Natural Science
Vol.5 No.7(2013), Article ID:33918,9 pages DOI:10.4236/ns.2013.57094
Morphological and chemical aspects of Chlorella pyrenoidosa, Dunaliella tertiolecta, Isochrysis galbana and Tetraselmis gracilis microalgae
![]()
1Department of Chemical Engineering, Federal University of Rio de Janeiro (UFRJ), Rio de Janeiro, Brazil; *Corresponding Author: tianemsilva@yahoo.com.br
2Federal Education Institute, Science and Technology of Rio de Janeiro (IFRJ), Rio de Janeiro, Brasil
Copyright © 2013 Cristiane Mesquita da Silva Gorgônio et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 10 May 2013; revised 11 June 2013; accepted 19 June 2013
Keywords: Microalgae; Macronutrients; Chromatography
ABSTRACT
This study evaluates the growth and chemical composition of the following marine microalgae: Dunaliella tertiolecta, Isochrysis galbana, and Tetraselmis gracilis and the chemical composition of Chlorella pyrenoidosa. Microalgae can produce a number of compounds of high commercial value for the industry, mainly for the food industry. The growth kinetics, cell volume, pigments, carbohydrates, proteins, lipids, and fatty acid and amino acid composition were evaluated. I. galbana had the largest number of cells per mL−1 (107), concentration of carotenoids (6.33 μg∙mL−1), and carbohydrates (34.32%). D. tertiolecta and T. gracilis had the highest cell volume (560.6 and 592.7 μm3, respectively), the highest amount of total dry biomass. D. tertiolecta had the highest chlorophyll concentration (9.05 μg∙mL−1), and C. pyrenoidosa had the highest protein (48.16%) and lipid (14.30%) content. The marine species D. Tertiolecta, I. galbana, and T. gracilis had high levels of monounsaturated fatty acids (C18:1 n9), and C. pyrenoidosa was high in polyunsaturated fatty acids (C18:2 n6 and C18:3 n3), indicating the present high nutritional value fatty acids. The microalgae studies showed a composition of amino acids that meet the nutritional requirements recommended by the FAO g∙100 g−1 (FAO/WHO/UN, 1985) for adults and children (2 - 5 years), indicating that these proteins can be used in foods.
1. INTRODUCTION
Microalgae are photosynthetic microorganisms with simple growing requirements. Its biomass can be used to produce human dietary supplements, animal feed and others. The demand for health beneficial substances has been growing on the world market, and most species studied produce optimal concentrations of compounds with high commercial value and interest to the food industry such as proteins, carbohydrates, lipids, enzymes, antibiotics, vitamins, and pigments. The biomass, merchandized as powdered formulations, tablets, capsules, and extracts can be used in pastry, snacks, sweets, drinks, and nutritional supplements [1-9]. Some species can produce 15% - 40% oil and are a promising alternative due to their rapid growth and accumulation of triglycerides [10,11]. The carbohydrates produced affect the osmotic equilibrium of the cell or act as energy store (starch and crisolaminarin), thus being a source of energy for the consumers [12-14]. The Microalgae D. tertiolecta, I. galbana and T. gracilis are flagellated marine species. The D. tertiolecta has been cultivated for the extraction of carotenoids used as natural dyes. The T. gracilis presents rapid growth and high tolerance to growing conditions, being mainly produced to feed aquatic organisms. I. galbana is rich in chlorophyll, carotenoids (fucoxanthin, β-carotene, diadinoxantin, diatoxantin) and crisolaminarin (polysaccharide derived glucose glycosidic type β-1,3). The freshwater specie C. pyrenoidosa, not flagellated, is rich in chlorophyll and is already being marketed as vitamin supplement in human nutrition [15-18].
They are known for their high levels of protein and essential amino acid composition, which are not synthesized by the human body and have lower levels of phenylalanine; therefore, they can offer additional benefits for people with Phenylketonuria (PKU). The aim of the present study is to investigate the morphological characteristics and chemical composition of some species of microalgae as well as their use as food.
2. MATERIALS AND METHODS
2.1. Microalgae
The three species of marine flagellate microalgae, Dunaliella tertiolecta, Isochrysis galbana, and Tetraselmis gracilis (Figure 1) were grown in duplicate at the Institute of Eco-development of Baía da Ilha Grande (POMAR/IED-BIG Project). The lyophilized microalgae Chlorella pyrenoidosa was obtained from the Galena® company (Campinas-SP, Brazil). The three marine species were selected due to their rapid growth in monoalgal culture and Chlorella also due to its chemical composition and by offering greater biomass for specific studies with protein hydrolysates.
2.2. Microalgal Culture
The microalgae were cultured in duplicate in 20 L polypropylene containers in Guillard (1975) medium prepared with filtered seawater [19]. The cultures were aerated with compressed air and were stored under a 24 h-photoperiod and fluorescent light (four 40 W fluorescent bulbs) at 20˚C (±2˚C). The pH equilibrium (7 to 8) was measured once daily and maintained using a TRIS buffer. The culture growth was evaluated by direct cell counting using Fuchs-Rosenthal chambers in the morning up to the eighth day, and was concentrated by tangential filtration and lyophilization. Table 1 shows the initial cells’ density (number of cells per ml).
2.3. Cell Volume Measurements
The cell volumes were determined [20]. The microalgae, fixed in a Lugol’s solution, were photographed using an Olympus Microscope BX5 (S20) using a cell^B Image acquisition software (3.0) to obtain the biovolume measurements (m3); the cell measurements were taken assuming an ellipsoid cell shape as shown in Eq.1.
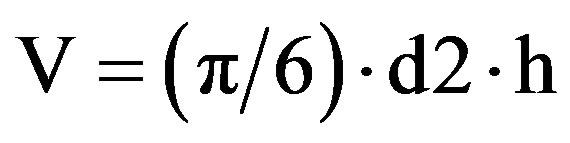
where: V = cell volume; π = 3.14; d = cell diameter; h = cell average height.
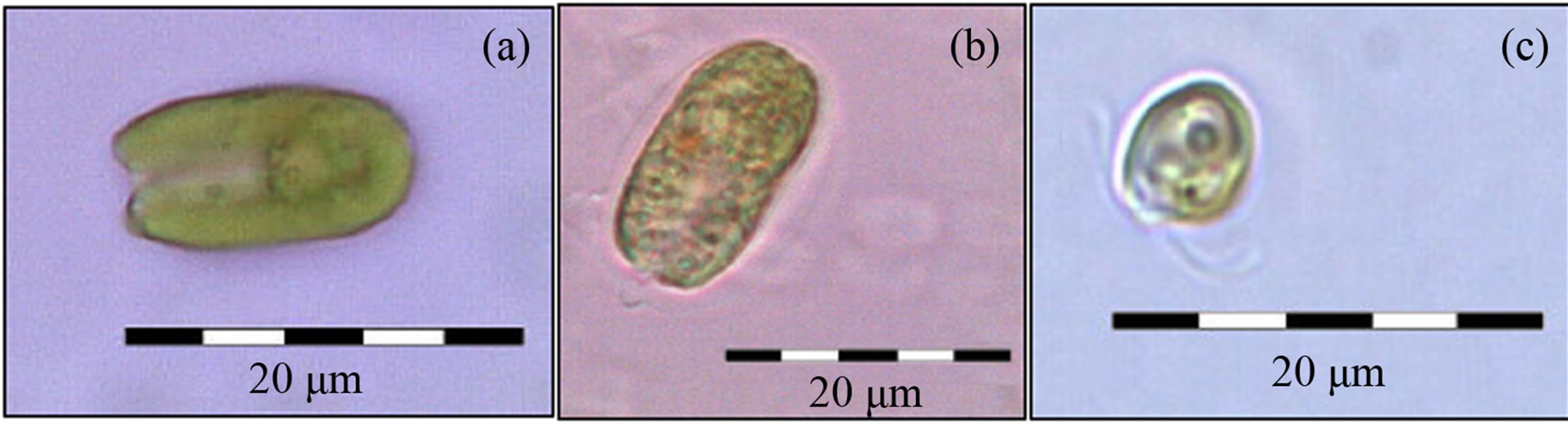
Figure 1. Marine microalgae D. tertiolecta (a) T. gracilis (b) and I. galbana (c) examined with a Olympus S20 microscope (100×).

Table 1. Average initial density of cells used in the culture.
2.4. Pigments (Chlorophyll and Carotenoids)
Five ml-aliquots of the cultures were filtered through borosilicate filter and homogenized in 5 ml of 90% acetone in triplicate. The sample was stored in the dark under refrigeration for 20 hours at 4˚C for pigment extraction. The extract was centrifuged at 3000 rpm for 10 min, and the absorbance of the supernatant (3 mL) was determined by spectrophotometry in semi-darkness [21-23].
2.5. Biomass Dry Weight
An aliquot of 4 mL of concentrated cell culture was vacuum filtered using a Sartorius filtration system. The residue retained on the filter was dried in an oven at 50˚C and determined gravimetrically to constant weight [15].
2.6. Chemical Composition of Biomass
2.6.1. Total Carbohydrates Determination
Intracellular and extracellular carbohydrate analysis was performed using 2 mg of dry weight. The analysis was based on complex sugars, and derivatives from yellow-orange colored complexes with phenol and concentrated sulfuric acid that absorb light at 485 nm [24].
2.6.2. Total Protein Determination
The intracellular proteins were determined using 2 mg of dry weight of cells by the interaction between the dye “Coomassie blue” and macromolecules containing basic side chains or aromatic amino acids. The interaction between the high molecular weight protein and the dye changes the equilibrium of the dye to the anionic form, which absorbs light at 595 nm due to the pH [25,26].
2.6.3. Total Lipid Determination
The lipid extraction [27] was performed using 500 mg of dry cells. Total lipids were estimated gravimetrically based on the dry mass of lipids and cells.
2.7. Determination of the Lipid Composition by the Methyl Ester Profile
The extracted lipids were subjected to methylation [28]. The fatty acid methyl esters were separated by the addition of distilled water: PA hexane (1:1), and the upper phase (hexane) was collected and concentrated at 60˚C. The dried sample was resuspended in 300 μL of hexane, and 1 μL was analyzed by gas chromatography using a Shimadzu GC-2014 with Quadrex Carbowax 20 M column (30m × 0.32 mm × 0.25 µm) and oven at 200˚C, flow of 20 ml/min, and Split injector and FID at 250˚C [29]. The esters were identified, and the profile was obtained by the integrated peak areas.
2.8. Determination of Amino Acid Composition
The total amino acids were determined by High Performance Liquid Chromatography (HPLC) using precolumn derivatization [30] by 6-Aminoquinolyl-N-Hydroxysuccinimidyl Carbamate (AQC), which enabled the detection of sulforated, acid hydrolysis, and tryptophan amino acids using fluorescence detection. The separation of the derivatized amino acids was optimized using an AccQ∙TagTM C18 column 3.9 × 150 mm, 4 mm particle size, and their quantitation was performed using external standardization with a standard solution containing a mixture of free amino acids. The results were expressed as mg/100g of sample (AOAC 982.30).
2.9. Statistical Analysis
The Statistica software package (for Windows version 4.0 Statsoft, Inc., 1993) was used, and the means were subjected to the Tukey test at 5% significance level.
3. RESULTS AND DISCUSSION
3.1. Microalgae Culture
According to Figure 2, I. galbana (8.56 × 107) had the highest average number of cells from the beginning of the culture up to the 8th day. D. tertiolecta (0.94 × 106) and T. gracilis (1.31 × 106) showed similar growth curve and a smaller number of cells. However, the number of cells must be evaluated together with the cell volume (cell size) and the biomass dry-weight because a greater number of cells do not necessarily mean larger biomass. Other authors have also found maximum cell density of 106 cells/mL for the D. tertiolecta and T. gracilis in the stationary phase after the 8th day of culture varying aeration, the culture medium and its nutrients, temperature, and lighting [3,14,31-33]. Most of these studies have reported curve in the order 106 - 107 cells/mL for I. galbana [3,34-36]. In the present study, a total of 107 cel/mL was found for I. galbana and 106 cells/mL for D. tertiolecta and T. gracilis.
3.2. Cell Volume Measurements
According to Table 2, the length and width of D. tertiolecta and T. gracilis was very similar. Their volume was
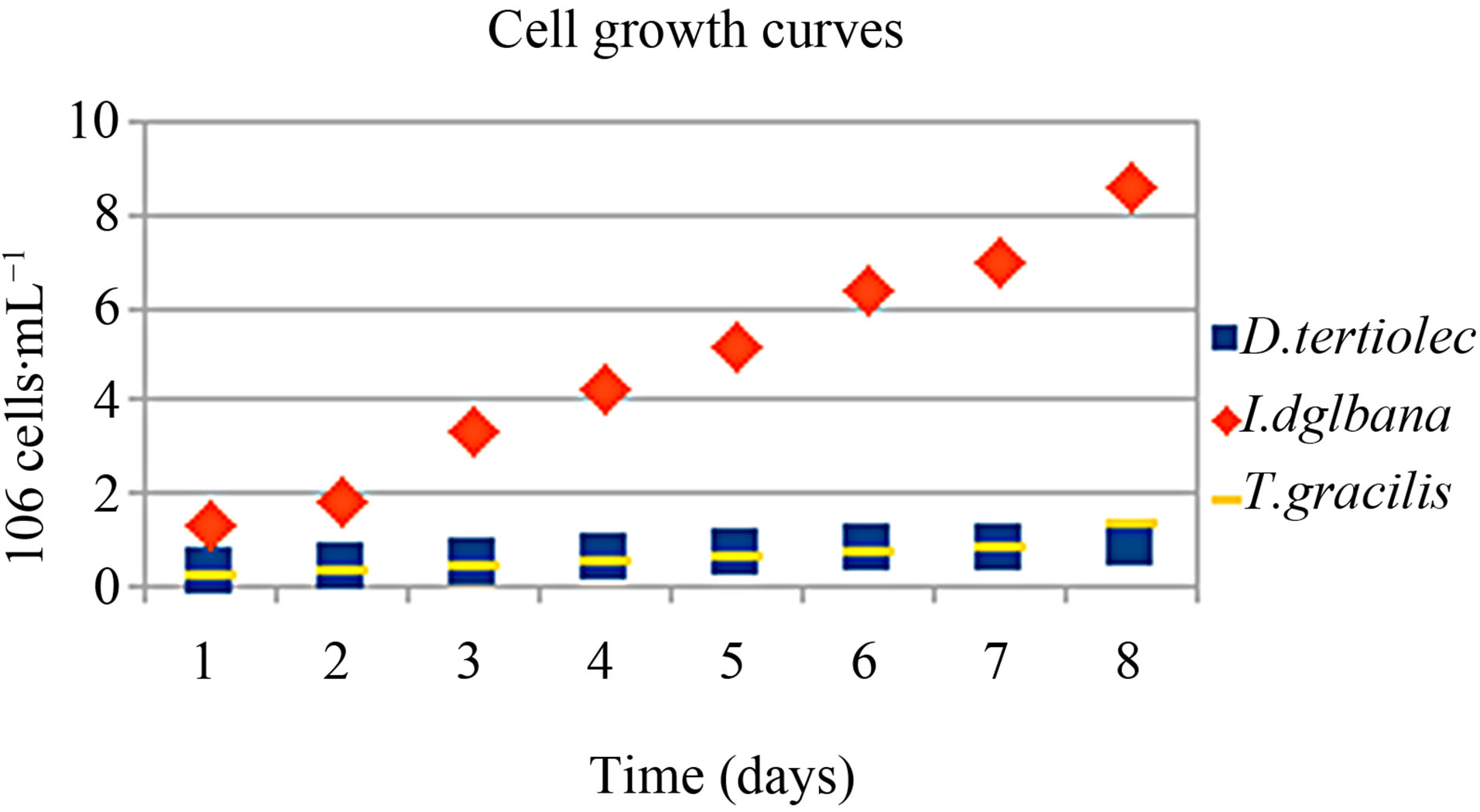
Figure 2. Growth curves of the average number of cells (mL−1) of the three marine microalgae studied.

Table 2. Average cell size and volume.
about 7 times larger than that of I. galbana. According to the literature, the length and width of D. tertiolecta and T. gracilis was 8:13 and 6.36 μm and 9.59 μm and 14.1, respectively; these values are smaller than those found in the present study, which may be due to the nitrogen content used in the culture [32]. Other authors found a cell volume range from 127 to 215 μm3, 57.3 to 58.3 μm3, and 152 to 391 μm3 for D. tertiolecta, I. galbana, and T. gracilis, respectively [3,32]. The average cell volume (μm3) found in this study was higher than that reported in the literature, and it represents an increase of 185.35% and 74% when compared to the highest volumes found. I. galbana would have greater advantage over the other two species in terms of cell number, but it has smaller cell volume and cellular dry weight when compared to these other species.
3.3. Pigments (Chlorophyll and Carotenoids)
According to Table 3, chlorophyll a was the non-degraded pigment found in the largest amount in the three microalgae species studied. D. tertiolecta showed a higher content of chlorophyll a and b (52.98 and 31.87 μg∙L−1, respectively). The second pigment found in the largest amount in D. tertiolecta and in T. gracilis was chlorophyll b, and in I. galbana it was chlorophyll c. Chlorophyll a accounted for the highest levels of pigment found since it is the main photosynthetic pigment and the other chlo-

Table 3. Average number of pigments present in the microalgae D. tertiolecta, I. galbana, and T. gracilis.
rophyll pigments are accessories which may or may not be in combined with chlorophyll a [32]. The degradation product of chlorophyll (pheophytin a) was found mainly in T. gracilis, and I. galbana showed higher levels of carotenoids, specially observed in the yellow colored culture.
Chlorophyll c is an intermediate link in the process of energy transfer from the carotenoids and chlorophyll pathway, and it is the pathway of chlorophyll biosynthesis of chlorophyll a and b [37]. I. galbana was the species with the most carotenoids and chlorophyll c, confirming this theory. Therefore, combining the cell dry weight and culture density, the average sum of chlorophylls values found was from 2:46 to 5:48 μg∙g−1 and 5:58 to 0.98 μg∙g−1 of carotenoids in the microalgae studied. When comparing with traditional foods rich in chlorophyll (Table 3), it is clear that the content of these pigments in these microalgae is small; however, it is significant in large-scale cultivation. In other studies, the values of 1.12, 0.32, and 0.93 μg/mL chlorophyll a were found in D. tertiolecta, I. galbana, and T. gracilis, respectively. The amount of 142.3 μg/L of chlorophyll a has been found in I. galbana, and 0.66, 0:10 and 0.56 μg/mL of carotenoids in D. tertiolecta, I. galbana, and T. gracilis, respectively. The microalgae investigated in the present study are chlorophyll sources with potential for modulation of xenobiotic metabolism and antioxidant and antimutagenic activity, which are effective in preventing gastrointestinal cancer [3,38]. However, it is non that chlorophylls are unstable pigments and they can be better preserved by HTST (High Temperature Short Time) treatment; moreover, their effectiveness is improved with the intake of fresh food. Carotenoids can be used as food or food additive since they can reduce atherosclerosis progression and enhance immune response preventing the formation of free radicals and reactive oxygen species that can damage mitochondrial enzymes, plasma membrane, and DNA [39-42].
3.4. Biomass Chemical Composition
Meats and soybean are rich in protein; meat is the richest available source of protein (more than 43%). Rice contains more than 77% carbohydrate. Milk and soybeans contain balanced proportions of protein, carbohydrates, and lipids [9,43].
The macronutrient composite-on of the microalgae studied was compared with that of commodities.
According to Table 4, the content of these macronutrients in these microalgae can reachlevels close to those found in soybeans and milk. The highest carbohydrate content was found in C. pyrenoidosa (34.34%), I. galbana (34.32%), and T. gracilis (29.96%). The highest lipid (14:30%) and protein content (48.16%) was found in C. pyrenoidosa. The amount of carbohydrates and proteins increased or decreased according to the species, and the lipid content in D. Tertiolecta and I. galbana showed no significant difference.
According to the literature, the content of protein in D. Tertiolecta and C. pyrenoidosa is higher (50% to 57% and 26% to 70%, respectively) [3,5,26,31,33,4-46]. Researcher have found that monosaccharides present in microalgae contain carbohydrates, mainly glucose (21% - 87%), galactose (1% - 20%), and mannose (2% - 46%) and varying amounts (0% - 17%) of arabinose, fucose, rhamnose, ribose, and xylose. According to one of these authors, species rich in mannose causes low digestibility in animals, and the composition of carbohydrates may be different depending on the phase of the cell cycle [12]. The composition of monosaccharides present in Prymnesiophyte (I. galbana), Clorophyta (C. pyrenoidosa and D. tertiolecta) and Prasinophyte (T. gracilis) can be seen in Figure 3. Some authors found 9.0 21.69% carbohydrates, 63.6% protein, and 2% to 20.5% lipids in D. tertiolecta [7,12,25,47]. Other authors found 12% to 50.8% protein, 21.7% to 21.9% lipids, and 7.6% to 14.2% carbohydrates in I. galbana [34,47]. In a study conducted in 2010, it was found more proteins in T. gracilis (33.6%) compared to that of I. galbana (29.4%) and D. tertiolecta (26.0%) in Conway medium [3]. Under temperatures between 25˚C and 35˚C, the content of protein may decrease and that of carbohydrates may increase, showing influence of temperature on the chemical composition [48].
The seawater used in the preparation of the culture medium can contain varied amounts of non-conservative nutrients such as phosphate and nitrate. The nutrient composition of the culture medium and the differences between species can also explain variations in the content of protein, carbohydrates, and lipids.

Table 4. Chemical composition of microalgae macronutrients and commodities.
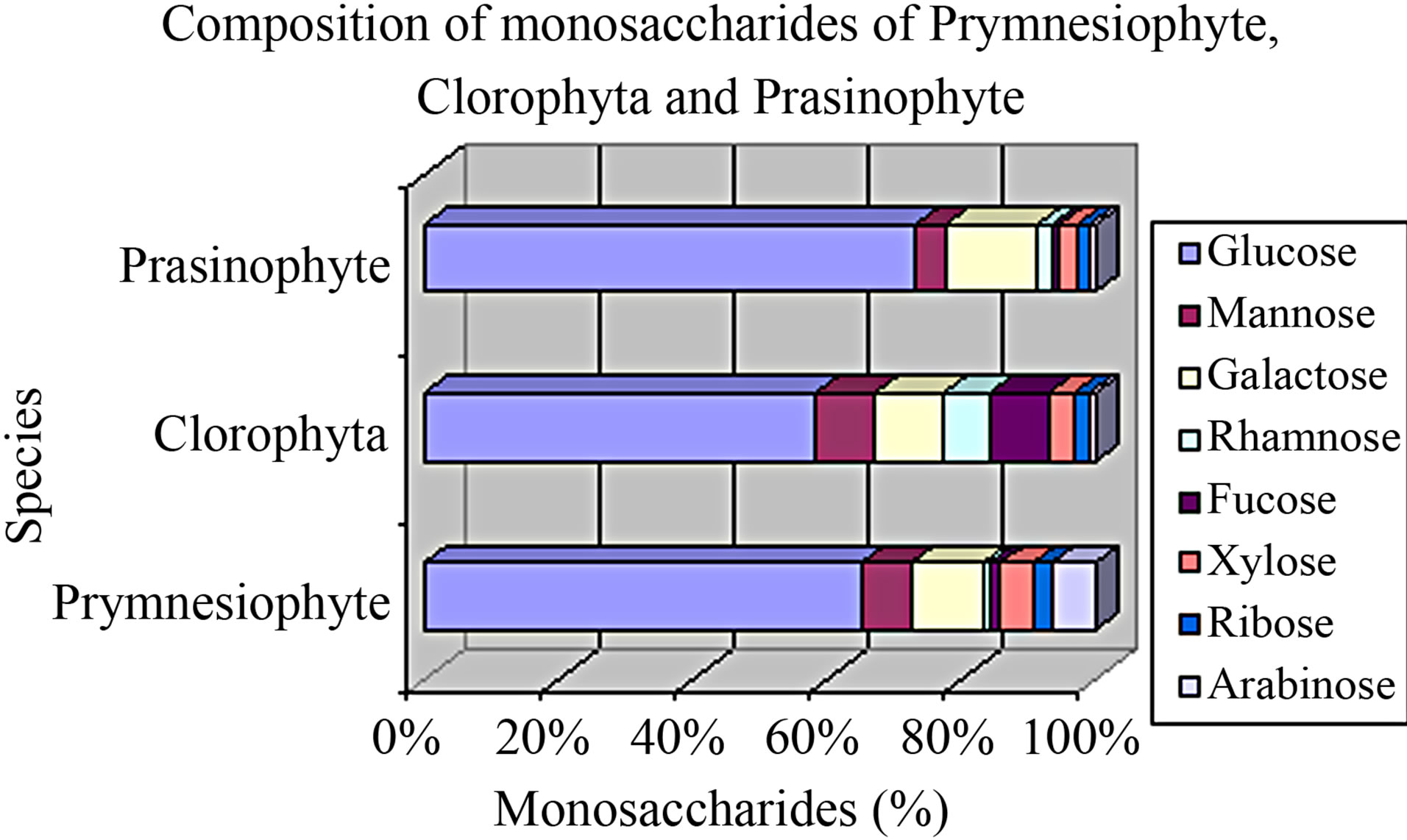 (Adapted from Brown et al. [12])
(Adapted from Brown et al. [12])
Figure 3. Composition of monosaccharides present in Prymnesiophyte, Clorophyta, and Prasinophyte.
3.4.1. Lipid Composition
Table 5 shows the fatty acid profile of the species studied and of some traditional sources of lipids. Palm stands out for its high content of C16:0 (palmitic acid: 59.12%) and C18:1 (oleic acid: 29.69%); beef tallow for C16:0 (21.83%), C18:0 (stearic acid - 23.99%), and C18:1 (40.84%); soybean for C18:1 (23:31%) and C18:2 (linoleic acid: 55.31%); and olive oil for C18:1 (67.99%) and C18:2 (2.15%). Similar results were found in the literature [49]. All marine species had high content of monounsaturated fatty acids (MUFA), and freshwater C. pyrenoidosa had the highest PUFA content (61.17%), indicating good nutritional lipid profile. The amount of C18:1 n-9 was high in all marine species; but only I. galbana had high C14:0 (myristic acid). Researchers have found 41.8%, 22.0%, and 32.9% of SAFA, MUFA, and PUFA in D. tertiolecta [3]. High content of MUFA
(54.59%), mainly C18:1 and SAFA (33.75%), was found in the present study. Some authors found the following composition of fatty acids in I. galbana: 11.1% C14:0, 15.5% C16:0, 1.1% C16:1, 2.0% C18:0, 28.6% C18:1, 7.4% of C18:2, and 3.6% C18:3 on the 8th day of culture; on the 4th day, however, the levels of C14:0, C16:1 and C18:3 were higher (13.5%, 1.4% and 4.6%, respectively), showing increased concentration of higher unsaturated acids in this microalgae. Other authors found 11.4% and 38.9% C14:0, 14.5% to 25.2% of C16:0, 16.1% to 19.6% C18:1, 8.6% C18:2, and 15.4% C18:4 [8,42,48], as well as 48.06% SAFA, 39.16% MUFA, and 12.72% PUFA in I. galbana [44], which are different from the levels found in the present study. The amount of 25.4% of C18:1, 15.2% C16:0, 14.6% C18:3, 9.0% C18:4, 6.4% C18:2 were found in T. gracilis [50]. In the present study, on the other hand, the levels of C16:0, C18:1 were higher and C18:3 was lower. In 2010, 52.26%, 23.16%, and 24.51% of SAFA, MUFA and PUFA. Higher amounts of MUFA (54.70%) and lower amounts of SAFA (35.06%) were found in the present study. The microalgae showed high contents of C16:0, 21:47% to 30.90%, characteristic of palm and beef tallow, and 37.11% to 41.32% of C18:1. It is worth mentioning that the fatty acids eicosapentaenoic acid (C20:5 n3) and docosahexaenoic (C22:6 n3) were not found in the present study; however, other authors have found these fatty acids in these species [8,11].
3.4.2. Amino Acid Composition
According to Table 6, the most abundant amino acids in all species were glutamic acid and aspartic acid, and histidine was found in the smallest amount. These results are similar to those found in Conway culture medium. [3]. Larger amounts of arginine were found in D. Tertiolecta and T. gracilis (g/100g 19.34 and 16.09, respectively), which can occasionally be regarded as essential. Some studies have reported that two species of Tetraselmis (T. suecica and T. Chuii) contained higher amount of arginine compared to that of other species [44]. Another study on types and amount of amino acids in microalgae has reported similar compositions between the species studied, suggesting proteins of similar quality. Aspartate and glutamine were found in higher concentrations (7.1% to 12.9%); cysteine, methionine, tryptophan, and histidine were found in lower amounts (0.4% to 3.2%), and other amino acids were found in amounts between 3.2% to 13.5% suggesting that the protein contains essential amino acids [12]. C. pyrenoidosa had the highest levels of tryptophan. The essential amino acid profile was similar to that of some legumes such as beans, peas, soybeans, and lentils [52-55]. All species showed levels of essential amino acids that meet the nutritional requirement recommended by FAO g/100g (FAO/WHO/ UN, 1985) for adults and children (2 - 5 years).

Table 5. Composition of lipids present in the microalgae D. tertiolecta, I. galbana, and T. gracilis and traditional sources of lipids.
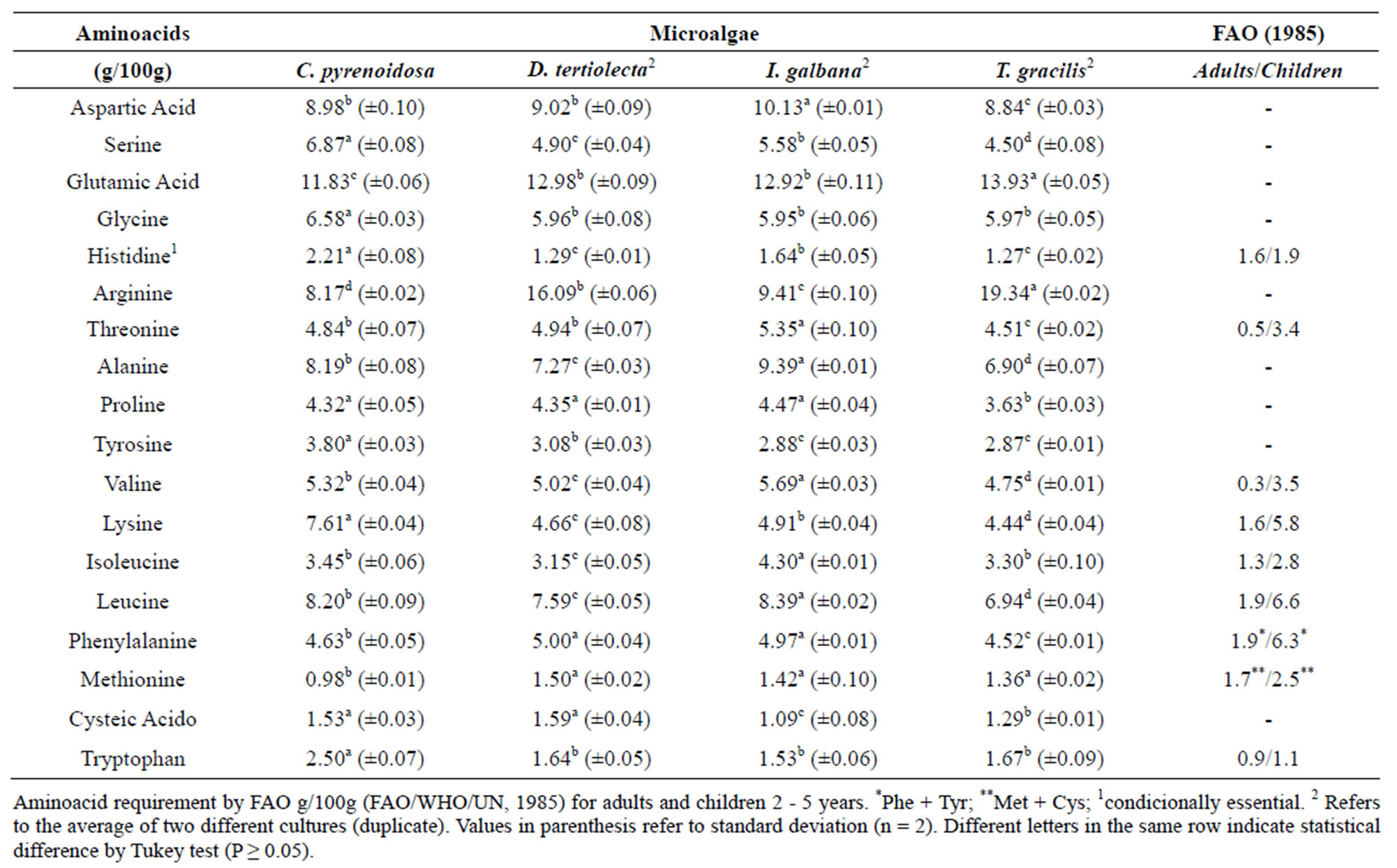
Table 6. Composition of total amino acids present in the microalgae D. tertiolecta, I. galbana and T. gracilis (g/100g).
4. CONCLUSION
D. tertiolecta and T. gracilis had the highest amount of biomass. D. tertiolecta had the highest content of chlorophyll, and I. galbana had the highest content of carotenoids. Higher levels of PUFA were found in C. pyrenoidosa showing better nutritional quality of this oil. The marine species stand out for the presence of MUFA, especially oleic acid (C18:1 n9). The concentration of essential amino acids meets the nutritional guidelines for children and adults (FAO/1985) and suggests that the microalgae proteins can be used in the production of food as well as combined with other protein sources. The bioavailability of nutrients and the composition of nitrogenous and carbohydrate compounds in the microalgae are issues that have been little studied but should be further investigated.
REFERENCES
- Andrade, M.R. and Costa, J.A.V. (2008) Cultivo da microalga Spirulina platensis em fontes alternativas de nutrientes. Ciênc Agrotec Lavras, 32, 1551-1556.
- Bertoldi, F.C., Sant’anna, E. and Oliveira, J.L.B. (2008) Revisão: Biotecnologia de microalgas. B. CEPPA, 26, 9-20.
- Campos, V.B., Barbarino, E. and Lourenço, S.O. (2010) Crescimento e composição química de dez espécies de microalgas marinhas em cultivos estanques. Ciência Rural, 40, 339-347.
- Derner, R.B., Ohse, S., Villela, M., et al. (2006) Microalgas, produtos e aplicações. Ciência Rural, 36, 1959- 1967. doi:10.1590/S0103-84782006000600050
- Harun, R., Singh, M., et al. (2009) Bioprocess engineering of microalgae to produce a variety of consumer products. Renew Sustain Energy, 14, 1037-1047. doi:10.1016/j.rser.2009.11.004
- Herrero, M., Cifuentes, A. and Ibañez, E. (2006) Sub and supercritical fluid extraction of functional ingredients from different natural sources: Plants, foodby-products, algae and microalgae: A review. Food Chemistry, 98, 136-148. doi:10.1016/j.foodchem.2005.05.058
- Mata, T.M., Martins, A.A. and Caetano, N.S. (2010) Microalgae for biodiesel production and other applications: A review. Renewable and Sustainable Energy Reviews, 14, 217-232. doi:10.1016/j.rser.2009.07.020
- Radmann, E.M. and Costa, J.A.V. (2008) Conteúdo lipí- dico e composição de ácidos graxos de microalgas expostas aos gases CO2, SO2 e NO. Quim. Nova, 31, 1609. doi:10.1590/S0100-40422008000700002
- Spolaore, P., Joannis-Cassan, C. and Duran, E. (2006) Review: Commercial applications of microalgae. Journal of Bioscience and Bioengineerin, 101, 87-96. doi:10.1263/jbb.101.87
- Chen, W., Zhang, C., Song, L., et al. (2009) A high throughput Nile red method for quantitative measurement of neutral lipids in microalgae. Journal of Microbiological Methods, 27, 41-47. doi:10.1016/j.mimet.2009.01.001
- Huang, G., Chen, F., Wei, D., et al. (2010) Biodiesel production by microalgal biotechnology. Applied Energy, 87, 38-46. doi:10.1016/j.apenergy.2009.06.016
- Brown, M.R., Jeffrey, S.W., Volkman, J.K., et al. (1997) Nutritional properties of microalgae for mariculture. Aquaculture, 151, 315-331. doi:10.1016/S0044-8486(96)01501-3
- Martínez-Fernández, E.., Salmón, H. and Southgate, A.P.C. (2006) The nutritional value of seven species of tropical microalgae for black-lip pearl oyster (Pinctada margaritifera, L.) larvae. Aquaculture, 257, 491-503. doi:10.1016/j.aquaculture.2006.03.022
- Strickland, J.D.H. and Parsons, T.R. (1968) A practical handbook of seawater analysis. Journal of the Fisheries Research Board of Canada., Ottawa, 311 pages.
- Lourenço, S.O. (2006) Cultivo de microalgas marinhas: Princípios e aplicações. Rima, São Carlos, 606 pages.
- Sawayama, S., Minowa, T. and Yokoyama, S.Y. (1999) Possibility of renewable energy production and CO2 mitigation by thermochemical liquefaction of microalgae. Biomass and Bioenergy, 17, 33-39. doi:10.1016/S0961-9534(99)00019-7
- Thorondsen, J. (1993) Identfying marine phytoplankton. Academic Press, New York, 591-729.
- Zelitch, I. (1971) Photosynthesis, photorespiration and plant productivity. Academic Press, 275.
- Guillard, R.R.L. (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith, W.L. and Chanley, M.H., Eds., Culture of Marine Invertebrates Animals, Plenum Publishing, New York, 29-60. doi:10.1007/978-1-4615-8714-9_3
- Hillebrand, H., Dürselem, C., Kirshtel, D., et al. (1999) Biovolume calculation for pelagic and benthic microalgae. Journal of Phycology, 35, 403-424. doi:10.1046/j.1529-8817.1999.3520403.x
- Jeffrey, S.W. and Humphrey, G. F. (1975) New spectrophotometric equations for determinig chlorophylls a b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochemie und Physiologie der Pflanzen, 167, 191-194.
- Lorenzen, C.J. (1967) Determination of chlorophyll and phaeophytin: Spectrophotometric equations. Limnology and Oceanography, 12, 343-346 doi:10.4319/lo.1967.12.2.0343
- Zaia, D.A.M., Zaia, C.T.B.V. and Lichtig, J. (1998) Determinação de proteínas totais via espectrofometria: vantagens e desvantagens dos métodos existentes. Química Nova, 21, 787-793. doi:10.1590/S0100-40421998000600020
- Dubois, M., Guilles, K.A., Hamilton, J.K., et al. (1956) Calorimetric method for the determination of sugars and related substances. Analytical Chemistry, 18, 350-356. doi:10.1021/ac60111a017
- Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248-254. doi:10.1016/0003-2697(76)90527-3
- Viêgas, C.V. (2010) Extração e caracterização dos lipí- deos da microalga Chlorella pyrenoidosa visando à produção de ésteres graxos. Dissertação Universidade Federal do Rio Grande (FURG), Rio Grande.
- Folch, J., Lees, M. and Stanley, G.H.S. (1956) A simple method for the isolation and purification of total lipids from animal tissues. The Journal of Biological Chemistry, 497-509. http://www.jbc.org
- Yoo, C., Jun, S.Y., Lee, J.Y., et al. (2010) Selection of microalgae for lipid production under high levels carbon dioxide. Bioresource Technology, 101, 71-74. doi:10.1016/j.biortech.2009.03.030
- EN14103 (2003) Derivados de óleo e gordura-Ésteres Metílicos de Ácidos Graxos (FAME)-Determinação quantitativa de Ésteres e Ésteres Metílicos de Ácido Linoléico.
- Cohen, A.M. and Michaud, D.P. (1993) Synthesis of fluorescent derivatizing reagent, 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate, and its application for the analysis of hydrolysate amino acids via high-performance liquid chromatography. Analytical Biochemistry, 211, 279- 287. doi:10.1006/abio.1993.1270
- Fábregas, J., Ferrón, L., Gamallo, Y., et al. (1994) Improvement of growth and cell productivity by aeration rate in cultures of the marine microalga Dunaliella tertiolecta. Bioresource Technology, 48, 107-111. doi:10.1016/0960-8524(94)90196-1
- Lavin, P.L.S. (2000) Nitrogênio orgânico intracelular: Extração, papel fisiológico e acumulação por microalgas marinhas, em cultivos estanques. Dissertação Universidade Federal Fluminense, Niterói, Rio de Janeiro.
- Ohse, S., Derner, R.B., Ozório, R.A., et al. (2009) Produção de biomassa e teores de carbono, hidrogênio, nitrogênio e proteínas em microalgas. Ciencia Rural, 39, 1760- 1767. doi:10.1590/S0103-84782009000600019
- Fidalgo, J.P., Cid, A., Torres, E., et al. (1998) Effects of nitrogen source and growth phase on proximate biochemical composition, lipid classes and fatty acid profile of the marine microalga Isochrysis galbana. Aquaculture, 166, 105-116. doi:10.1016/S0044-8486(98)00278-6
- Junior, A.M.M., Neto, E.B., Koening, M.L., et al. (2006) Composição química de microalgas em cultivo semiintensivo: Chaetoceros gracilis Schutt, Isochrysis galbana Parke e Thalassiosira weissflogii (Grunow) G. Fryxell & Hasle. Revista Ciência Agronômica, 37, 142-148.
- Valenzuela-Espinoza, E., Millán-Núñez, R. and NúñezCebrero, F. (2002) Protein, carbohydrate, lipid and chlorophyll a content in Isochrysis aff. galbana (clone Tiso) cultured with a low cost alternative to the f/2 medium. Aquacultural Engineering, 25, 207-216. doi:10.1016/S0144-8609(01)00084-X
- Falkowski, P.G. and Raven, J.A. (1997) Aquatic photosynthesis. Blackwell Scientific, Oxford.
- Ferruzzi, M.G. and Blakeslee, J. (2007) Digestion, absorption, and cancer preventative activity of dietary chlorophyll derivatives. Nutrition Research, 27, 1-12. doi:10.1016/j.nutres.2006.12.003
- Gomes, F.S. (2007) Carotenóides: Uma possível proteção contra o desenvolvimento de câncer. Revista de Nutrição, 20, 537-548.
- Gomes, M.M., Saunders, C. and Accioly, E. (2005) Papel da vitamina A na prevenção do estresse oxidativo em recém-nascidos. Revista Brasileira de Saúde Materno Infantil, 5, 275-282.
- Lanfer-Marquez, U.M. (2003) O papel da clorofila na alimentação humana: Uma revisão. Revista Brasileira de Ciências Farmacêuticas, 39, 227-242.
- Oliveira, G.S., Figueiredo, Á.S.P., Santos, R.S., et al. (2007) Efeito da suplementação de beta-caroteno na pressão arterial de ratos. Revista de Nutrição, 20, 39-45. doi:10.1590/S1415-52732007000100004
- Franco, G. (2002) Tabela de composição química dos alimentos. 5th Edition, Atheneu, São Paulo.
- Brown, M. (1991) The amino-acid and sugar composition of 16 species of microalgae used in mariculture. Journal of Experimental Marine Biology and Ecology, 145, 79-99. doi:10.1016/0022-0981(91)90007-J
- Sánches, S., Martinez, M.E. and Espinola, F. (2000) Biomass production and biochemical variability of marine microalga Isochrysis galbana in relation to culture medium. Biochemical Engineering Journal, 6, 13-18. doi:10.1016/S1369-703X(00)00071-1
- Soares, A. and Clavico, E. (2005) Propriedades físicoquímicas da água. Universidade Federal Fluminense, Niterói. http://www.uff.br/ecosed/PropriedadesH2O.pdf
- Renaud, S.M., Luong-van, T., Lambrinidis, G., et al. (2002) Effect of temperature on growth, chemical composition and fatty acid composition of tropical Australian microalgae grown in batch cultures. Aquaculture, 211, 195-214. doi:10.1016/S0044-8486(01)00875-4
- Ogbonna, J.C. and Tanaka, H. (1996) Night biomass loss and changes in biochemical composition of cells during light/dark cyclic culture of Chlorella pyrenoidosa. Journal of Fermentation and Bioengineering, 82, 558-564. doi:10.1016/S0922-338X(97)81252-4
- Moraes, M.S.A. (2008) Biodiesel de sebo: Avaliação de propriedades e testes de consumo em motor a diesel. Dissertação Universidade Federal do Rio Grande do Sul, Porto Alegre.
- Reitan, K.I., Rainuzzo, J.R., Oie, G., et al. (1997) A review of the nutritional effects of algae in marine fish larvae. Aquaculture, 155, 207-221. doi:10.1016/S0044-8486(97)00118-X
- Aued-Pimentel, S., Takemoto, E., Kumagai, E.E., et al. (2008) Determinação da diferença entre o valor real e o teórico do triglicerídeo ECN 42 para a detecção de adulteração em azeites de oliva comercializados no Brasil. Quim. Nova, 31, 31-34. doi:10.1590/S0100-40422008000100006
- Bertol, T.M., Moraes, N. and Franke, M.R. (2001) Substituição parcial do farelo de soja por proteína texturizada de soja na dieta de leitões desmamados. Revista Brasileira de Zootecnia, 30, 141-149. doi:10.1590/S1516-35982001000100021
- Khattab, R.Y., Arntfield, S.D. and Nyachoti, C.M. (2009) Nutritional quality of legume seeds as affected by some physical treatments, part 1: Protein quality evaluation. Food Science and Technology, 42, 1107-1112.
- Karr-Lilienthal, L.K., Grieshop, C.M., Spears, J.K., et al. (2005) Amino acid, carbohydrate, and fat composition of soybean meals prepared at 55 commercial US soybean processing plants. Journal of Agricultural and Food Chemistry, 53, 2146-2150. doi:10.1021/jf048385i
- Rozan, P., Yu-Haey, K. and Lambein, F. (2001) Amino acids in seeds and seedlings of the genus Lens. Phytochemistry, 58, 281-289. doi:10.1016/S0031-9422(01)00200-X
- FAO. (1985) Energy and protein requirements. Technical Report, Joint FAO/WHO/ONU Expert Consulation.

