Paper Menu >>
Journal Menu >>
 Journal of Behavioral and Brain Science, 2011, 1, 160-166 doi:10.4236/jbbs.2011.13021 Published Online August 2011 (http://www.SciRP.org/journal/jbbs) Copyright © 2011 SciRes. JBBS Inhibition in Action―Inhibitory Components in the Behavioral Activation System Stefan Sütterlin1, Stein Andersson2, Claus Vögele1 1Integrative Research Unit on Social and Individual Development (INSIDE), University of Luxembourg, Luxembourg City, Luxembourg 2Oslo University Hospital―Rikshospitalet, Oslo, Norway E-mail: stefan.suetterlin@uni.lu Received June 8, 2011; revised July 19, 2011; accepted July 26, 2011 Abstract Over the past two decades, the neurobiological substrates of the reinforcement theory have been discussed in terms of a behavioral activation system (BAS) and a behavioral inhibition system (BIS). While the BAS has been conceptualized as both an activating system and an approach-related system, the empirical evidence for either approach remains inconclusive. In the current study we hypothesize that the inclusion of self-regula- tory capacity contributes to a better understanding of the BAS. In a sample of 29 volunteers motor response inhibition elicited by a stop-signal task and heart rate variability (HRV) as a proxy of self-regulatory capacity were related to BAS scores (BIS/BAS scales [1]). Results show significant positive associations between inhibitory capacity and the sensitivity of the behavioral activation system, suggesting markers of self-regu- lation as components of the BAS. Keywords: Behavioral Activation System, Heart Rate Variability, Stop-Signal Task, Self-Regulation 1. Introduction Over the past two decades, extensive research has been conducted to investigate the reinforcement sensitivity the- ory [2,3], its neurobiological substrates, related personality traits [4] and psychopathology [5], and physiological in- dicators. In the original formulation of their model, Gray and colleagues [2,3] suggested a behavioral activation system (BAS) and a behavioral inhibition system (BIS), which are typically operationalized with the BIS/BAS- Scales [1] at self-report level. However, based on their research on the neurobiological substrates of these sys- tems, Sutton and Davidson [6] conceptualized the be- havioral approach system (BAS), which is opposed to the behavioral inhibition system (BIS). If the BAS scales indeed measure behavioral activation, independent from behavioral direction, and in the more comprehensive sense of intended alterations of spatial proximity (approach or active avoidance), then this would be contrary to purely approaching behavior. BAS scores should, therefore, be positively related to physiological indicators of efficient self-and emotion-regulation. Self- regulation describes the individual’s ability to adapt behaviorally, emotionally and cognitively to constantly changing environmental demands. This includes goal-directed behavior, the abil- ity to resist temptations, to overcome competing or pre- potent action tendencies, to make elaborated decisions in order to regulate emotional, cognitive and motor res- ponses to optimize future outcome (overview [7]). Self- regulation is conceived as a personality trait and can be objectively assessed under laboratory conditions, typi- cally via physiological and behavioral indicators of pre- frontally mediated inhibitory control mechanisms, using motor response paradigms. Motor response inhibition paradigms, such as the stop-signal task (SST), induce suppression of automatized, pre-potent motor behavior in pre-defined, infrequent and unpredictable cases; they require focused attention, stimulus discrimination, choice of the appropriate reaction and its execution. These pro- cesses can be subsumed under the broader term execu- tive functions. In the present study it is hypothesized that performance in a motor response inhibition paradigm is positively associated with BAS scores. Resting vagal tone has been identified as a peripheral physiological correlate of BAS scores. Early research reported a positive relationship between approach-related  S. SÜTTERLIN ET AL. Copyright © 2011 SciRes. JBBS 161 behavior and resting vagal tone [8,9], preparing the ground for later findings with Carver and White’s [1] BAS-scale by researchers comparing physiological meas- ures and BAS scores [10,11]. The positive relationship between vagal tone and BAS scores has been interpreted in terms of mechanisms of emotional, self-regulatory, and behavioral processes, according to the evolutionary theory proposed by Porges [12-14]. Nevertheless, vagal tone at rest can also be conceived as a measure of self- regulatory and inhibitory capacity. Executive functions and their association with regulatory competence and their corresponding neurophysiological substrates have been outlined in a model of neurovisceral integration, which is complementary to Porges’ more philogenetic approach. The model of neurovisceral integration de- scribes inhibitory cortico-cardiac interactions mediated by the vagus nerve and supported by the inhibitory trans- mitter γ-aminobutyric acid (GABA) [15-17]. A first aim of the present study was to replicate the reported positive association between vagal tone and BAS scores. The main aim of the present study, however, was the investi- gation of the role of inhibition in the organization of cog- nition, behavior and affect. Inhibitory processes are a crucial component of behavioral adaptation. In the pre- sent study measures of inhibitory capacity are operation- alized as motor response inhibition performance (per- centage of correctly inhibited motor responses), inhibi- tory speed (stop-signal reaction time), and heart rate va- riability (HRV), the latter indicating vagally mediated in- hibitory cardiac control. We hypothesize that these mea- sures of inhibitory control show a positive association with BAS scores, thus supporting the assumption of BAS resembling a behavioral activation system, which is closely linked to executive functions tapping inhibitory re- sources required for action planning and control. 2. Materials and Methods 2.1. Participants Twenty-nine healthy participants (20 women, 9 men) were recruited via advertisement from the staff of the Oslo University Hospital. Age ranged from 19 to 47 years (M = 29.3, SD = 6.5). Participants received a fi- nancial compensation for taking part in the study. Exclu- sion criteria were self-reports of current and previous psychiatric, neurological, or cardiovascular diagnoses, and medication affecting the central nervous or cardio- vascular system. The study was approved by the Re- gional Ethical Committee of South-Eastern Norway and all subjects gave written informed consent to participate, in accordance with the Helsinki Declaration of 1975 (as revised in 1983). 2.2. Material and Experimental Tasks Stop-signal task: The “GO” stimuli consisted of the let- ters “S” or “B”, presented on a 19-inch computer display using E-Prime software (v2.0, Psychology Software Tools, Pittsburgh, PA, 2007). Stimuli were presented in black on white background, viewing distance from the screen was 80 - 90 cm. Stimuli covered an angle of ap- proximately 3.5˚ × 2˚ of the visual field. “GO” stimuli were presented for 500 ms, followed by an intertrial in- terval (ITI) of 1500 ms. The total number of trials was 600; in 150 trials (25%) the “GO” stimulus was followed by an acoustic signal (1000 Hz, 500 ms) acting as a stop signal. Stimulus onset asynchrony (SOA) between “GO” and “STOP” signal was 100 ms, 200 ms, or 300 ms, as determined by a performance-related staircase-tracking algorithm [18], ensuring a similar level of subjective difficulty of about 50% accuracy for all participants. Par- ticipants were instructed to press a button as fast as pos- sible as soon as either letter appears on the screen, but to inhibit their response in those cases where the auditory stop signal occurred. Recovery breaks after 200 and 400 items provided the possibility to relax. 2.3. Physiological Assessment Electrocardiographic recording: Electrocardiogram (ECG) was monitored using the Einthoven configuration with disposable electrodes attached to the non-dominant wrist and the opposite ankle. To reduce the probability of move- ment artifacts and ensure regular breathing cycles par- ticipants were instructed to relax and close their eyes while monitoring ensued for a period of 10 min. ECG raw data were recorded using a Neuroscan polygraph (Neuroscan, Charlotte/NC), sampled at 512 Hz. 2.4. Data Reduction and Statistical Analysis Stop-signal task: Stop-signal reaction time (SSRT) and percentage of correctly suppressed reactions in “STOP” trials were calculated following the recommendations made by Logan (for details see: [19,20]), collapsing the rank-ordered reaction times of “GO” trials into a single distribution where the SSRT is identified on basis of the probability of a response in “STOP” trials. This process is repeated for each stop signal delay for each subject. The results are then averaged over subjects within and sometimes cross stop signal delays. Stop-signal reaction time estimates the speed of the inhibitory process in mil- liseconds, with lower value reflecting faster inhibitory processing. Vagal tone: offline analyses of ECG included the ex- 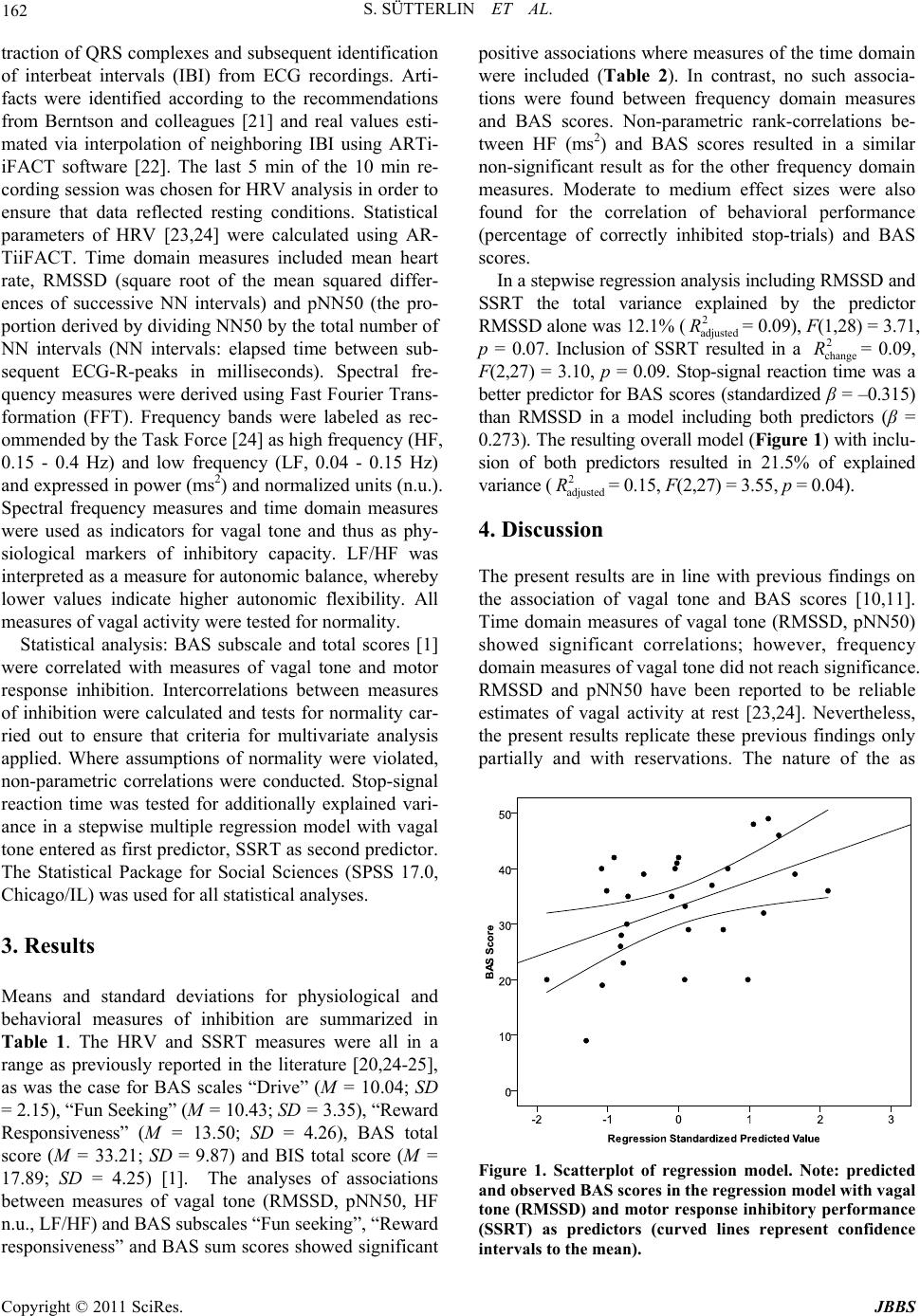 S. SÜTTERLIN ET AL. Copyright © 2011 SciRes. JBBS 162 traction of QRS complexes and subsequent identification of interbeat intervals (IBI) from ECG recordings. Arti- facts were identified according to the recommendations from Berntson and colleagues [21] and real values esti- mated via interpolation of neighboring IBI using ARTi- iFACT software [22]. The last 5 min of the 10 min re- cording session was chosen for HRV analysis in order to ensure that data reflected resting conditions. Statistical parameters of HRV [23,24] were calculated using AR- TiiFACT. Time domain measures included mean heart rate, RMSSD (square root of the mean squared differ- ences of successive NN intervals) and pNN50 (the pro- portion derived by dividing NN50 by the total number of NN intervals (NN intervals: elapsed time between sub- sequent ECG-R-peaks in milliseconds). Spectral fre- quency measures were derived using Fast Fourier Trans- formation (FFT). Frequency bands were labeled as rec- ommended by the Task Force [24] as high frequency (HF, 0.15 - 0.4 Hz) and low frequency (LF, 0.04 - 0.15 Hz) and expressed in power (ms2) and normalized units (n.u.). Spectral frequency measures and time domain measures were used as indicators for vagal tone and thus as phy- siological markers of inhibitory capacity. LF/HF was interpreted as a measure for autonomic balance, whereby lower values indicate higher autonomic flexibility. All measures of vagal activity were tested for normality. Statistical analysis: BAS subscale and total scores [1] were correlated with measures of vagal tone and motor response inhibition. Intercorrelations between measures of inhibition were calculated and tests for normality car- ried out to ensure that criteria for multivariate analysis applied. Where assumptions of normality were violated, non-parametric correlations were conducted. Stop-signal reaction time was tested for additionally explained vari- ance in a stepwise multiple regression model with vagal tone entered as first predictor, SSRT as second predictor. The Statistical Package for Social Sciences (SPSS 17.0, Chicago/IL) was used for all statistical analyses. 3. Results Means and standard deviations for physiological and behavioral measures of inhibition are summarized in Table 1. The HRV and SSRT measures were all in a range as previously reported in the literature [20,24-25], as was the case for BAS scales “Drive” (M = 10.04; SD = 2.15), “Fun Seeking” (M = 10.43; SD = 3.35), “Reward Responsiveness” (M = 13.50; SD = 4.26), BAS total score (M = 33.21; SD = 9.87) and BIS total score (M = 17.89; SD = 4.25) [1]. The analyses of associations between measures of vagal tone (RMSSD, pNN50, HF n.u., LF/HF) and BAS subscales “Fun seeking”, “Reward responsiveness” and BAS sum scores showed significant positive associations where measures of the time domain were included (Table 2). In contrast, no such associa- tions were found between frequency domain measures and BAS scores. Non-parametric rank-correlations be- tween HF (ms2) and BAS scores resulted in a similar non-significant result as for the other frequency domain measures. Moderate to medium effect sizes were also found for the correlation of behavioral performance (percentage of correctly inhibited stop-trials) and BAS scores. In a stepwise regression analysis including RMSSD and SSRT the total variance explained by the predictor RMSSD alone was 12.1% (2 adjusted R= 0.09), F(1,28) = 3.71, p = 0.07. Inclusion of SSRT resulted in a 2 change R= 0.09, F(2,27) = 3.10, p = 0.09. Stop-signal reaction time was a better predictor for BAS scores (standardized β = –0.315) than RMSSD in a model including both predictors (β = 0.273). The resulting overall model (Figure 1) with inclu- sion of both predictors resulted in 21.5% of explained variance (2 adjusted R= 0.15, F(2,27) = 3.55, p = 0.04). 4. Discussion The present results are in line with previous findings on the association of vagal tone and BAS scores [10,11]. Time domain measures of vagal tone (RMSSD, pNN50) showed significant correlations; however, frequency domain measures of vagal tone did not reach significance. RMSSD and pNN50 have been reported to be reliable estimates of vagal activity at rest [23,24]. Nevertheless, the present results replicate these previous findings only partially and with reservations. The nature of the as Figure 1. Scatterplot of regression model. Note: predicted and observed BAS scores in the regression model with vagal tone (RMSSD) and motor response inhibitory performance (SSRT) as predictors (curved lines represent confidence intervals to the mean).  S. SÜTTERLIN ET AL. Copyright © 2011 SciRes. JBBS 163 Table 1. Inhibitory measures. n Min Max Mean SD HRV RMSSD 29 19.90 79.00 39.32 15.65 pNN50 29 1.00 54.20 20.76 17.12 HF (ms2) 29 58 555 217 150 HF (n.u.) 29 17.00 65.70 44.26 14.97 LF/HF 29 0.52 3.88 1.31 0.87 SST SSRT (ms) 29 134 279 201 38.35 Correct inh. (%) 29 25.79 94.83 55.77 18.54 Table 2. Correlations of inhibitory measures and BIS/BAS scores. BAS DriveBAS FS BAS RR BAS SumBIS Sum HRV RMSSD 0.13 0.38* 0.35* 0.36* –0.10 pNN50 0.13 0.39* 0.34* 0.37* –0.03 HF (ms2) 0.02 0.10 –0.04 0.06 –0.08 HF (n.u.) 0.13 0.02 –0.16 –0.07 –0.20 LF/HF –0.09 –0.02 0.17 0.06 0.17 SST SSRT (ms) –0.20 –0.25 –0.37* –0.39* –0.23 Correct inh. (%) 0.41* 0.41* 0.38* 0.27 0.11 Note: values represent Pearson’s correlation coefficient r for all variables with the exception of HF (ms2). Correlations with HF (ms2) are rank correlated (Spearman’s rs); * p < 0.05. ** p < 0.01, (one-tailed). FS = fun seeking, RR = reward responsiveness. sumed and previously reported association between parasympathetic activation at rest and a pronounced be- havioral approach or activation trait has not been specified yet, with explanations limited to, e.g., “emotional, self- regulatory, and behavioral processes” [11]. Vagal tone reflects the activity of the X. cranial nerve, mediating the cortico-cardiac modulation indicated by HRV. Neuro- biological models have approached the phenomenon of respiration-induced heart-rate oscillations at rest from different perspectives. Previously, the philogenetic per- spective suggested by Porges [12,13,26] was referred to as an explanatory model for the observed association. In the present study a different but complementary theo- retical approach was taken by deriving explicitly inhibi- tion-oriented hypotheses from the model of neurovisceral integration [17] as a key process in the proposed central autonomic network (CAN). This network has been de- scribed as crucially depending on frontal inhibitory input and includes GABAergic neuronal networks involved in inhibitory action in emotional, cognitive, and behavioral domains [15,17,27,28]. The CAN depicts a model of neurovisceral integration, in which frontal inhibitory in- put provides the means for self-regulated action and regulated emotional responding via an extensive cortico- cardiac network enabling the organism to adapt flexibly to changing environmental needs, to focus attention, and to facilitate executive functioning in terms of planning and executing goal-directed behavior. Vagal activity is known to be related to inhibition-intensive processing such as working memory [29], and executive function [17] and has recently also been shown to play a role in higher-order decision-making processes such as over- coming distracting emotional biases in individual or so- cial context [30,31]. Based on the present results it is argued that the association of vagal tone and BAS scores is linked to frontal inhibitory capacity as a component of  S. SÜTTERLIN ET AL. Copyright © 2011 SciRes. JBBS 164 executive control. This interpretation is supported by the positive relation on a behavioral level between stop-signal reaction time representing effectiveness of inhibitory pro- cesses interrupting pre-potent motor responses and BAS scores. In the light of the present findings, previous no- tions suggesting that vagal activity and the BAS scores are positively correlated could be revised and extended insofar as measures of inhibitory capacity are positively related to BAS scores. We concede that further research is needed to replicate these findings in larger samples, possibly applying alternative measures of inhibitory ca- pacity such as, e.g., antisaccadic eye-movements and be- havioral measures of executive control. The role of inhibitory processes for executive func- tions might explain the close association with BAS scores. Executive functions and their underlying components such as goal-directed behavior, working memory, and re- gulated emotional responding make intensive use of pre- frontally originating inhibitory processes [32]. In contrast to behavioral inhibition as indexed by the BIS-scale, the BAS occasionally requires conscious decision-making and self-regulatory competences mirrored in delay of gratifica- tion, sequential action plans and higher-order processing. Components of action control such as these are linked to prefrontal functions and inhibition in particular. They increase the likelihood of successful action and thus in- crease the probability of behavior as assessed by the BAS-scale. Regarding the debate of BAS as a behavioral activa- tion or behavioral approach system, the present findings support the idea of a behavioral activation system re- gardless of locomotive or motivational direction, defined as either approach or active avoidance. The concept of executive functions describes the neuronal and physio- logical basis for consciously planned and goal-directed behavioral competence regardless of its direction, exactly as does frontal inhibition as indexed by performance in the stop-signal task, inhibitory event-related potentials and vagal tone. Inhibitory measures constitute the organ- ism’s adaptability regardless of direction, but dependent on prefrontal neuronal activity. The present study aimed to contribute to the understanding of the mechanisms underlying the behavioral activation system with par- ticular respect to the nature of its postulated association with vagal tone. Recent research on relative frontal activation largely supports the concept of a behavioral activation system. In contrast to Sutton and Davidson [6], Harmon-Jones and Allen [33] reported bilateral activity to be associated with increased BAS scores. These findings were repli- cated by Wacker and colleagues [34], suggesting that the BAS is a behavioral activation system facilitating goal- directed behavior regardless of direction. Further con- firmation for the notion of a behavioral activation system (as opposed to a behavioral approach system) comes from a study by Hewig and colleagues [35]. In summary, these results are in line with the earlier suggestion by Gray and McNaughton [3] that active avoidance is part of the BAS. Hewig and colleagues [35,36] dissected the components of motivation and affective state, and re- ported motivational direction to be associated with fron- tal asymmetry, but behavioral activation per se to be re- lated to greater bilateral activity. Given the controversially discussed issue regarding the operationalization of BIS/BAS and anterior asymme- try, we restricted our research to the investigation of un- derlying processes promoting relatively higher BAS- scores. Our results suggest inhibitory capacity as an endo- phenotypic trait marker of a pronounced behavioral acti- vation system. We further suggest that the associations for various markers of inhibitory measures reported in the literature [10] and the results presented in the present study are in line with the assumption of a behavioral ac- tivation system, indicating higher behavioral regulation competence in individuals scoring high on the BAS scale. Correlations of inhibitory measures and BIS score were not subject to the present study. The lack of corre- lations between inhibitory measures and the “behavioral inhibitory system” might appear counter-intuitive. BIS does not involve action, but the interruption and avoid- ance of action. High BIS scores have been reported to be associated with high reactivity to negative and poten- tially threatening cues and anxiety [37], the opposite of “regulated emotional responding”, which has been asso- ciated with prefrontal function and vagal tone [17,27]. High BIS scores reflect poor emotion regulation. In con- trast, the inhibition of behavior intuitively suggests a positive association between physiological correlates of inhibitory capacity and BIS scores, which is supported by empirical data linking dorsolateral prefrontal cortex (DLPFC) activity with BIS scores [38]. Heart rate vari- ability is not a specific measure and involves the large multilevel model of CAN. As such it is exposed to vari- ous influences of diverging directions as they are re- flected in BIS items. The BIS scale as it is conceptual- ized does not find an equivalent in the CAN or inhibition measures as such, particularly not a linear relationship. 5. Conclusions The present results are in line with previous findings reporting a positive association of vagal tone and BAS score. This association was exceeded by a positive rela- tionship of BAS score and motor response inhibition performance as well as the stop-signal reaction time, a  S. SÜTTERLIN ET AL. Copyright © 2011 SciRes. JBBS 165 measure of inhibitory efficacy [19]. Taken together, both measures of self-regulation and via inhibitory control complement each other in predicting BAS scores on the BIS/BAS scale. Thus, the positive association of inhibi- tory capacity and BAS scores provide arguments for the notion that the BAS represents a behavioral activation system, not a behavioral approach system. Inhibitory control is both, a key element of behavioral activation and executive functioning. 6. References [1] C. S. Carver and T. L. White, “Behavioral Inhibition, Behavioral Activation, and Affective Responses to Im- pending Reward and Punishment: The BIS/BAS Scales,” Journal of Personality and Social Psychology, Vol. 67, No. 2, 1994, pp. 319-333. HHdoi:10.1037/0022-3514.67.2.319 [2] J. A. Gray, “Précis of the Neuropsychology of Anxiety: An Enquiry into the Functions of the Septo-Hippocampal System,” The Behavioral and Brain Sciences, Vol. 5, No. 3, 1982, pp. 469-534. HHdoi:10.1017/S0140525X00013066 [3] J. A. Gray and N. McNaughton, “The Neuropsychology of Anxiety: Reprise,” In: D. A. Hope, Ed., Perspectives on Anxiety, Panic and Fear, University of Nebraska Press, Lincoln, 1996, pp. 61-134. [4] J. J. Simon, S. W. Walther, C. J. Fiebach, H. C. Fried- erich, C. Stippich, M. Weisbrod and S. Kaiser, “Neural Processing is Modulated by Approach- and Avoidance- Related Personality Traits,” NeuroImage, Vol. 49, No. 2, 2010, pp. 1868-1874. HHdoi:10.1016/j.neuroimage.2009.09.016 [5] M. R. M. Scholten, J. van Honk, A. Aleman and R. S. Kahn, “Behavioral Inhibition System (BIS), Behavioral Activation System (BAS) and Schizophrenia: Relation- ship with Psychopathology and Physiology,” Journal of Psychiatric Research, Vol. 40, No. 7, 2006, pp. 638-645. [6] S. K. Sutton and R. J. Davidson, “Prefrontal Brain Asym- metry: A Biological Substrate of the Behavioral Approach and Inhibition Systems,” Psychological Science, Vol. 8, No. 3, 1997, pp. 204-210. HHdoi:10.1111/j.1467-9280.1997.tb00413.x [7] R. F. Baumeister and K. D. Vohs, “Handbook of Self- Regulation,” The Guilford Press, New York, 2004. [8] J. E. Richards, “Respiratory Sinus Arrhythmia Predicts Heart Rate and Visual Responses during Visual Attention in 14- and 20-Week-Old Infants,” Psychophysiology, Vol. 22, No. 1, 1985, pp. 101-109. HHdoi:10.1111/j.1469-8986.1985.tb01567.x [9] N. A. Fox, “Psychophysiological Correlates of Emotional Reactivity during the First Year of Life,” Developmental Psychology, Vol. 25, No. 3, 1989, pp. 364-372. HHdoi:10.1037/0012-1649.25.3.364 [10] S. L. Brenner, T. P. Beauchaine and P. D. Sylvers, “A Comparison of Psychophysiological and Self-Report Mea- sures of BAS and BIS Activation,” Psychophysiology, Vol. 42, No. 1, 2005, pp. 108-115. HHdoi:10.1111/j.1469-8986.2005.00261.x [11] H. L. Movius and J. J. B. Allen, “Cardiac Vagal Tone, Defensiveness, and Motivational Style,” Biological Psy- chology, Vol. 68, No. 2, 2005, pp. 147-162. HHdoi:10.1016/j.biopsycho.2004.03.019 [12] S. W. Porges, “Vagal Tone: An Autonomic Mediator of Affect,” In: J. Garber and K. A. Dodge, Eds., Principles of Psychophysiology: Physical, Social, and Inferential Ele- ments, Cambridge University Press, Cambridge, 1991, pp. 456-512. [13] S. W. Porges, “Orienting in a Defensive World: Mam- malian Modifications of Our Evolutionary Heritage. A Polyvagal Theory,” Psychophysiology, Vol. 32, No. 4, 1995, pp. 301-318. HHdoi:10.1111/j.1469-8986.1995.tb01213.x [14] N. Eisenberg, R. A. Fabes, B. Murphy, P. Maszk, M. Smith and M. Karbon, “The Role of Emotionality and Regulation in Children’s Social Functioning: A Longitu- dinal Study,” Child Development, Vol. 66, No. 5, 1995, pp. 1360-1384. HHdoi:10.2307/1131652 [15] E. E. Benarroch, “The Central Autonomic Network: Functional Organization, Dysfunction, and Perspective,” Mayo Clinic Proceedings, Vol. 68, No. 10, 1993, pp. 988- 1001. [16] J. F. Thayer and J. F. Brosschot, “Psychosomatics and Psychopathology: Looking up and down from the Brain,” Psychoneuroendocrinology, Vol. 30, No. 10, 2005, pp. 1050-1058. HHdoi:10.1016/j.psyneuen.2005.04.014 [17] J. F. Thayer and R. D. Lane, “Claude Bernard and the Heart-Brain Connection: Further Elaboration of a Model of Neurovisceral Integration,” Neuroscience and Biobe- havioral Reviews, Vol. 33, No. 2, 2009, pp. 81-88. HHdoi:10.1016/j.neubiorev.2008.08.004 [18] M. Boecker, M. M. Buecheler, M. L. Schroeter and S. Gauggel, “Prefrontal Brain Activation during Stop-Signal Response Inhibition: An Event-Related Functional Near- Infrared Spectroscopy Study,” Behavioural Brain Re- search, Vol. 176, No. 2, 2007, pp. 259-266. HHdoi:10.1016/j.bbr.2006.10.009 [19] G. D. Logan and W. B. Cowan, “On the Ability to Inhibit thought and Action: A Theory of an Act of Control,” Psychological Review, Vol. 91, No. 3, 1984, pp. 295-327. HHdoi:10.1037/0033-295X.91.3.295 [20] G. D. Logan, “On the Ability to Inhibit Thought and Ac- tion: A User’s Guide to the Stop-Signal Paradigm,” In: D. Dagenbach and T. H. Carr, Eds., Inhibitory Processes in Attention, Memory, and Language, Academic Press, San Diego, 1994, pp. 184-239. [21] G. G. Berntson, K. S. Quigley, J. F. Jang and S. T. Boy- sen, “An Approach to Artifact Identification: Application to Heart Period Data,” Psychophysiology, Vol. 27, No. 5, 1990, pp. 586-598. HHdoi:10.1111/j.1469-8986.1990.tb01982.x [22] T. Kaufmann, S. Sütterlin, S. M. Schulz and C. Vögele, “ARTiiFACT: A Tool for Heart Rate Artifact Processing and Heart Rate Variability Analysis,” Behavior Research Methods, published online, 14 May 2011. HHdoi:10.3758/s13428-011-0107-7  S. SÜTTERLIN ET AL. Copyright © 2011 SciRes. JBBS 166 [23] B. Allen, A. S. Chambers and D. N. Towers, “The Many Metrics of Cardiac Chronotropy: A Pragmatic Primer and a Brief Comparison of Metrics,” Biological Psychology, Vol. 74, No. 2, 2007, pp. 243-262. HHdoi:10.1016/j.biopsycho.2006.08.005 [24] Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysi- ology, “Heart Rate Variability―Standards of Measure- ment, Physiological Interpretation, and Clinical Use,” European Heart Journal, Vol. 17, 1996, pp. 354-381. [25] G. J. M. van Boxtel, M. W. van der Molen, J. R. Jennings and C. H. M. Brunia, “A Psychophysiological Analysis of Inhibitory Motor Control in the Stop-Signal ParaDigm,” Biological Psychology, Vol. 58, No. 3, 2001, pp. 229-262. HHdoi:10.1016/S0301-0511(01)00117-X [26] S. W. Porges, J. A. Doussard-Roosevelt and A. K. Maiti, “Vagal Tone and the Physiological Regulation of Emo- tion,” In: Society for Research in Child Development, Ed., The Development of Emotion Regulation: Biological and Behavioral Considerations, Blackwell Publishing, New York, 1994, pp. 167-186. [27] B. M. Appelhans and L. J. Luecken, “Heart Rate Vari- ability as an Index of Regulated Emotional Responding,” Review of General Psychology, Vol. 10, No. 3, 2006, pp. 229-240. HHdoi:10.1037/1089-2680.10.3.229 [28] V. Napadow, R. Dhond, G. Conti, N. Makris, E. N. Brown and R. Barbieri, “Brain Correlates of Autonomic Modula- tion: Combining Heart Rate Variability with fMRI,” NeuroImage, Vol. 42, No. 1, 2008, pp. 169-177. HHdoi:10.1016/j.neuroimage.2008.04.238 [29] A. L. Hansen, B. H. Johnsen and J. F. Thayer, “Vagal Influence on Working Memory and Attention,” Interna- tional Journal of Psychophysiology, Vol. 48, No. 3, 2003, pp. 263-274. HHdoi:10.1016/S0167-8760(03)00073-4 [30] S. Sütterlin, C. Herbert, M. Schmitt, A. Kübler and C. Vögele, “Frames, Decisions, and Cardiac-Autonomic Con- trol,” Social Neuroscience, Vol. 6, No. 2, 2011, pp. 169- 177. HHdoi:10.1080/17470919.2010.495883 [31] S. Sütterlin, C. Herbert, M. Schmitt, A. Kübler and C. Vögele, “Overcoming Selfishness: Reciprocity, Inhibition, and Cardiac Autonomic Control in the Ultimatum Game,” Frontiers in Psychology, Vol. 2, No. 173, 2011, pp. 1-8. doi: 10.3389/fpsyg.2011.0017 [32] S. Funahashi, “Neuronal Mechanisms of Executive Con- trol by the Prefrontal Cortex,” Neuroscience Research, Vol. 39, No. 2, 2001, pp. 147-165. HHdoi:10.1016/S0168-0102(00)00224-8 [33] E. Harmon-Jones and J. J. B. Allen, “Behavioral Activa- tion Sensitivity and Resting Frontal EEG Asymmetry: Covariation of Putative Indicators Related to Risk of Mood Disorders,” Journal of Personality and Social Psy- chology, Vol. 74, No. 5, 1997, pp. 1310-1316. HHdoi:10.1037/0022-3514.74.5.1310 [34] J. Wacker, M. Heldmann and G. Stemmler, “Separating Emotion and Motivational Direction in Fear and Anger: Effects on Frontal Asymmetry,” Emotion, Vol. 3, No. 2, 2003, pp. 167-193. HHdoi:10.1037/1528-3542.3.2.167 [35] J. Hewig, D. Hagemann, J. Seifert, E. Naumann and D. Bartussek, “On the Selective Relation of Frontal Cortical Asymmetry and Anger-Out versus Anger-Control,” Jour- nal of Personality and Social Psychology, Vol. 87, No. 6, 2004, pp. 926-939. HHdoi:10.1037/0022-3514.87.6.926 [36] J. Hewig, D. Hagemann, J. Seifert, E. Naumann and D. Bartussek, “The Relation of Cortical Activity and BIS/ BAS on the Trait Level,” Biological Psychology, Vol. 71, No. 5, 2006, pp. 42-53. HHdoi:10.1016/j.biopsycho.2005.01.006 [37] A. F. Jorm, H. Christensen, A. S. Henderson, P. A. Jacomb, A. E. Korten and B. Rodgers, “Using the BIS/ BAS Scales to Measure Behavioural Inhibition and Be- havioural Activation: Factor Structure, Validity and Norms in a Large Community Sample,” Personality and Indivi- dual Differences, Vol. 26, No. 1, 1998, pp. 49-58. HHdoi:10.1016/S0191-8869(98)00143-3 [38] A. J. Shackman, B. W. McMenamin, J. S. Maxwell, L. L. Greischar and R. J. Davidson, “Right Dorsolateral Pre- frontal Activity and Behavioral Inhibition,” Psychologi- cal Science, Vol. 20, No. 12, 2009, pp. 1500-1506. HHdoi:10.1111/j.1467-9280.2009.02476.x |

