Journal of Minerals and Materials Characterization and Engineering
Vol.02 No.06(2014), Article ID:51459,11 pages
10.4236/jmmce.2014.26060
Clay Characterization and Optimisation of Bleaching Parameters for Palm Kernel Oil Using Alkaline Activated Clays
T. O. Salawudeen1, A. O. Arinkoola1, M. O. Jimoh1*, B. A. Akinwande2
1Chemical Engineering Department, Ladoke Akintola University of Technology, Ogbomoso, Nigeria
2Department of Food Science and Engineering, Ladoke Akintola University of Technology, Ogbomoso, Nigeria
Email: *jimonseur2013@gmail.com
Academic Editor: Jiann-Yang (Jim) Hwang, Michigan Technological University, USA
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 6 August 2014; revised 12 September 2014; accepted 14 November 2014
ABSTRACT
The optimum bleaching parameters (clay dose and bleaching time) for palm kernel oil were determined using alkaline activated clay locally sourced in Nigerian. Activation was carried out using NaOH and KOH. The raw and activated clay samples were characterized using XRD and XRF. Bleached oil was analyzed for % colour reduction (%CR) and percentage free fatty acid (FFA) content before and after bleaching. Alkaline activation resulted to total dissolution of Mg2+ and Ca2+ ions in the octahedral sheet. The activation also affected the tetrahedral sheet by reducing the % composition of the silica and alumina ion after the modification. The results showed that alkaline activation had significant effect on mineralogical and chemical composition of the clay samples. The optimum bleaching conditions (clay dosage and bleaching time) of 3%/weight of oil and 30 mins resulted in 33.3% CR and %FFA of 2.2. It was discovered that NaOH activated clay resulted to higher %CR while KOH activated clay shows affinity for reduced %FFA but at higher concentration of 5 M. This research has further established that alkaline activated clays are feasible alternative for acid activated clays in vegetable oil bleaching.
Keywords:
Bleaching, Optimisation, Clay, Characterization, Palm Kernel Oil

1. Introduction
Bleaching is an important process in vegetable oil refining process. It involves the removal of variety of impurities from vegetable oil such as phosphatides, gum, trace metals and free fatty acid. Bleaching process reduces color pigments produced by compounds such as carotene, carotenoids, xanthophylls, and chlorophyll present in the oil to more acceptable levels [1] . It is well known that clay minerals can be used to decolorize oils because of its high adsorptive capacity. In other to enhance this adsorptive property, acid activation of clay is the most commonly used technique. This area of clay modification has received enormous interest from various researchers [2] - [7] . Generally, acid activation process consists of two steps which are substitution of the exchangeable cations by protons and the dissolution of metals ions from the clay structure, by depopulating the octahedral sheet [8] . However, a lot of problems associated with bleached oil have been attributed to using acid activated clay, and such problems include residual acid effect on the bleached oil and soap formation during neutralization. Washing of the acid activated clay to reduce the residual acid is also another major challenge because it increases the cost and time of activation. Also, the cost of acid used is relatively high, hence the need for alternative method of improving clay adsorptive properties that will give us a contaminant free oil.
Akinwande et al. [9] investigated the suitability of alkaline activated clays on vegetable oil refining, a case study of Shea butter. The authors discovered that alkaline activation had significant effect on the structure, morphology and adsorptive power of the clay. The result indicated that alkaline concentration required to achieve maximum bleaching power is 0.5 M and 5 M for NaOH and KOH respectively. The percentage free fatty acid content of the Shea oil was reduced by 65% and 75% for oil bleached with NaOH activated clays and KOH activated clays respectively. Okwara and Osoka [10] studied the influence of caustic activation of Nigerian local clays on palm oil bleaching. Their findings indicated that the adsorption capacity for bleaching earth increased up to 79% at the optimum concentration of 1.0 N NaOH. Compared to the caustic treatment, acid treatment shows a much higher adsorption activity increased up to 99%. Even though there are few studies for the alkaline activation effect on the adsorption capacity of bleaching earth, it was believed that base activation is a better alternative for use in palm oil bleaching. This is because surface activation of clay can be achieved at a lesser cost compared to using the conventional inorganic acid like H2SO4 and HCl.
Many factors have been reported to influence the bleaching performance of activated clay; these include concentration of activating agent, activation temperature, bleaching temperature, bleaching time, clay dosage, moisture content, clay quality and particle size. Determination of optimum parameters for achieving highest bleaching efficiency is important to prevent formation of undesirable compounds in the final product. Roberto [11] , stated in his article “achieving optimal bleaching performance” that to achieve the most economical bleaching process, it is necessary to consider the following: type and quality of degummed and refined oil, characteristics of sorbent used, processing condition and layout of process equipment employed. The author also mentioned that efficient bleaching process must be able to remove the appropriate contaminant hence, good refined oil must be low in phosphorus, free fatty acids and soaps. Dubravka et al. [12] studied the optimization of bleaching parameter for soya bean oil. Their result showed that the amount of clay had the greatest influence on bleaching efficiency especially on transparency and phosphorous content. Temperature and clay dose significantly increased oxidative stability while increased clay dose reduced the tocopherol content of the bleached oil. The authors also stated that optimum bleaching time depended on bleaching temperature and the quality of oil. However, this optimum condition varies depending on the type of oil and clay materials, therefore this study aimed at characterization and optimization of bleaching parameters of Nigeria clay activated with alkalis using Palm Kernel Oil (PKO) as reference vegetable oil.
2. Material and Methods
Clay material was collected using hand auger to scoop the clay materials from dug pits. Naturally occurring clay sample B was obtained at a mining site located outskirt of Awo which falls within latitude 7˚45'N and 7˚47'N of the equator and longitude 4˚24'E and 4˚25'E of Greenwich meridian in Egbedore local government area of Osun state, Nigeria. This clay sample was mined at an approximate dept of 10.5 meters below the top soil. The location and accessibility map of the sampling location is shown in Figure 1.
This clay is off white in colour and gave high plasticity when mixed with water. Crude palm kernel oil was obtained at a local refinery at Aroje, Ogbomoso, Oyo state, Nigeria. Reagents used such as sodium hydroxide, potassium hydroxide, ethanol, n-hexane were analytical grade, products of Mayer and Baker, Bagenham, England.
Figure 1. Location and accessibility map of the study area.
3. Experimental
3.1. Clay Characterization
The mineralogical compositions of the raw and activated samples were determined using X-ray diffraction (Empyrean diffractometer DY 674 Panalytical, Holland). The measurement were made using CuKα radiation = 1.5418 Å, tube current was 40 mA and the tension was 45 VA. The chemical composition was determined using X-ray fluorescence.
3.2. Clay Activation
The clay sample was wet sieved to remove impurities. The sieved clay (100 g) was dispersed in 60 ml of distilled water to form slurry. The slurry was pre heated to 60˚C for 5minutes. 100 ml of alkaline, 0.5 M NaOH and 5 M KOH was added to the pre heated slurry and the mixture heated to 90˚C for 80 mins [9] . The slurry formed was dried conventionally in a drier to reduce the moisture content. Drying was done at a temperature of 70˚C for 12 hours [13] . The dried clay was grinded to the appropriate size for use as bleaching clay. The activated clay was grinded so that at least 98% of the resulting products are less than 100 microns (160 mesh) in size.
3.3. Degumming
500 ml of crude palm kernel oil was measured into a 2000 ml beaker and 1000 ml of boiled water was added to it. The hot water-oil was continuosly stirred for 10 minutes. The gums and water was separated from the oil while still hot using a separating funnel. The process was repeated thrice to ensure thorough removal of all hydratable gums.
3.4. Bleaching of Palm Kernel Oil
100 g of Palm kernel oil was weighed in to a round bottom flask and heated to a temperature of about 50˚C. A known weight of activated clay was added to the preheated oil under continuous stirring in a rotary evaporator (RE-52A, Biochemistry instrument factory, Shanghai). The oil-clay mixture was gradually heated to a bleaching temperature of 95˚C for specific time (according to the design of experiment). After the bleaching process, the oil treatment, the sludge was filtered off through a filter paper (Whatman No. 1) under vacuum using suction apparatus (Model YX932D) with filtration set. The process was carried out for both NaOH and KOH activated clay.
3.5. Determination of Physicochemical Properties of Bleached Oil
3.5.1. Acid Value
The acid value is the number of mg of potassium hydroxide required to neutralize the free acid in 1 g of the substance. Acid value and free fatty acid content of the oil before and after bleaching was determined using titrimetric method [14] . After the experimental procedure, the acid value was calculated using the relation:
Acid value = 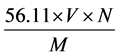 (1)
(1)
where, V is the volume of Potassium hydroxide used in ml;
N is the exact normality;
M is the mass in g of oil sample.
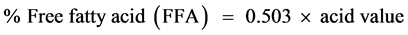 (2)
(2)
3.5.2. Percentage Colour Reduction
The percentage colour reduction of the oil after bleaching was determined using colorimeter (Uniscope, SM- 3004). A full absorption spectrum was made over all the wave lengths, between 420 nm and 670 nm. The absorbance of the samples was determined at the maximum wavelength (530 nm) using hexane as reference. The samples were diluted in n-hexane in the proportion 0.1 g of oil to 2 ml of solvent. The bleaching capacity of the adsorbent was determined using the following relation:
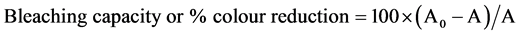 (3)
(3)
where A0 and A are absorbance of crude and bleached oil respectively.
3.6. Optimization
Optimization was carried out using CCD of Surface Response Methodology with two factors (clay dose and the bleaching time) and two responses (%FFA and %CR). Statistical analysis was carried out to determine correlation coefficients of the model as a function of the responses. The statistical summary of the design is shown in Table 1 and the Experimental Design is shown in Table 2.
4. Results and Discussion
4.1. Mineralogical Composition of Clay Sample Using X-Ray Diffraction Technique
The effect of alkaline activation on the mineralogical composition of the clay sample was studied using the XRD. The result of the raw sample, Figure 2 showed that illite (potassium aluminium silicate hydroxide hydrate) is the dominant mineral in the sample as indicated by the peak at an intensity count above 800 in the XRD plot. Other minerals present include Kaolinite (aluminum silicate hydrate) and rutile (titanium oxide). Illite and Kaolinite are clay minerals while rutile is an ore present in the clay sample.
Figure 3 and Figure 4 showed the XRD patterns of alkali activated clay samples. It was observed that there were no changes in the mineral composition of the clay samples after activation as shown in the Figures but the
Table 1. Statistical summary of experimental design (Bleaching).
Table 2. Design of experiment for oil bleaching.
Figure 2. XRD pattern of raw clay sample.
Figure 3. XRD pattern of sample B activated with 0.5 M Sodium hydroxide.
Figure 4. XRD pattern of sample B activated with 5 M potassium hydroxide.
intensity or counts changes from 800 as in the raw sample to 1000 and 400 for clay activated with 0.5 M NaOH and 5 M KOH respectively. This is an indication that the percentage composition of the constituting minerals changed with the activation and this change is expected to have a positive impact on the adsorption efficiency of the activated clay.
4.2. Chemical Composition of Clay Samples Using X-Ray Fluorescence (XRF)
The chemical analysis of the raw and activated samples presented in mass % in Table 3 showed that the major oxides present in the clay samples were SiO2, Al2O3, Na2O, K2O, MgO, CaO, TiO2, MnO, Fe2O3 and P2O5. The XRF result showed that some interlayer cations were removed as a result of the activation indicating that more active sites are generated on the clay surface.
The dominant oxides in the clay samples are oxides of silica, alumina and potassium. Comparing the activated samples with raw sample, the percentage composition of silica oxide decreased from 52.4% to 47.3% and 43.9% after activation with 0.5 M NaOH and 5 M KOH respectively. Alumina content increased after NaOH activation (from 34.2% to 36.5%) and dropped (from 34.2% to 29.2%) after KOH activation. This variation explains the extent of isomorphic substitution in the activated samples when two different alkaline were used. The lower percentage composition of silica oxide in the potassium hydroxide activated clay (KOHAC) showed that isomorphic substitution of Al3+ for Si4+ was higher than in sodium hydroxide activated clay (NaOHAC). This implied that more negative charge will be produced in the crystal of KOHAC than that of NaOHAC hence, the higher adsorption property exhibited by KOHAC.
The appearance of TiO2 is due to the presence of impurities such as rutile in the clay sample. The exchangeable cations in the octahedral sheet such as Ca2+ and Mg2+ present in the clay sample were completely dissolved after action with 5 M KOH. These divalent exchangeable cations may be said to have been replaced by monovalent hydrogen ion during the activation process. Part of Mg2+ and Fe2+/3+ were removed when activation was carried out with 0.5 M NaOH. Phosphorus oxide was not detected in the raw sample but was discovered in trace after activation with both alkaline. This substitution produced more negative charge on the surface of the clay and these negative charges attract any positive charges in order to balance up and hence, in the process improved adsorption efficiency.
4.3. Optimization of Bleaching Process Parameters
Table 4 shows the physiochemical properties of oil bleached with activated clays. The bleaching process resulted in pH and % free fatty acid reduction. More pigment was removed with NaOH activated clay (22% CR) than KOH activated clay (18.5% CR). However, free fatty acid removal was favoured most by KOH activated clay. There was a slight decrease in the density values, this decrease may be attributed to the removal of pigments in the oil which subsequently affect its weight.
Table 3. Chemical composition of raw and activated clay samples.
ND = Not Detected.
Table 4. Physicochemical properties of PKO bleached with activated clays.
AC = Alkaline Concentration. AT = Activation Time. %CR = Percentage colour reduction. %FFA = Percentage free fatty acid.
Optimization of the bleaching parameters was done using the NaOHAC (sample B). NaOHAC was chosen because a relatively low concentration was required for the activation hence, lower cost of activation compared to KOHAC. An experiment was designed varying the clay dosage in grams and the bleaching time minutes. The result of the oil bleached with NaOHAC-sample B is presented in Table 5. %CR and FFA were used as responses to test the effect of the factors (clay dose and contact time) on the bleached oil.
4.3.1. Statistical Analyses
Table 6 and Figure 7 shows the analysis of variances of the fitted quadratic models for percentage colour reduction and percentage free fatty acid respectively for oil bleached with sodium hydroxide activated clays for sample B. The models were chosen because of their high correlation coefficient (actual and adjusted) value as shown in Table 8. A and B are clay dose and contact time respectively.
4.3.2. Test for Significance and Accuracy of the Model
The levels of significance of the models were tested using the p-value. As shown in Table 7 and Table 8 p- value less than 0.05 indicate the model terms are significant and p-values greater than 0.1 indicate model terms are not significant (Expert, 2002). From both tables, it can be seen that all the model terms A, B, A2 and B2 are significant. Both the linear and quadratic effect of the factors is significant. A and B represents clay dosage and bleaching time respectively. It can be inferred from this test that varying the clay dose and bleaching time will have effect on the physicochemical properties of the bleached oil. The significance of the model was tested
Table 5. Experimental result for oil bleached with Sample B (NaOH activated clay).
Table 6. Analysis of variance for response surface quadratic model (%CR)―Sample B.
P < 0.05 indicates the model terms are significant.
Table 7. Analysis of variance for response surface quadratic model (%FFA)―Sample B.
P < 0.05 indicates the model terms are significant.
Table 8. Measurement of significance using R2 and adequate precision.
further by plotting the actual experimental values against the model prediction values as shown in Figure 5. It can also be observed from the plots that some points lie on the 45˚ line while others are not too far from it showing that the model predictive.
4.3.3. Effect of Individual Parameters on Responses
Figure 6(a), Figure 6(b), Figure 7(a) and Figure 7(b) showed the interaction of individual factor (clay dosage
Figure 5. Model prediction values and actual experimental values plot (a) %CR for NaOHAC (b) %FFA for NaOHAC.
Figure 6. Interaction plots for NaOHAC (a) one factor plot of clay dosage and colour reduction (b) one factor plot of contact time and colour reduction (c) combined interaction plot of clay dosage and contact time on colour reduction.
and bleaching time) on percentage colour reduction and percentage free fatty acid respectively. Figure 6(c) and Figure 7(c) presents the interaction of the two factors on the responses (%CR and %FFA). From Figure 6a, %CR of oil increases with increased clay dosage. Similarly, %CR increases with increased bleaching time as shown in Figure 6(b). The %CR increases with increase in both factors as shown in Figure 6(c). %FFA decreases with increased clay dosage and contact time (Figures 7(a)-(c)), however, the decrease was more with increased clay dose. This result is in line with the work of Durbrvka et al. (2012) which stated that high clay content enhances the removal of impurities in oil.
The equations generated from the fitted surface response quadratic models for the bleaching operation are shown in Equations (4) and (5). These equations are mathematical representation of the experimental results and can be use to determine the bleaching parameters required to achieve a particular physicochemical property of oil prior the experiment.


4.4. Optimizations Result
The bleaching parameters were optimized subject to constraint shown in Table 9. The objective is to maximize percentage colour reduction and minimize percentage free fatty acid content of the oil. 8 solutions were found and the solution with the highest desirability was chosen. In other to verify the optimization results, the selected parameters were validated in the laboratory with suggested values as shown in Table 10 and Table 11. The result from the experiments confirmed the selected solutions.
Figure 7. Interaction plots for NaOHAC (a) one factor plot of clay dosage and FFA (b) one factor plot of contact time and FFA (c) combined interaction plot of clay dosage and contact time on FFA.
Table 9. Optimization constraint for the bleaching optimization.
Table 10. Optimization solution for bleaching parameters (Sample B).
Table 11. Validation result for NaOH activated clay (Sample B).
CD = Clay dosage; CT = Contact time.
5. Conclusion
Investigation shows that alkaline activation resulted in significant modification of the clay structure. The octahedral sheet cations (Ca2+ and Mg2+) were completely dissolved with 5 M KOH. NaOHAC and KOHAC were both effective for the adsorption bleaching of PKO. However, lower concentration required to achieve optimum bleaching efficiency gave NaOHAC an advantage over KOHAC. Also, more soap was formed when KOHAC was used for the adsorption bleaching which affected the filterability of the oil-clay mixture. Optimization of the bleaching parameters (clay dosage and bleaching time) shows that bleaching efficiency increases with increased clay dosage and longer activation time. The optimum conditions are 3 w/w clay dose and 30 mins contact time. However, more oil may be retained if filtration was not carried out under high pressure.
References
- Wu, Z.S. and Li, C. (2009) Kinetics and Thermodynamics of β-Carotene and Chlorophyll Adsorption onto Acid- Activated Bentonite from Xinjiang in Xylene Solution. Journal of Hazardous Materials, 171, 582-587. http://dx.doi.org/10.1016/j.jhazmat.2009.06.047
- Kashani Motlagh, M.M., Youzbashi, A.A. and Amiri Rigi, Z. (2011) Effect of Acid Activation on Structural and Bleaching Properties of a Bentonite. Iranian Journal of Materials Science & Engineering, 8, 50-56.
- Christidis, G.E., Scott, P.W. and Dunham, A.C. (1997) Acid Activation and Bleaching Capacity of Bentonites from the Islands of Milos and Chios, Aegean, Greece. Applied Clay Science, 12, 329-347. http://dx.doi.org/10.1016/S0169-1317(97)00017-3
- Fransisco, R.V. and de Souza Santos, P. (2001) Studies on the Acid Activation of Brazilian Smectite Clay. University of San Paulo, San Paulo.
- Suarez Barrios, M., Flores González, L.V., Vicente Rodríguez, M.A. and Martin Pozas, J.M. (1995) Acid Activation of a Palygorskite with HCl: Development of Physico-Chemical, Textural and Surface Properties. Applied Clay Science, 10, 247-258. http://dx.doi.org/10.1016/0169-1317(95)00007-Q
- Novakovic, T., Rožić, L., Petrovic, S. and Rozic, A. (2008) Synthesis and Characterization of Acid Activated Serbian Smectite Clays Obtained by Statistically Designed Experiments. Chemical Engineering Journal, 137, 436-442. http://dx.doi.org/10.1016/j.cej.2007.06.003
- David, H., Christian, P.F. and Jorge, B. (2002) Processes for Producing Bleaching Clay Product. United States Patent; US6759359BI.
- Fernandes, C., Catrinescu, C., Castilho, P., Russo, P.A., Carrott, M.R. and Breen, C. (2007) Catalytic Conversion of Limonene over Acid Activated Serra de Dentro (SD) Bentonite. Applied Catalysis A: General, 318, 108-120. http://dx.doi.org/10.1016/j.apcata.2006.10.048
- Akinwande, B.A., Salawudeen, T.O., Arinkoola, A.O. and Jimoh, M.O. (2014) A Suitability Assessment of Alkali Activated Clay for Application in Vegetable Oil Refining. International Journal of Engineering and Advanced Technology Studies, 2, 1-12.
- Okwara, C.A. and Osoka, E.C. (2006) Caustic Activation of Local Clays for Palm Oil Bleaching. Journal of Engineering and Applied Sciences, 1, 526-529.
- Roberto, R. (2006) Achieving Optimal Bleaching Performance. Oil Mill Gazetter, 112, 2-6.
- Dubravka, S., Tomislav, D., Klara, K., Jasenka, G.K., Sandra, N. and Marko, O. (2012) Soybean Oil Bleaching Parameters. Food Technology and Biotechnology, 50, 199-207.
- Rohdenburg, H., Csernitzky, K., Chikany, B., Peredi, J., Borodi, A. and Fabicsne Ruzics, A. (1999) Degumming Process for Plant Oils. US Patent 5,239,096.
- ISO (2009) Animal and Vegetable Fats and Oil: Determination of Acid Value and Acidity. ISO Method 660:2009, Geneva.
NOTES
*Corresponding author.










