Paper Menu >>
Journal Menu >>
 Advances in Microbiology, 2012, 2, 310-316 http://dx.doi.org/10.4236/aim.2012.23037 Published Online September 2012 (http://www.SciRP.org/journal/aim) Saffron (Crocus sativus L.) Inhibits Aflatoxin B1 Production by Aspergillus parasiticus Cryssa Tzanidi, Charalampos Proestos, Panagiota Markaki* Department of Chemistry, Laboratory of Food Chemistry, National and Kapodistrian University of Athens, Athens, Greece Email: *markaki@chem.uoa.gr Received May 2, 2012; revised June 1, 2012; accepted June 14, 2012 ABSTRACT Aflatoxin B1 (AFB1) is a carcinogenic metabolite produced by certain Aspergillus species. The aim of the present study was to investigate the effect of saffron stigmas on A. parasiticus growth and AFB1 production in Yeast Extract Sucrose (YES) medium. AFB1 was extracted from cultures and purified with immunoaffinity columns followed by high performance liquid chromatography (HPLC) coupled to fluorescence detector (FL) analysis. Methods’ recovery and limit of detection were 95.3% and 0.02 ng AFB1·ml–1 of YES respectively. Results indicated that AFB1 produc- tion in samples of YES inoculated with A. parasiticus after the addition of saffron dried stigmas (100 mg·flask–1) was significantly lower (p < 0.05) compared to control samples (inoculated without the addition of saffron stigmas) throughout the entire incubation period (18 days), at the same time as mycelial growth was noticeable. In addition, mycelial growth was observed and AFB1 was detected, after the 7th day of observation in cultures with saffron alone. Maximum production was observed on the 12th day (0.018 μg AFB1·flask–1) and on the 9th day (0.051 μg AFB1·flask–1) for samples of YES with the addition of saffron inoculated with A. parasiticus and samples with saffron alone (non-inoculated), respectively. In control samples (inoculated without saffron) the maximum production on the same days 12 and 9 was 75.31 μg AFB1·flask–1 and 64 μg AFB1·flask–1 respectively. Conclusively when saffron was added to YES inoculated with A. parasiticus, AFB1 production decreased by 99.9% compared to control cultures without saffron addition. This inhibition can be attributed to the antioxidant capacity of saffron con- stituents. Keywords: Aflatoxin B1; A. parasiticus; HPLC-FL Analysis; Saffron; Antioxidants 1. Introduction Aflatoxins are mycotoxins of great significance in foods and feeds and are mainly produced by Aspergillus (A) flavus, A. parasiticus and A. nomius. Aflatoxin B1 (AFB1) is usually found at highest concentrations in contami- nated food and feed. Additionally it is regarded to be genotoxic and the most potent liver carcinogen for many animal species as well humans [1,2]. Saffron consists of dried stigmas of the herb Crocus sativus L. belonging to the Iridaceae family. It is culti- vated not only in Mediterranean countries such as Greece, Spain and France but also in India and Iran [3]. There is limited knowledge about the physiology of vegetative development of saffron. It has been shown that the corm size, water availability and cultivation conditions have a significant effect on vegetative development of saffron. The biomass contribution of leaves and mother corm for maintaining the vegetative development of saffron varies throughout the growing season. Photosynthetic rate is consistently very high throughout the year but is reduced in the largest corms [4]. In addition saffron is world’s most expensive spice and several studies indicate its po- tential as anticancer, anti-inflammatory and hypolipi- daemic agent [5-9]. Asdaq and Inamdar [10] investigated the potential of saffron and its constituent, crocin, as hypolipidemic and antioxidant agent in rats and reported that saffron was found to be superior to crocin indicating the involvement of other potential constituents of saffron apart from crocin for its synergistic behaviour of quench- ing free radicals. Moreover according to Kumar et al. [11] saffron is known to have antioxidant-like properties. Furthermore, extracts of several spices and herbs have been shown to reduce A. parasiticus growth and AFB1 production [12]. Moreover, essential oils have been con- sidered to inhibit A. flavus growth and AFB1 production [13,14]. Pawar and Thaker [15] investigated 75 essential oils against the fungus A. niger growth and saffron was found to have no effect on the fungus. According to Igawa et al. [16], in addition to production of inhibitors, the development of plants that can inhibit mycotoxins is a promising approach. *Corresponding author. C opyright © 2012 SciRes. AiM  C. TZANIDI ET AL. 311 Saffron contains chemical constituents such as cro- cetin, picrocrocin and safranal which are responsible for color, flavor and aroma respectively. Anthocyanins, fla- vonoids, vitamins, amino acids, proteins, starch, minerals and other chemical compounds have also been described in saffron [15]. It is important to note that under basic conditions and following the drying process, picrocrocin is converted to safranal [17]. Additionally crocetin ester is degraded upon thermal treatment, although it was af- fected by external factors [18]. Giaccio [3] reported that crocetin protects against oxidation damage in rats’ pri- mary hepatocytes, in particular a suppression of AFB1- induced hepatotoxic lesions by crocetin. The reduction of toxicity is due to the property of crocetin to stimulate defense mechanisms in the liver cells with the increase of the glutathion-S-transferase and also with the decrease of the alleged AFB1-DNA. To our knowledge there is little information in the lit- erature concerning the saffron biological activity as crude product and its effect on AFB1 production. Hence the aim of the present study was to investigate the effect of saf- fron dried stigmas on A. parasiticus growth and AFB1 production in Yeast Extract Sucrose medium. 2. Materials and Methods 2.1. Apparatus and Reagents A laminar flow (Telstar Bio IIA, Spain), an incubator WTB Binder (Tuttlinger, Germany), and a centrifuge Sorvall RC-5B (HS-4) (Norwalk, USA) were used. AFB1 standard was purchased from Sigma-Aldrich Chemical Co, USA. Millipore filters HVLP (0.45 μm) were pur- chased from Waters (Millipore, USA). The Aflatest im- munoaffinity columns were obtained from Vicam, USA. All reagents used were of analytical grade (Sigma Al- drich, USA) while HPLC solvents were of HPLC grade and were purchased from Fisher Scientific, UK. Trifluo- roacetic acid was from Fluka, The Netherlands. 2.2. Samples Samples of saffron were collected from the Athens mar- ket during 2010. Saffron purchased in packages was used before the expiration date. The packages were little pots made of glass labeled as “Cooperative de Saffran Ko- zani”. The content of each pot was 1 g of saffron stigmas. All samples were stored in dark and cool place prior to analysis. Just before examination saffron stigmas were transferred in sterile polyethylene bags. Representative sub samples of saffron stigmas (100 mg) were collected aseptically and also used in the present study. 2.3. Media Aspergillus flavus parasiticus agar (AFPA) was prepared by dissolving 4 g of yeast extract (Oxoid) (Hamshire, UK), 2 g of bacteriological peptone (Oxoid) 0.1 g of fer- ric ammonium citrate, 0.2 ml of Dichloran 0.2% in etha- nol (Fluka Steinheim, The Netherlands), 0.02 g of chlo- ramphenicol (Oxoid) and 3 g of agar (Oxoid) in 200 ml of distilled water, final pH 6.0 - 6.5. Czapek Dox agar (CZA) was prepared by dissolving 0.4 g of sodium nitrite, 0.1 g of potassium chloride, 0.1 g of magnesium sulfate, 0.002 g of ferric sulfate, 0.2 g of dipotassium phosphate, 6 g of sucrose, 3 g of agar, 0.002 g of zinc sulfate and 0.001 g of copper sulfate in 200 ml of distilled water, final pH 6.0 - 6.5 [19]. 2.4. Preparation of Spore Inoculum The aflatoxigenic strain A.parasiticus spear (IMI 283883) utilized throughout this study was obtained from the In- ternational Mycological Institute (Engham Surrey, UK). An inoculum was obtained by growing the mold on a slant of stock cultures of CZA, which were maintained at 5˚C. Spore inoculum was prepared by growing A. para- siticus on CZA for 7 days at 30˚C and spores were har- vested aseptically using 10 mL of sterile 0.01% v/v Tween 80 solution. AFB1 carried over from the initial growth was minimized by centrifuging the spore sus- pension (1000 g for 10 min) and resuspending the bio- mass in 10 mL of sterile Tween 80 solution twice. Dilu- tions (0.1, 0.01, 0.001 and 0.0001) were prepared from the initial spore in sterile tubes containing 10 mL of Tween 80 0.05% v/v suspension [19]. The spore concen- tration was determined by the spread plate surface count technique, using 0.1 mL of each dilution on four AFPA plates after incubation at 30˚C for 2 days. The population size was estimated by the reverse intense yellow/orange coloration of the colonies. For obtaining an inoculum containing 100 conidia, plates with 10 - 100 colony form- ing units (cfu) were selected and the desired 100 spore quantity used in the present study was estimated.The quantity of 100 spores flask-1 was chose as it was the minimum concentration found in the literature producing a detectable amount of AFB1 by aspergilli [20]. 2.5. Inoculation For each day of observation, 6 flasks containing 10 mL of YES medium were inoculated with 100 conidia·flask–1 of A. parasiticus. 100 mg of saffron stigmas with natural microbiota (NM), were added before inoculation into each of the three flasks for each day of observation. Fur- thermore, 100 mg of saffron with NM were added to three additional flasks of non-inoculated YES, for each day of observation. We should mention that all flasks with YES were sterilized by autoclaving at 121˚C for 15 min. Saffron was added after sterilization and before in- oculation. All flasks were incubated under stationary Copyright © 2012 SciRes. AiM  C. TZANIDI ET AL. 312 conditions at 30˚C. Immediately after autoclaving (115˚C, 30 min) for safety reasons [21] the mycelial growth was determined and AFB1 was assayed on days 0, 3, 7, 9, 12, 15 and 18 of incubation. The experiments were repeated in triplicate. 2.6. AFB1 Determination The content of each flask containing YES medium with saffron and flasks control (without saffron addition) was mixed with 30 mL of methanol and shaken well for 10 min. After filtration, an aliquot of 1 ml from each flask was used for AFB1 analysis. The aliquot of 1 ml from the filtrate was mixed with 10 ml of distilled water. The mixture was transferred onto an Aflatest immunoaffinity column with flow rate 6 ml·min–1 and washed twice with 10 ml of distilled water. The column was then allowed once more to dry by passing air through it. AFB1 was eluted with 2 ml of acetonitrile (flow rate 0.3 ml·min–1) [22]. A derivative of AFB1 (AFB2a, hemiacetal of AFB1) was prepared by adding 200 μl of hexane and 200 μl of trifluoroacetic acid to the evaporated solution of AFB1 eluate, heating at 40˚C in a water bath for 10 min, evaporating to dryness under nitrogen, redissolving in 200 - 500 μl in appropriate volume with water/acetoni- trile 9:1, v/v to give concentration of <5 ng·ml–1 and analyzing by HPLC (volume injected = 40 μl). AFB2a shows enhanced fluorescence compared to AFB1 [23]. On the other hand AFB2a is less toxic compared to AFB1 because of its protein binding properties, since it is not being absorbed from the gastrointestinal tract and there- fore it is non toxic to experimental animals. Moreover AFB2a does not interact with nuclear DNA [24]. 2.7. Determination of Mycelial Mass in YES Medium After cooling, mycelia were filtered through filters that were previously dried (24 h at 80˚C) and weighed. The mycelium was washed with distilled water and allowed to dry for 24 h at 80˚C. The dry weight of the mycelium was then measured. 2.8. HPLC-FL Analysis HPLC was performed using a Hewlett-Packard 1050 (Waldbornn, Germany) liquid chromatograph equipped with a JASCO FP-920 (Jasco Ltd, JAPAN) fluorescence detector and an HP integrator 3395. The HPLC column used was a C18 Nova-Pak (250 × 4.6 mm, 4 μm particle size) purchased from Waters (Millipore, USA). For AFB1 determination isocratic elution was employed and mobile phase consisted of water/acetonitrile/methanol, 20:4:3, v/v/v. Prior to injection samples were filtered through Millipore HVLP (0.45 μm) filters. Detection of the AFB1 hemiacetal derivative (AFB2a) was carried out at λex = 365 nm and λem = 425 nm. The flow rate was 1 ml·min–1, and the retention time for AFB2a was ~15 min. 3. Results and Discussion Effect of Saffron on Mycelial Growth and AFB1 Production The in-house characterization of the method for deter- mining AFB1 in YES medium has been reported in detail by Vergopoulou et al. [19]. The recovery factor of the method is 95.3% (RSD 9.6%) and the detection limit of the derivatized AFB1 (AFB2α) was found to be 0.02 ng AFB1 ml–1 of YES. In the present study YES medium was chosen since is an optimum medium for AFB1 bio- synthesis. Gqaleni et al. [25] reported that in YES liquid static culture at 30˚C, AFB1 production by A. flavus was higher compared to agar media. In addition, measuring AFB1 rather than all four AFBs (AFB1, AFG1, AFB2, and AFG2) was followed throughout this study. AFB1 is the prevalent form and also the most potent of these toxins [26]. A. parasiticus was used as most A. parasiticus strains are rather stable in aflatoxin production in cul- tures [27]. Table 1 shows that the mycelial growth was observed in control cultures inoculated (without saffron addition) and in cultures with saffron, inoculated and non- inoculated with A. parasiticus, as well. It is obvious that molds occur along with other microorganisms in saffron natural microbiota. The natural microbiota of saffron stigmas examined, consisted of bacteria (<1000 cfu/gr) and potentially aflatoxigenic molds (authors unpublished results). In that case mycelium growth is likely. Palumbo et al. [28] reported that the growth of A. flavus was com- pletely inhibited when antagonist bacteria were present in microbiological media. In the present study microbial competition in cultures between saffron natural microbi- ota and A. parasiticus was not proved since the mycelial growth was just slightly reduced compared to cultures control (only inoculated with the fungus, Table 1). The addition of saffron stigmas to cultures inoculated with A. parasiticus in this study, inhibited significantly AFB1 production compared to control cultures (inocu- lated with A. parasiticus without the addition of saffron (Table 2, Figure 1). AFB1 production in cultures inocu- lated with A. parasiticus with the addition of saffron was observed up to the 15th day of incubation, while on the 18th day AFB1 was not detectable. Moreover AFB1 pro- duction was also observed at lower levels after the 7th day of incubation in non- inoculated cultures containing saf- fron (100 mg· f la s k –1). AFB1 in cultures only with saffron was not detectable the days 0 and 3 of incubation, indi- cating that the saffron utilized throughout this work was not originally contaminated with AFB1 at detectable lev- els. Previously Martins et al. [29] reported that AFB1 was Copyright © 2012 SciRes. AiM 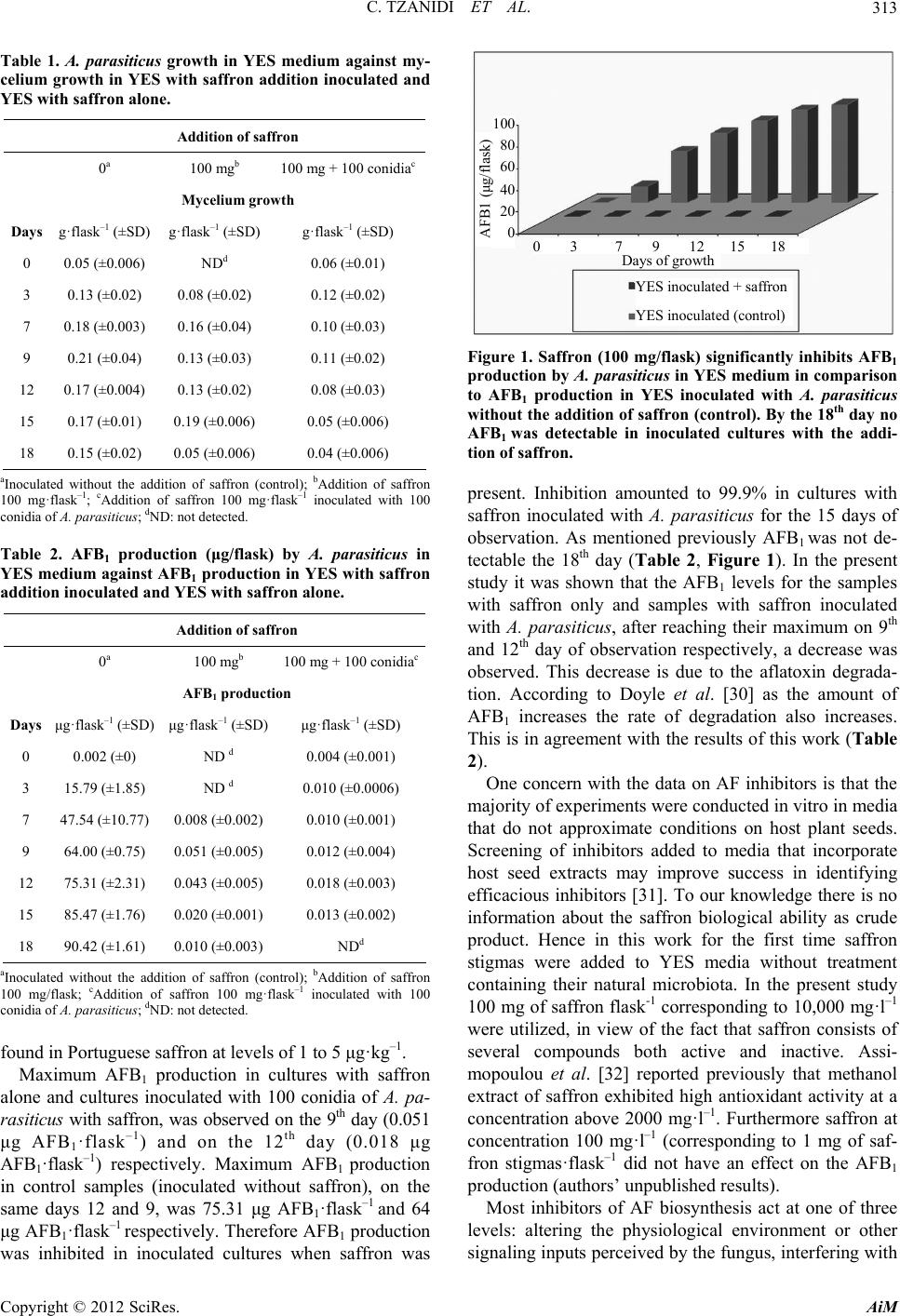 C. TZANIDI ET AL. 313 Table 1. A. parasiticus growth in YES medium against my- celium growth in YES with saffron addition inoculated and YES with saffron alone. 100 80 60 40 20 0 Addition of saffron 0 a 100 mgb 100 mg + 100 conidiac Mycelium growth Days g·flask–1 (±SD) g·flask–1 (±SD) g·flask–1 (±SD) 0 0.05 (±0.006) NDd 0.06 (±0.01) 3 0.13 (±0.02) 0.08 (±0.02) 0.12 (±0.02) 7 0.18 (±0.003) 0.16 (±0.04) 0.10 (±0.03) 9 0.21 (±0.04) 0.13 (±0.03) 0.11 (±0.02) 12 0.17 (±0.004) 0.13 (±0.02) 0.08 (±0.03) 15 0.17 (±0.01) 0.19 (±0.006) 0.05 (±0.006) 18 0.15 (±0.02) 0.05 (±0.006) 0.04 (±0.006) AFB1 (μg/flask) Days of growth 0 3 7 9 12 15 18 YES inoculated + saffron YES inoculated (control) aInoculated without the addition of saffron (control); bAddition of saffron 100 mg·flask–1; cAddition of saffron 100 mg·flask–1 inoculated with 100 conidia of A. parasiticus; dND: not detected. Table 2. AFB1 production (μg/flask) by A. parasiticus in YES medium against AFB1 production in YES with saffron addition inoculated and YES with saffron alone. Addition of saffron 0 a 100 mgb 100 mg + 100 conidiac AFB1 production Days μg·flask–1 (±SD) μg·flask–1 (±SD) μg·flask–1 (±SD) 0 0.002 (±0) ND d 0.004 (±0.001) 3 15.79 (±1.85) ND d 0.010 (±0.0006) 7 47.54 (±10.77) 0.008 (±0.002) 0.010 (±0.001) 9 64.00 (±0.75) 0.051 (±0.005) 0.012 (±0.004) 12 75.31 (±2.31) 0.043 (±0.005) 0.018 (±0.003) 15 85.47 (±1.76) 0.020 (±0.001) 0.013 (±0.002) 18 90.42 (±1.61) 0.010 (±0.003) NDd aInoculated without the addition of saffron (control); bAddition of saffron 100 mg/flask; cAddition of saffron 100 mg·flask–1 inoculated with 100 conidia of A. parasiticus; dND: not detected. found in Portuguese saffron at levels of 1 to 5 μg·kg–1. Maximum AFB1 production in cultures with saffron alone and cultures inoculated with 100 conidia of A. pa- ra s iticu s with saffron, was observed on the 9th day (0.051 μg AFB1·flask–1) and on the 12th day (0.018 μg AFB1·flask–1) respectively. Maximum AFB1 production in control samples (inoculated without saffron), on the same days 12 and 9, was 75.31 μg AFB1·flask–1 and 64 μg AFB1·flask–1 respectively. Therefore AFB1 production was inhibited in inoculated cultures when saffron was Figure 1. Saffron (100 mg/flask) significantly inhibits AFB1 production by A. parasiticus in YES medium in comparison to AFB1 production in YES inoculated with A. parasiticus without the addition of saffron (control). By the 18th day no AFB1 was detectable in inoculated cultures with the addi- tion of saffron. present. Inhibition amounted to 99.9% in cultures with saffron inoculated with A. parasiticus for the 15 days of observation. As mentioned previously AFB1 was not de- tectable the 18th day (Table 2, Figure 1). In the present study it was shown that the AFB1 levels for the samples with saffron only and samples with saffron inoculated with A. parasiticus, after reaching their maximum on 9th and 12th day of observation respectively, a decrease was observed. This decrease is due to the aflatoxin degrada- tion. According to Doyle et al. [30] as the amount of AFB1 increases the rate of degradation also increases. This is in agreement with the results of this work (Table 2). One concern with the data on AF inhibitors is that the majority of experiments were conducted in vitro in media that do not approximate conditions on host plant seeds. Screening of inhibitors added to media that incorporate host seed extracts may improve success in identifying efficacious inhibitors [31]. To our knowledge there is no information about the saffron biological ability as crude product. Hence in this work for the first time saffron stigmas were added to YES media without treatment containing their natural microbiota. In the present study 100 mg of saffron flask-1 corresponding to 10,000 mg·l–1 were utilized, in view of the fact that saffron consists of several compounds both active and inactive. Assi- mopoulou et al. [32] reported previously that methanol extract of saffron exhibited high antioxidant activity at a concentration above 2000 mg·l–1. Furthermore saffron at concentration 100 mg·l–1 (corresponding to 1 mg of saf- fron stigmas·flask–1 did not have an effect on the AFB1 production (authors’ unpublished results). Most inhibitors of AF biosynthesis act at one of three levels: altering the physiological environment or other signaling inputs perceived by the fungus, interfering with Copyright © 2012 SciRes. AiM  C. TZANIDI ET AL. 314 signal transduction and gene expression regulatory net- works upstream of AF biosynthesis, or blocking enzy- matic activity [31]. Previously, Zaica and Buchanan [33] reported that a large number of compounds were found to inhibit AFB1 biosynthesis mainly through their effect on fungal growth. In the current study on the 9th and 12th days of maximum production of AFB1 in samples with saffron alone and samples with saffron inoculated with A. parasiticus re- spectively was 1254 - 1751 and 4183 - 5333 folds lower compared with samples control (inoculated with fungus). Nevertheless the mycelial mass of the same samples at the same days was only 1.3 - 1.6 and 1.9 - 2.1 folds lower, compared to samples inoculated with A. parasiticus (control). Therefore AFB1 decrease was proved and it is assumed that is not correlated to fungus growth. Re- cently López and Gómez-Gómez [34] reported that in- fection of plants by pathogens activates a complex sys- tem of signal transduction pathways, which results in the activation of defense genes. So the authors isolated and characterized a new chitinase gene from saffron design- nated Safchi A, which showed chitinase activity in vitro. Chitin is a major component of fungal walls. Upon pathogen attack plants produce chitinases that degrade chitin to chito-oligomers which were shown to elicit strong defense responses in many plant species [35]. The results reported by López and Gómez-Gómez [34] indicated that saffron develops an active defense response to prevent colonization by the fungus. This statement is in agree- ment with the results presented in the current study since A. parsiticus growth was slightly inhibited in the pres- ence of saffron (Table 1). The most interesting approach to the study of the in- hibitory effect of saffron on A. parasiticus growth and AFB1 production is that saffron acts as an antioxidant [36]. Oxidative stress has been shown to stimulate afla- toxin biosynthesis in A. parasiticus [37,38]. The stimula- tion and suppression of AF biosynthesis by oxidants and antioxidants, respectively indicates that perturbations in the oxidative state of the fungal cell influence AF bio- synthesis. Redox reactions are fundamental to cellular catabolism and anabolism, and antioxidants may hinder vital processes. Inhibition of mitochondrial or perox- isomal b-oxidation by antioxidants may limit the avail- ability of carbon skeletons for polyketide pathways dur- ing growth on lipid-rich seeds. Antioxidants could inter- fere, thereby, reducing the pool of nicotinamide adenine dinucleotide phosphate (reduced form) available for AF biosynthetic reactions [39]. Furthermore a survey of in- hibitors of AF biosynthesis has illustrated that many in- hibitors have antioxidant activity [31]. In the present study, the significant inhibitory effect of saffron on AFB1 production should probably be attrib- uted to a synergistic action of the main bioactive con- stituents crocin and safranal, which is obtained by picro- crocin degradation. Assimopoulou et al. [32] reported that saffron methanol extract exhibited high antioxidant activity and established that crocin and safranal showed high scavenging activity which is involved in aging process, anti-inflammatory, anticancer and wound heal- ing activity as well. Additionally Papandreou et al. [40] showed the antioxidant properties of saffron stigmas ex- tract and its crocin constituents. It is interesting to report that, in the present study, AFB1 production in cultures inoculated with A. parasiticus with the addition of saf- fron before autoclaving, was not significantly different compared to control cultures (without saffron) (authors unpublished results). Autoclaving possibly inactivates crucial antioxidants such as safranal (boiling point 70˚C at 1 mm Hg) and crocetin as already mentioned previ- ously. In the present study the significance was given to the saffron activity as crude material since the antioxidant activity of the principal components has been already shown in the literature as mentioned above. Therefore although saffron consists of many components, both ac- tive and inactive, in the present study has been revealed that antioxidant properties dominate. As a result, when saffron stigmas were added to cultures of YES inoculated with A. parasiticus, AFB1 production was down 99.9% compared to control cultures, (without saffron addition) throughout the period of observation. Conclusively tak- ing account saffron’s unique properties, it might be util- ized under specific conditions for processing agricultural products, to prevent AFB1 production. 4. Acknowledgements This work was supported in part by the University of Athens, Special Account for Research Grants (70/4/ 8786). REFERENCES [1] M. J. Sweeney and A. D. W. Dobson, “Mycotoxin Pro- duction by Aspergillus, Fusarium and Penicillium Spe- cies,” International Journal of Food Microbiology, Vol. 43, No. 3, 1998, pp. 141-158. doi:10.1016/S0168-1605(98)00112-3 [2] E. Sánchez, N. Heredia and S. Garcia, “Inhibition of Growth and Mycotoxin Production of Aspergillus flavus and Aspergillus parasiticus by Extracts of Agave Spe- cies,” International Journal of Food Microbiology, Vol. 98, No. 3, 2005, pp. 271-278. doi:10.1016/j.ijfoodmicro.2004.07.009 [3] M. Giaccio, “Crocetin from saffron: An Active Compo- nent of an Ancient Spice,” Critical Reviews in Food Sci- ence and Nutrition, Vol. 44, No. 3, 2004, pp. 155-172. doi:10.1080/10408690490441433 [4] B. Renau-Morata, S. G. Nebauer, M. Sanchez and R. V. Copyright © 2012 SciRes. AiM  C. TZANIDI ET AL. 315 Molina, “Effect of Corm Size, Water Stress and Cultiva- tion Conditions on Photosynthesis and Biomass Parti- tioning during the Vegetative Growth of Saffron (Crocus sativus L.),” Industrial Crops and Products, Vol. 39, No. 1, 2012, pp. 40-46. doi:10.1016/j.indcrop.2012.02.009 [5] S. C. Nair, S. K. Kurumboor and J. H. Hasegawa, “Saf- fron Chemoprevention in Biology and Medicine: A Re- view,” Cancer Biotherapy, Vol. 10, No. 4, 1995, pp. 257- 264. doi:10.1089/cbr.1995.10.257 [6] J. L. Ríos, M. C. Recio, R. M. Giner and S. Máňez, “An Update Review of Saffron and Its Active Constituents,” Phytotherapy Research, Vol. 10, No. 3, 1996, pp. 189- 193. doi:10.1002/(SICI)1099-1573(199605)10:3<189::AID-PT R754>3.0.CO;2-C [7] J. Escribano, G. L. Alonso, M. Coca-Prados and J. A. Fernandez, “Crocin, Safranal and Picrocrocin from Saf- fron (Crocus sativus L.) Inhibit the Growth of Human Cancer Cells in Vitro,” Cancer Letters, Vol. 100, No. 1-2, 1996, pp. 23-30. doi:10.1016/0304-3835(95)04067-6 [8] H. Hosseinzadeh and H. M. Younesi, “Antinociceptive and Anti-Inflammatory Effects of Crocus sativus L. Stig- ma and Petal Extracts in Mice,” BMC Pharmacology, Vol. 2, 2002, p. 7. doi:10.1186/1471-2210-2-7 [9] J. P. Melnyk, S. Wang and M. F. Marcone, “Chemical and Biological Properties of the World’s Most Expensive Spice: Saffron,” Food Research International, Vol. 43, No. 8, 2010, pp. 1981-1989. doi:10.1016/j.foodres.2010.07.033 [10] S. M. B. Asdaq and M. N. Inamdar, “Potential of Crocus sativus (Saffron) and Its Constituent, Crocin, as Hypolip- idemic and Antioxidant in Rats,” Applied Biochemistry and Biotechnology, Vol. 162, No. 2, 2010, pp. 358-372. doi:10.1007/s12010-009-8740-7 [11] R. Kumar, V. Singh, K. Devi, M. Sharma, M. K. Singh and P. S. Ahuja, “State of Art of Saffron (Crocus sativus L.) Agronomy: A Comprehensive Review,” Food Re- views International, Vol. 25, No. 1, 2009, pp. 44-85. doi:10.1080/87559120802458503 [12] F. Olojede, G. Engelhardt, P. R. Wallnofer and G. O. Adegoke, “Decrease of Growth and Aflatoxin Production in Aspergillus parasiticus Caused by Spices,” World Journal of Microbiology and Biotechnology, Vol. 9, No. 5, 1995, pp. 605-606. doi:10.1007/BF00386307 [13] A. L. E. Mahmoud, “Antifungal Action and Antiaflatoxi- genic Properties of Some Essential Oil Constituents,” Letters in Applied Microbiology, Vol. 19, No. 2, 1994, pp. 110-113. doi:10.1111/j.1472-765X.1994.tb00918.x [14] R. Montes-Belmont and M. Carvajal, “Control of Asper- gillus flavus in Maize with Plant Essential Oils and Their Components,” Journal of Food Protection, Vol. 61, 1998, pp. 616-619. [15] V. C. Pawar and V. S. Thaker, “In Vitro Efficacy of 75 Essential Oils against Aspergillus Niger,” Mycoses, Vol. 49, No. 4, 2006, pp. 316-323. doi:10.1111/j.1439-0507.2006.01241.x [16] T. Igawa, N. Takahashi-Ando, N. Ochiai, S. Ohsato, T. Shimizu, T. Kudo, I. Yamaguch and M. Kimura, “Re- duced Contamination by the Fusarium Mycotoxin Zeara- lenone in Maize Kernels through Genetic Modification with a Detoxification Gene,” Applied Environmental Mi- crobiology, Vol. 73, No. 5, 2007, pp. 1622-1629. doi:10.1128/AEM.01077-06 [17] A. V. Loskutov, C. W. Beninger, G. L. Hosfield and K. C. Sink, “Development of an Improved Procedure for Ex- traction and Quantitation of Safranal in Stigmas of Cro- cus sativus L. Using High Performance Liquid Chroma- tography,” Food Chemistry, Vol. 69, No. 1, 2000, pp. 87- 95. doi:10.1016/S0308-8146(99)00246-0 [18] A. M. Sánchez, M. Carmona, S. A. Ordoudi, M. Z. Tsimidou and G. L. Alonso, “Kinetics of Individual Cro- cetin Ester Degradation in Aqueous Extracts of Saffron (Crocus sativus L.) upon Thermal Treatment in the Dark,” Journal of Agricultural and Food Chemistry, Vol. 56, No. 5, 2008, pp. 1627-1637. doi:10.1021/jf0730993 [19] S. Vergopoulou, D. Galanopoulou and P. Markaki, “Methyl Jasmonate Stimulates Aflatoxin B1 Biosynthesis by As- pergillus parasiticus,” Journal of Agricultural and Food Chemistry, Vol. 49, No. 7, 2001, pp. 3494-3498. doi:10.1021/jf010074+ [20] D. Abramson and R. M. Clear, “A Convenient Method for Assessing Mycotoxin Production in Cultures of As- pergilli and Penicillia,” Journal of Food Protection, Vol. 59, 1996, pp. 642-644. [21] K. K. Sinha, A. K. Sinha and G. Prasad, “The Effect of Clove and Cinnamon Oils on Growth of an Aflatoxin Production by Aspergillus flavus,” Letters in Applied Mi- crobiology, Vol. 16, No. 3, 1993, pp. 114-117. doi:10.1111/j.1472-765X.1993.tb01373.x [22] E. Daradimos, P. Markaki and M. Koupparis, “Evaluation and Validation of Two Fluorometric HPLC Methods for the Determination of Aflatoxin B1 in Olive Oil,” Food Additives and Contaminants, Vol. 17, No. 1, 2000, pp. 65-73. doi:10.1080/026520300283603 [23] R. D. Stubblefield, “Optimum Conditions for Formation of Aflatoxin M1-Trifluoroacetic Acid Derivative,” Jour- nal Association of Official Analytical Chemists, Vol. 7, 1987, pp. 1047-1049. [24] D. S. P. Patterson, “Structure, Metabolism and Toxicity of Aflatoxins: A Review,” Cahiers de Nutrition et de dietetique, Vol. 11, 1976, pp. 71-76. [25] N. Gqaleni, J. E. Smith and J. Lacey, “Co-Production of Aflatoxins and Cyclopiazonic Acid in Isolates of Asper- gillus flavus,” Food Additives and Contaminants, Vol. 13, No. 6, 1996, pp. 677-675. doi:10.1080/02652039609374453 [26] G. B. Burow, T. C. Nerbitt, J. Dunlap and N. P. Keller, “Seed Lipoxygenase Products Modulate Aspergillus My- cotoxin Biosynthesis,” Molecular Plant Microbiology In- ternational, Vol. 10, No. 3, 1997, pp. 380-387. doi:10.1094/MPMI.1997.10.3.380 [27] M. A. Klich, “Environmental and Developmental Factors Influencing Aflatoxin Production by Aspergillus flavus and Aspergillus parasiticus,” Mycoscience, Vol. 48, No. 2, 2007, pp. 71-80. doi:10.1007/s10267-006-0336-2 [28] J. D. Palumbo, J. L. Baker and N. E. Mahoney, “Isolation Copyright © 2012 SciRes. AiM  C. TZANIDI ET AL. Copyright © 2012 SciRes. AiM 316 of Bacterial Antagonists of Aspergillus flavus from Al- monds,” Microbioal Ecology, Vol. 52, No. 1, 2006, pp. 45-52. doi:10.1007/s00248-006-9096-y [29] M. L. Martins, H. M. Martins and F. Bernardo, “Afla- toxins in Spices Marketed in Portugal,” Food Additives and Contaminants, Vol. 18, 2001, pp. 315-319. [30] M. P. Doyle, R. S. Applebaum, E. Brackett and E. H. Marth, “Physical,Chemicalandbiological Degradation of Mycotoxins in Foods and Agricultural Commodities,” Journal of Food Protection, Vol. 45, 1982, pp. 964-971. [31] R. A. Holmes, R. S. Boston and G. A. Payne, “Diverse Inhibitors of Aflatoxin Biosynthesis,” Applied Microbi- ology and Biotechnology, Vol. 78, No. 4, 2008, pp. 559- 572. doi:10.1007/s00253-008-1362-0 [32] A. N. Assimopoulou, Z. Sinakos and V. P. Papageorgiou, “Radical Scavenging Activity of Crocus sativus L. Ex- tract and Its Bioactive Constituents,” Phytotherapy Re- search, Vol. 19, No. 11, 2005, pp. 997-1000. doi:10.1002/ptr.1749 [33] L. L. Zaika and R. L. Buchanan, “Review of Compounds Affecting the Biosynthesis or Bioregulation of Aflato- xins,” Journal of Food Protection, Vol. 50, 1987, pp. 691-708. [34] R. C. Lopéz and L. Goméz-Goméz, “Isolation of a New Fungi and Wound-Induced Chitinase Class in Corms of Crocus sativus,” Plant Physiology and Biochemistry, Vol. 47, No. 5, 2009, pp. 426-434. doi:10.1016/j.plaphy.2009.01.007 [35] N. Shibuya and E. Minami, “Oligosaccharide Signalling for Defence Responses in Plant,” Physiological and Mo- lecular Plant Pathology, Vol. 59, No. 5, 2001, pp. 223- 233. doi:10.1006/pmpp.2001.0364 [36] S. K. Verma and A. Bordia, “Antioxidant Properties of Saffron in Man,” Indian Journal of Medicinal Science, Vol. 52, 1998, pp. 205-207. [37] T. Jayashree and C. Subramanyam, “Oxidative Stress as a Prerequisite for Aflatoxin Production by Aspergillus parasiticus,” Free Radical Biology and Medicine, Vol. 29, No. 10, 2000, pp. 981-985. doi:10.1016/S0891-5849(00)00398-1 [38] M. Reverberi, A. A. Fabbri, S. Zjalic, A. Ricellil, F. Punelli and C. Fanelli, “Antioxidant Enzymes Stimulation in Aspergillus parasiticus by Lentinula Edodes Inhibits Aflatoxin Production,” Applied Microbiology and Bio- technology, Vol. 69, No. 2, 2005, pp. 207-215. doi:10.1007/s00253-005-1979-1 [39] L. A. Maggio-Hall, R. A. Wilson and N. P. Keller, “Fun- damental Contribution of β-Oxidation to Polyketide My- cotoxin Production in Planta,” Molecular Plant-Microbe International, Vol. 18, No. 8, 2005, pp. 783-793. doi:10.1094/MPMI-18-0783 [40] M. A. Papandreou, C. D. Kanakis, M. G. Polyssiou, S. Efthimiopoulos, P. Cordopatis and M. Margarity, “In- hibitory Activity Onamyloid-β Aggregation and Antioxi- dant Properties of Crocus sativus Stigmas Extract and Its Crocin Constituents,” Journal of Agricultural and Food Chemistry, Vol. 54, No. 3, 2006, pp. 8762-8768. doi:10.1021/jf061932a |

