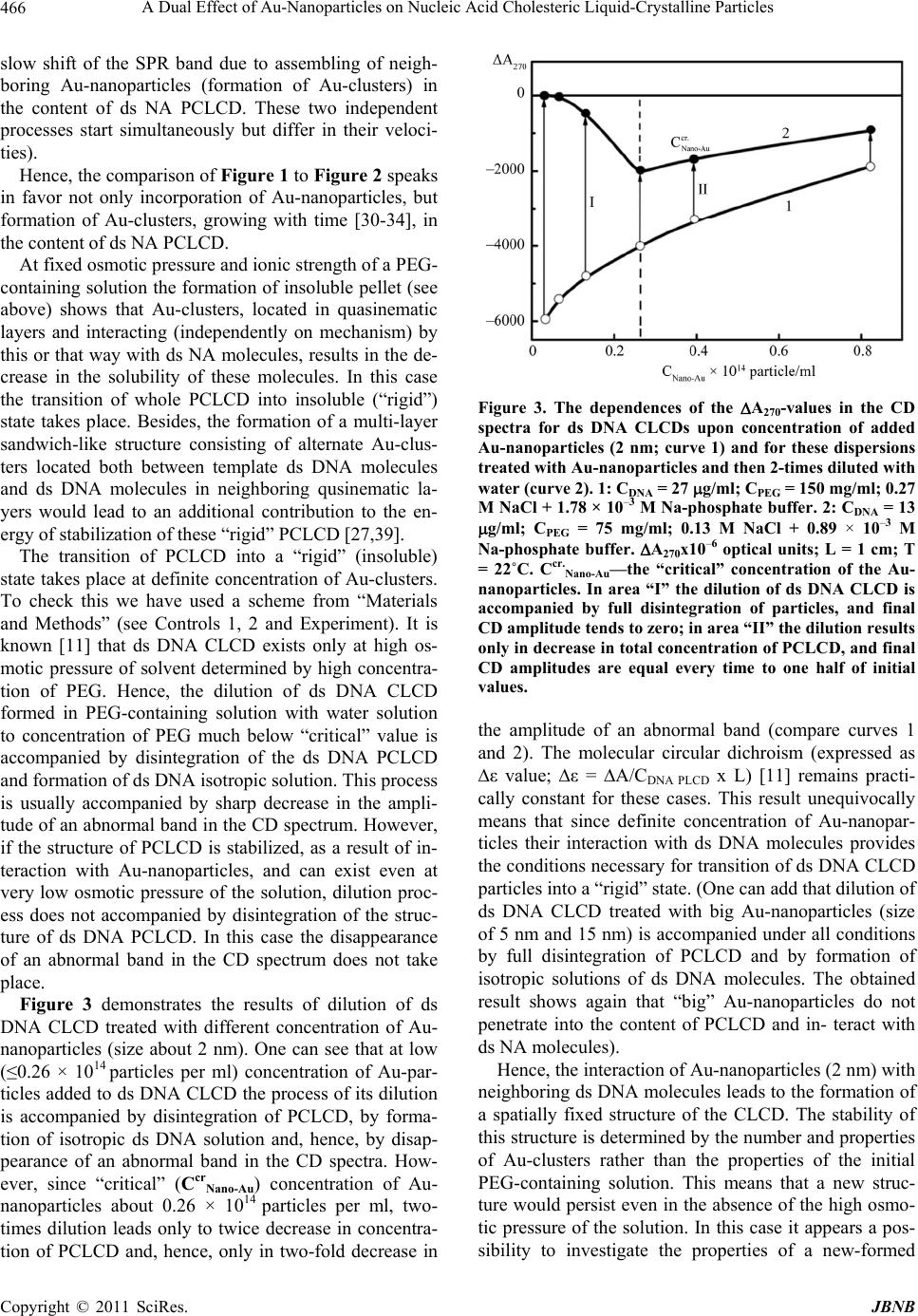 A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles 466 slow shift of the SPR band due to assembling of neigh- boring Au-nanoparticles (formation of Au-clusters) in the content of ds NA PCLCD. These two independent processes start simultaneously but differ in their veloci- ties). Hence, the comparison of Figure 1 to Figure 2 speaks in favor not only incorporation of Au-nanoparticles, but formation of Au-clusters, growing with time [30-34], in the content of ds NA PCLCD. At fixed osmotic pressure and ionic strength of a PEG- containing solution the formation of insoluble pellet (see above) shows that Au-clusters, located in quasinematic layers and interacting (independently on mechanism) by this or that way with ds NA molecules, results in the de- crease in the solubility of these molecules. In this case the transition of whole PCLCD into insoluble (“rigid”) state takes place. Besides, the formation of a multi-layer sandwich-like structure consisting of alternate Au-clus- ters located both between template ds DNA molecules and ds DNA molecules in neighboring qusinematic la- yers would lead to an additional contribution to the en- ergy of stabilization of these “rigid” PCLCD [27,39]. The transition of PCLCD into a “rigid” (insoluble) state takes place at definite concentration of Au-clusters. To check this we have used a scheme from “Materials and Methods” (see Controls 1, 2 and Experiment). It is known [11] that ds DNA CLCD exists only at high os- motic pressure of solvent determined by high concentra- tion of PEG. Hence, the dilution of ds DNA CLCD formed in PEG-containing solution with water solution to concentration of PEG much below “critical” value is accompanied by disintegration of the ds DNA PCLCD and formation of ds DNA isotropic solution. This process is usually accompanied by sharp decrease in the ampli- tude of an abnormal band in the CD spectrum. However, if the structure of PCLCD is stabilized, as a result of in- teraction with Au-nanoparticles, and can exist even at very low osmotic pressure of the solution, dilution proc- ess does not accompanied by disintegration of the struc- ture of ds DNA PCLCD. In this case the disappearance of an abnormal band in the CD spectrum does not take place. Figure 3 demonstrates the results of dilution of ds DNA CLCD treated with different concentration of Au- nanoparticles (size about 2 nm). One can see that at low (≤0.26 × 1014 particles per ml) concentration of Au-par- ticles added to ds DNA CLCD the process of its dilution is accompanied by disintegration of PCLCD, by forma- tion of isotropic ds DNA solution and, hence, by disap- pearance of an abnormal band in the CD spectra. How- ever, since “critical” (CcrNano-Au) concentration of Au- nanoparticles about 0.26 × 1014 particles per ml, two- times dilution leads only to twice decrease in concentra- tion of PCLCD and, hence, only in two-fold decrease in Figure 3. The dependences of the A270-values in the CD spectra for ds DNA CLCDs upon concentration of added Au-nanoparticles (2 nm; curve 1) and for these dispersions treated with Au-nanoparticles and then 2-times diluted with water (curve 2). 1: CDNA = 27 g/ml; CPEG = 150 mg/ml; 0.27 M NaCl + 1.78 × 10–3 M Na-phosphate buffer. 2: CDNA = 13 g/ml; CPEG = 75 mg/ml; 0.13 M NaCl + 0.89 × 10–3 M Na-phosphate buffer. A270x10–6 optical units; L = 1 cm; T = 22˚C. Ccr.Nano-Au—the “critical” concentration of the Au- nanoparticles. In area “I” the dilution of ds DNA CLCD is accompanied by full disintegration of particles, and final CD amplitude tends to zero; in area “II” the dilution results only in decrease in total concentration of PCLCD, and final CD amplitudes are equal every time to one half of initial values. the amplitude of an abnormal band (compare curves 1 and 2). The molecular circular dichroism (expressed as ∆ε value; ∆ε = ∆A/CDNA PLCD x L) [11] remains practi- cally constant for these cases. This result unequivocally means that since definite concentration of Au-nanopar- ticles their interaction with ds DNA molecules provides the conditions necessary for transition of ds DNA CLCD particles into a “rigid” state. (One can add that dilution of ds DNA CLCD treated with big Au-nanoparticles (size of 5 nm and 15 nm) is accompanied under all conditions by full disintegration of PCLCD and by formation of isotropic solutions of ds DNA molecules. The obtained result shows again that “big” Au-nanoparticles do not penetrate into the content of PCLCD and in- teract with ds NA molecules). Hence, the interaction of Au-nanoparticles (2 nm) with neighboring ds DNA molecules leads to the formation of a spatially fixed structure of the CLCD. The stability of this structure is determined by the number and properties of Au-clusters rather than the properties of the initial PEG-containing solution. This means that a new struc- ture would persist even in the absence of the high osmo- tic pressure of the solution. In this case it appears a pos- sibility to investigate the properties of a new-formed C opyright © 2011 SciRes. JBNB  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles Copyright © 2011 SciRes. JBNB 467 structure. (Here one can remind again, that a “liquid” mode of spatial packing of ds DNA molecules in the PCLCD prevents their immobilization on the surface of membrane filters. However, if poorly soluble (or insolu- ble) PCLCD consisting of molecules of the (ds DNA-Au) complexes are formed, the immobilization of these parti- cles on the surface of the nuclear membrane filter be- comes possible and the size and shape of these particles can be investigated). For this purpose, the insoluble PCLCD formed by (ds DNA-Au) complexes were immobilized on the surface of a nuclear membrane filter and the AFM images of these particles were registered. As an example, Figure 4 de- monstrates the images (2D and 3D) of ds DNA PCLCD treated with 0.82 × 1014 Au-nanoparticles per ml. The shape of the particles is close to elongated spheroids. One can stress that this shape is very similar to the shapes of the rigid PCLCD formed as a result of formation of nanobridges between neighboring ds DNA molecules, or decrease in solubility of ds NA molecules as a result of their treatment with the salt of rare-earth elements [40]. In the shown case, the sizes of particles are varied from 0.1 - 0.2 μm to 0.7 μm with 0.4 - 0.5 μm as the average, which is in good agreement with the data on the particle size theoretically calculated for the solutions with a con- stant osmotic pressure for the initial ds DNA CLCD. This means that the initial size of the PCLCD was not practically changed at interaction of Au-particles with ds DNA. Figure 4 confirms as well that ds DNA PCLCD treated with Au-particles exist as independent, nonsolu- ble, objects. The presence of single particles (Figure 4) testifies that at treatment of ds DNA PCLCD by Au- nanoparticles the “liquid” character of the DNA packing in these particles is disappeared and the formed particles have a “rigid” spatial structure. Besides, standard sizes of rigid PCLCD formed by (ds DNA-Au) complexes speaks against aggregation of Au-nanoparticles outside PCLCD. In favor of existence of ds DNA PCLCD in PEG- containing solutions as independent objects speaks the results obtained with confocal microscope. The ds DNA PCLCD formed in PEG-containing solution (170 mg/ml of PEG) was added with Au-nanoparticles and then was additionally treated with dye “SYBR Green”. This com- pound is capable not only of intercalating between nitro- gen bases of ds DNA, but also of retaining its high fluorescence, being incorporated even into content of PCLCD formed by ds DNA in PEG-containing solution. As an example, Figure 5 shows the fluorescence “im- ages” of particles of the ds DNA PCLCD treated with Au-nanoparticles and existing in solution. These images obtained by the confocal microscope demonstrate that particles of the ds DNA CLCD treated with Au-nanoparticles exist as isolated objects. The mean fluorescence “diameter” of these particles is simi- lar to mean diameter detected for initial ds DNA CLCD particles treated as well with “SYBR Green”. This indi- cates no deformation of the ds DNA secondary structure due to interaction with the Au-nanoparticles. The data shown in Figure 5 are important. They confirm that an appearance of the SPR band is connected with the as- sembling of Au-nanoparticles realized within individual particles of ds DNA CLCD, in the other words, with formation of Au-clusters between neighboring ds DNA molecules in quasinematic layers of PCLCD. Taking into account the above results, it was interest- ing to apply SAXS for investigation of phases formed by ds DNA CLCD treated with Au-nanoparticles. SAXS permits to probe an internal structure of the ob- ject at resolution from about 1 to 100 nm, and can pro- vide valuable structural information both about nano- scale inhomogeneities (particles or clusters) and about Figure 4. The AFM-images of the ds DNA CLCD particles treated with Au-nanoparticles (2 nm) and immobilized on the surface of a nuclear membrane (PETP) filter (2D: left panel; 3D: right panel). CNano-Au = 0.82 × 1014 particle/ml. (Dark spots are holes in the filter).  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles 468 Figure 5. The fuorescence “image” of PCLCD formed by ds DNA in PEG - containing solution and treated with Au- nanoparticles (2 nm) and then with SG. CDN A = 9 g/ml; CPEG = 150 mg/ml; 0.27 M NaCl + 1.78 × 10-3 M Na-phosphate buffer; CNano-Au = 0.82 × 1014 particle/ml; CSG = 4.11 × 10-6 M; T = 22˚C. the internal ordering in the sample. The size and shape of the scattering objects can be obtained from the portion of the scattering pattern close to the primary beam (central scattering), while the internal structure is determined using information containing in scattering profiles at higher scattering angles and Bragg peaks [41-44]. Small-angle scattering curves of samples obtained from these phases are shown in Figure 6. The cholesteric liquid-crystalline phase of initial ds DNA was used as a control sample (curve 1). First of all one can see that samples treated with Au-nanoparticles have diffuse Bragg maxima. Comparison of these curves shows that the interhelical distances (dinter) between ad- jacent ds DNA molecules in the analyzed samples con- taining Au-nanoparticles does not increase relatively to Au-free ds DNA CLCD sample. This means that the dis- tance between neighboring quasinematic layers is not changed as a result of incorporation of Au-nanoparticles into structure of ds DNA CLCD. One can say that local concentration of ds DNA molecules remains constant and structural transition due to change in concentration of ds DNA is impossible. Secondly, one can see that the higher Au content in the sample, the lower the amplitude of the Bragg peaks due to their flattening and broadening, which indicates that the ordering is diminished. Taking into account unchanged concentration of ds DNA mole- cules, this fact shows that the distortion of ordering of ds DNA molecules in neighboring quasinematic layers in Figure 6. The small-angle X-ray scattering curves of the ds DNA CLC phase (curve 1, control) and of the phases con- taining various concentrations of Au-nanoparticles (curves 2-4). 1 – CDNA = 27 g/ml, CPEG = 150 mg/ml, 0.27 M NaCl + 1.78 × 10-3 M Na-phosphate buffer, CNano-Au = 0; 2 – CNano-Au = 0.07 × 1014 particle/ml; 3 – CNano-Au = 0.33 × 1014 particle/ml; 4 – CNano-Au = 0.66 × 1014 particle/ml. Solvent, see conditions for curve 1. PCLCD is increased. This result correlates with the CD spectra shown in Figure 1, which gives evidence for a decrease in spatial ordering of ds DNA quasinematic layers when concentration of Au nanoparticles is in- creased. Hence, one can repeat again that the incorpora- tion of Au-nanoparticles into structure of quasinematic layers of ds DNA PCLCD conditions for collinear loca- tion of ds DNA molecules, i.e., this process is accompa- nied by untwisting of neighboring layers. Finally, sam- ples containing Au-nanoparticles are strongly scattering at very small angles, i.e., in the region of the central scattering, which points to a presence of sufficiently large polydisperse scattering objects (clusters of Au-na- noparticles). The viewpoint about possible linear arrangement of Au-nanoparticles and the results of size calculation of Au-clusters made by the program GNOM [45] allows one to suggest a hypothetical model reflecting the incor- poration of Au-nanoparticles between ds DNA molecules in quasinematic layers (Figure 7). According to this model, initial Au-nanoparticles have average diameter (size) of about 2 nm. These nanoparti- cles form linear clusters with an average length 13 - 15 nm and clusters with a maximal length of ~40 nm. For- mation of Au-clusters between ds DNA molecules in- duces violation of mutual orientation of neighboring qua- sinematic layers. Under these conditions the helical twisting of these layers in the structure of PCLCD is de- creased. Thus, the results of SAXS speaks in favor of the statement noted above, according to which the decrease in amplitude of an abnormal band in CD spectrum of ds C opyright © 2011 SciRes. JBNB  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles469 Figure 7. The hypothetical model of location of Au-clusters between ds DNA molecules forming the quasinematic layer in structure of PCLCD. DNA CLCD treated with Au-nanoparticles reflects the distortion in the arrangement of neighboring quasine- matic layers induced by formation of the Au-clusters between them [46]. As a result of this process, spatial structure of PCLCD loses its helical twisting and the transition of type “helically-twisted structure-structure without twisting” is induced. Besides, our calculations showed that the formation of Au-cross-links connecting neighboring ds DNA is possible. Due to the real position of neighboring ds DNA molecules in quasinematic layers, one can expect that in order to connect these molecules, the ends of Au-cross-link should wrap around the ds DNA molecules [29]. It is not excluded, that Au-cross- links can induce an additional stabilization of the “rigid” structure of ds DNA PCLCD. Hence, the obtained results demonstrate that interact- tion of Au-nanoparticles with ds NA CLCDs results in dual effect: i) it facilitates reorganization of the initial cholesteric structure, and ii) it induces the formation of Au-clusters in particles of ds NA CLCD. In conclusion, one can add, the effect of introducing Au-nanoparticles into cytoplasm of the cell has been in- vestigated by many scientific teams (see, for instance, [1,3,7,46]). However, yet little is known about effect of Au-nanopartricles at the nucleus of the cell. The cell nu- cleus functions to maintain all processes that occur with- in the cell, and any disruption within the nucleus would subsequently affect the cell’s DNA, thereby disturbing the highly regulated cell cycle. Here can remind that ds DNA CLCD particles reflect some structural and abnor- mal optical properties of Protozoa chromosomes. High efficiency of interaction of Au-nanoparticles with ds NA CLCDs allows one to suggest that PCLCD formed by ds NA molecules can, indeed, be considered as “molecular sponges”, easily “absorbing” nanoparticles of fixed size, interacting by this or that way with ds NA molecules. In the case of Au-nanoparticles such interaction is accom- panied by a strong change in the spatial structure of ini- tial PCLCD of ds NA. Therefore, the results of this study support the state- ment, according to which the distortion of mutual orien- tation of neighboring quasinematic layers formed by ds DNA molecule in spatial chromosome structure, induced by fixation of Au-nanoparticles between fragments of this molecule, can be one of the reasons for genotoxicity of these particles [2,3]. 4. Acknowledgements We want to express our thanks to Mikhail N. Savvateev (AIST-NT Co, Zelenograd, Russia) for his technical as- sistance at the study of the “rigid” PCLCD formed by (DNA-Au) complexes by means of AFM. REFERENCES [1] B. Kang, M. A. Mackey and M. A. El-Sayed, “Nuclear Targeting of Gold Nanoparticles in Cancer Cells Induces DNA Damage, Causing Cytokinesis Arrest and Apop- tosis,” Journal of the American Chemical Society, Vol. 132, No. 5, 2010, pp. 1517-1519. doi:10.1021/ja9102698 [2] V. Wiwanitkit, A. Sereemaspun and R. Rojanathanes, “Effect of Gold Nanoparticles on Spermatozoa: The First World Report,” Fertility & Sterility, Vol. 91, No. 1, 2009, pp. e7-e8. doi:10.1016/j.fertnstert.2007.08.021 [3] S. T. Zakhidov, T. L. Marshak, E. A. Malolina, A. Yu. Kulibin, I. A. Zelenina, S. M. Pavlyuchenkova, V. M. Ru- doy, O. V. Dement’eva, S. G. Skuridin and Y. M. Yevdo- kimov, “Gold Nanoparticles Impair Nuclear Chromatin Decondensation Process in Murine Sperm Cells in Vitro,” Biologicheskie membrany, Russian edition, Vol. 27, No. 4, 2010, pp. 349-353. [4] Y. P. Liu, W. Meyer-Zaika, S. Franzka, G. Schmid, M. Tsoli and H. Kuhn, “Gold-Cluster Degradation by Tran- sition of B-DNA into A-DNA and Formation of Nano- wires,” Angewandte Chemie, International Edition, Vol. 42, No. 25, 2003, pp. 2853-2857. doi:10.1002/anie.200250235 Copyright © 2011 SciRes. JBNB  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles 470 [5] M. Tsoli, H. Kuhn, W. Brandau, H. Esche and G. Schmid, “Cellular Uptake and Toxicity of Au55 Clusters,” Small, Vol. 1, No. 8-9, 2005, pp. 841-844. doi:10.1002/smll.200500104 [6] Y. Pan, S. Neuss , A. Leifert, M. Fischer, F. Wen, U. Si- mon, G. Schmid, W. Brandau and W. Jahnen-Dechent, “Size-Dependent Cytotoxicity of Gold Nanoparticles,” Small, Vol. 3, No. 11, 2007, pp. 1941-1949. doi:10.1002/smll.200700378 [7] E. Boisselier and D. Astruc, “Gold Nanoparticles in Na- nomedicine: Preparations, Imaging, Diagnostics, Thera- pies and Toxicity,” Chemical Society Reviews, Vol. 38, No. 6, 2009, pp. 1759-1782. doi:10.1039/b806051g [8] X. D. Zhang, M. L. Guo, H. Y. Wu, Y. Q. Ding, X. Feng and L. A. Zhang, “Irradiation Stability and Cytotoxicity of Gold Nanoparticles for Radiotherapy,” International Journal of Nanomedicine, Vol. 4, No. 9, 2009, pp. 165-173.doi:10.2147/IJN.S6723 [9] Y. Jin, M. Wu and X. Zhao, “Toxicity of Nanomaterials to Living Cells,” Technical Proceedings of the 2005 NSTI Nanotechnology Conference and Trade Show, Vol. 1, 2005, pp. 274-277. [10] J. Ai, E. Biazar, M. Jafarpour, M. Montazeri, A. Majdi, S. Aminifard, M. Zafari, H. R. Akbari and H. G. Rad, “Nanotoxicology and Nanoparticle Safety in Biomedical Designs,” International Journal of Nanomedicine, Vol. 6, No. 5, 2011, pp. 1117-1127. [11] Yu. M. Yevdokimov, V. I. Salyanov, S. V. Semenov and S. G. Skuridin, “DNA Liquid-Crystalline Dispersions and Nanostructures,” In: Yu. M. Yevdokimov, Ed., CRC Press, Taylor & Francis group, London, 2011, p. 320 [12] F. Livolant and A. Leforestier, “Condensed Phases of DNA: Structures and Phase Transitions,” Progress in Polymer Science, Vol. 21, No. 6, 1996, pp. 1115-1164. doi:10.1016/S0079-6700(96)00016-0 [13] Y. M. Yevdokimov, V. I. Salyanov, S. G. Skuridin, S. V. Semenov and O. N. Kompanets., “The CD Spectra of Doublestranded DNA Liquid-Crystalline Dispersions,” Nova Science Publishers, New York, 2011, p 103. [14] Y. M. Yevdokimov, S. G. Skuridin and B. A. Chernuha, “The Background for Creating Biosensors Based on Nu- cleic Acid Molecules,” In: A. P. F. Turner and Y. M. Ye- vdokimov, Eds., Advances in Biosensors, JAI Press, London, 1995, pp. 143-164. [15] J. Turkevich, P. C. Stevenson and J. Hillier, “A Study of the Nucleation and Growth Processes in the Synthesis of Colloidal Gold,” Discussions of the Faraday Society, Vol. 11, 1951, pp. 55-75. doi:10.1039/df9511100055 [16] K. R. Brown, D. G. Walter and M. J. Natan, “Seeding of Colloid-Dal Au-nanoparticle Solutions 2. Improved Con- trol of Particle Size and Shape,” Chemistry of Materials, Vol. 12, No. 2, 2000, pp. 306-313. doi:10.1021/cm980065p [17] D. G. Duff, A. Baiker and P. P. Edwards, “A New Hydrosol of Gold Clusters 1. Formation and Particle Size Variation,” Langmuir, Vol. 9, No. 9, 1993, pp. 2301-2309. doi:10.1021/la00033a010 [18] H. Zipper, H. Brunner, J. Bernhagen and F. Vitzthum, “Investigations on DNA Intercalation and Surface Bind- ing by SYBR Green I, Its Structure Determination and Methodological Implications,” Nucleic Acids Research, Vol. 32, No. 12, 2004, pp. e103. doi:10.1093/nar/gnh101 [19] O. N. Kompanets, “Portable Optical Biosensors for De- tection of Biologically Active and Toxic Compounds,” Uspekhi Fizicheskikh Nauk, Russian edition, Vol. 174, No. 6, 2004, pp. 686-690. [20] E. V. Shtykova, V. V. Volkov, V. I. Salyanov and Yu. M. Yevdokimov, “SAXS_Data_Based Structural Modeling of DNA-Gadolinium Complexes Fixed in Particles of Cholesteric Liquid-Crystalline Dispersions,” European Biophysics Journal, Vol. 39, No. 9, 2010, pp. 1313-1322. doi:10.1007/s00249-010-0584-0 [21] S. G. Skuridin, V. A. Dubinskaya, E. V. Shtykova, V. V. Volkov, V. M. Rudoy, O. V. Dement’eva, V. A. Kuzmin, E. S. Lisitsyna, S. T. Zakhidov, I. A. Zelenina and Yu. M. Yevdokimov, “Retention of Gold Nanoparticles in the Structure of Quasienematic Layers Formed by DNA Molecules,” Biologicheskie membrany, Russian edition, Vol. 28, No. 3, 2011, pp. 191–198. [22] P. V. Konarev, V. V. Volkov, A. V. Sokolova, M. H. J. Koch and D. I. Svergun, “PRIMUS: A Windows PC- Based System for Small-Angle Scattering Data Analy- sis,” Journal of Applied Crystallography, Vol. 36, 2003, No. 5, pp. 1277–1282. doi:10.1107/S0021889803012779 [23] V. A. Belyakov, V. P. Orlov, S. V. Semenov, S. G. Sku- ridin and Yu. M. Yevdokimov, “Comparison of Calcu- lated and Observed CD Spectra of Liquid Crystalline Dispersions Formed from Double-Stranded DNA and from DNA Complexes with Colored Compounds,” Liquid Crystals, Vol. 20, No. 6, 1996, pp. 777-784. doi:10.1080/02678299608033172 [24] C. A. Mirkin, R. L. Letsinger, R. C. Mucic and J. J. Stor- hoff, “A DNA-Based Method for Rationally Assembling Nanoparticles into Macroscopic Materials,” Nature, Vol. 382, No. 6592, 1996, pp. 607-609. doi:10.1038/382607a0 [25] A. P. Alivisatos, K. P. Johnsson, X. Peng, T. E. Wilson, C. J. Loweth, M. P. Bruchez and P. Scultz, “Organization of Nanocrystal Molecules Using DNA,” Nature, Vol. 382, No. 6592, 1996, pp. 609-611. doi:10.1038/382609a0 [26] A. Kumar, M. Pattarkine, M. Bhadbhade, A. B. Mandale, K. N. Ganesh, S. S. Datar, C. V. Dharmadhikari and M. Sastry, “Linear Superclusters of Colloidal Gold Particles by Electrostatic Assembly on DNA Templates,” Ad- vanced Materials, Vol. 13, No. 5, 2001, pp. 341-344. doi:10.1002/1521-4095(200103)13:5<341::AID-ADMA3 41>3.0.CO;2-X [27] M. Sastrya, A. Kumar, S. Datar and C. V. Dharmadhikari, “DNA-Mediated Electrostatic Assembly of Gold Nano- particles into Linear Arrays by a Simple Drop-Coating Procedure,” Applied Physics Letters, Vol. 78, No. 19, 2001, pp. 2943-2945. doi:10.1063/1.1370993 [28] M. G. Warner and J. E. Hutchison, “Linear Assemblies of Nanoparticles Electrostatically Organized on DNA Scaf- folds,” Nature Materials, Vol. 2, No. 4, 2003, pp. 272- 277. doi:10.1038/nmat853 C opyright © 2011 SciRes. JBNB  A Dual Effect of Au-Nanoparticles on Nucleic Acid Cholesteric Liquid-Crystalline Particles Copyright © 2011 SciRes. JBNB 471 [29] L. V. Zherenkova, P. V. Komarov and P. G. Khakatur, “Simulation of the Metallization of a Fragment of a De- Oxyribonucleic Acid Molecule with Gold Nanoparticles,” Colloid Journal, Russian edition, Vol. 69, No. 6, 2007, pp. 753- 765. [30] S. Link and M. A. El-Sayed, “Spectral Properties and Relaxation Dynamics of Surface Plasmon Electronic Os- cillations in Gold and Silver Nanodots and Nanorods,” The Journal of Physical Chemistry B, Vol. 103, No. 40, 1999, pp. 8410-8426. doi:10.1021/jp9917648 [31] S. L. Westcott, S. J. Oldenburg, T. R. Le and N. J. Halas, “Construction of Simple Gold Nanoparticle Aggregates with Controlled Plasmon-Plasmon Interactions,” Chemi- cal Physics Letters, Vol. 300, No. 5-6, 1999, pp. 651-655. doi:10.1016/S0009-2614(98)01410-9 [32] S. Link and M. A. El-Sayed, “Shape and Size Depen- dence of Radioactive, Non Radioactive and Photothermal Properties of gold Nanocrystals,” International Reviews in Physical Chemistry, Vol. 19, No. 3, 2000, pp. 409-453. doi:10.1080/01442350050034180 [33] K. H. Su, Q. H. Wei, X. Zhang, J. J. Mock, D. R. Smith and S. Schultz, “Interparticle Coupling Effects on Plas- mon Resonances of Nanogold Particles,” Nano Letters, Vol. 3, No. 8, 2003, pp. 1087-1090. doi:10.1021/nl034197f [34] W. Rechberger, A. Hohenau, A. Leitner, J. R. Krenn, B. Lamprecht and F. R. Aussenegg, “Optical Properties of Two Interacting Gold Nanoparticles,” Optics Communi- cations, Vol. 220, No. 1-3, 2003, pp. 137-141. doi:10.1016/S0030-4018(03)01357-9 [35] P. V. Kamat, “Photophysical, Photochemical and Photo- catalytic Aspects of Metal Nanoparticles,” The Journal of Physical Chemistry B, Vol. 106, No. 32, 2002, pp. 7729- 7744. doi:10.1021/jp0209289 [36] N. G. Khlebtsov, A. G. Melnikov, L A. Dykman. and V. A Bogatyrev, “Optical Properties and Biomedical Appli- cations of Nanostructures Based on Gold and Silver Bio- conjugates. Photopolarimetry in Remote Sensing,” In: G. Videen, Ya. S. Yatskiv and M. I. Mishchenko, Eds., NATO Science Series, II. Mathematics, Physics, and Chemistry, Vol. 161, 2004, pp. 265-308. [37] L. Dykman, V. Bogatyrev, S. Shchyogolev and N. Khle- btsov, “Gold Nanoparticles: Synthesis, Properties, Bio- medical Applications,” Nauka, Russian Edition, Moscow, 2008, pp. 70-78. [38] P. Mulvaney, “Surface Plasmon Spectroscopy of Nano- sized Metal Particles,” Langmuir, Vol. 12, No. 3, 1996, pp. 788-800. doi:10.1021/la9502711 [39] G. Decher, “Fuzzy Nanoassemblies: Toward Layered Po- lymeric Multicomposites,” Science, Vol. 277, No. 5330, 1997, pp. 1232-1237. doi:10.1126/science.277.5330.1232 [40] Yu. M. Yevdokimov, “From Particles of Liquid Crystal- line Dispersions to Rigid Deoxyribonucleic Acid Nano- constructions,” Liquid Crystals Today, Vol. 20, No. 1, 2011, pp. 2-19. doi:10.1080/1358314X.2011.549300 [41] A. Guinier and G. Fournet, “Small-Angle Scattering of X-Rays,” John Wiley and Sons, New York, 1955, p. 269. [42] O. Glatter and O. Kratky, “Small-Angle X-Ray Scatter- ing,” Academic Press, London, 1982, p. 515. [43] L. A. Feigin and D. I. Svergun, “Structure Analysis by Small-Angle X-ray and Neutron Scattering,” Plenum Press, New York, 1987. [44] H. D. Mertens, D. I. Svergun, “Structural Characteriza- tion of Proteins and Complexes Using Small-Angle X-ray Solution Scattering,” Journal of Structural Biology, Vol. 172, No. 1, 2010, pp. 128-141. doi:10.1016/j.jsb.2010.06.012 [45] D. I. Svergun, A. V. Semenyuk and L. A. Feigin, “Small-Angle-Scattering-Data Treatment by the Regu- larization Method,” Acta Crystallographica, Vol. A44, 1988, pp. 244-250. [46] A. Rocha, Y. Zhou, S. Kundu, J. M. Gonzalez, S. B. Vin- son and H. Liang, “In vivo Observation of Gold Nanopar- ticles in the Central Nervous System of Blaberus dis- coidalis,” Journal of Nanobiotechnology, Vol. 9, No. 1, 2011. doi:10.1186/1477-3155-9-5 Abbreviations Au nanoparticles: gold nanoparticles, CD: circular dichroism, OD: optical density, CLCD: cholesteric liquid-crystalline dispersion, Ds: double-stranded, NA: nucleic acid, PCLCD: particles of cholesteric liquid-crystalline disper- sion, SPR: surface plasmon resonance, AFM: atomic force microscopy, SAXS: small-angle X-ray scattering, SG: SYBR Green I, PEG: poly(ethylene glycol), PETP: poly(ethylene therephtalate).
|