Synthesis and Electrochemical Properties of Transparent Nanostructured BaTiO3 Film Electrodes ()
1. Introduction
Dye-sensitized solar cells (DSSCs) have attracted considerable attention in recent years due to their simple structure, low production cost and high performance [1] -[8] . The fundamental component of the DSSCs is the nanoporous electrode formed by nanocrystalline semiconductors. In order to improve the energy conversion efficiency, the development of new electrode materials seems to be one of the main research targets. In recent years, the metal oxides have been focused and employed typically such as wide band gap n-type semiconductors TiO2, SnO2, ZnO, et al. [9] - [12] .
An obvious characteristic of nanoporous electrodes is the large density of surface states. They are the most important when the energies of these states lie in the band gap. Electrochemical and spectroelectrochemical methods are valuable in the study of their properties [11] - [18] .
In comparison with these binary oxide semiconductors, the structure of oxide Barium titanate (BaTiO3) was similar to anatase TiO2, and could be loosely as a highly doped TiO2 structure. The titanium atoms were 6-foldoctahedral coordination in Barium titanate (BaTiO3). At the same time, the band gap of BaTiO3 was nearly the same as that of anatase TiO2 (3.2 ev) [19] [20] . But there is rarely report about nanostructured BaTiO3 films so far.
In this study, monodispersed BaTiO3 nanoparticles were prepared at low temperature and were fabricated as nanostructured BaTiO3 films electrodes. The properties of band energetics and the trap state at different pH values were investigated with electrochemical and spectroelectrochemical techniques.
2. Experiment
2.1. Materials
Ti(OCH(CH3)2)4, Ba(OH)2 and Ethyl cellulose were purchased from Tianjin Chemical Company. LiClO4, HClO4, terpineol and tetramethylammonium hydroxide were purchased from Shanghai Nuotai Chemical Company. Optically transparent electrodes (OTE) were fabricated on an F-doped SnO2-coated glass substrate. Water (R = 18.3 MW) was obtained from pure RF system. All the chemicals were reagent grade.
2.2. Preparation of Nanostructure BaTiO3 Electrodes
Preparation of BaTiO3 nanocrystals: BaTiO3 nanocrystals were synthesized using a slightly modified previously published technique [21] [22] . At first, 5 mL of Ti(n-OC4H9)4 and 1.5 mL of acetylacetone were mixed together. After addition of 10 mL of ethanol, the mixture solution was backflowed at 100˚C for 0.5 h. 4.7 g Ba(OH)2 was dissolved in 60 mL of boiling water under vigorously stirring. After Ba(OH)2 were completely dissolved, the former mixed solution was added to Ba(OH)2 solution slowly under constantly at 100˚C in 1h. Then the mixture solution turned white and white crystals precipitate was formed. The precipitation was eventually washed with distilled water and dried at room temperature.
Preparation of nanostructured BaTiO3 films: Resulting precipitate was washed with ethanol completely. 12.5 g of terpineol and
15 g
of 10 wt% ethyl cellulose ethanol solution were added to the BaTiO3 paste. Then the mixture was dispersed under sonication, and ethanol was removed by rotary-evaporator. The final solid were grounded with a mill.
BaTiO3 sol was spread on the substrates by a glass rod with adhesive tapes as spacers. The films were dried at 105˚C and sintered at 500˚C for 30 min in air and finally cooled to room temperature.
3. Results and Discussions
3.1. Characterization of Nanostructured BaTiO3 Film
X-ray diffraction (XRD) measurements were performed on a D 8 diffractometer (Bruker Co.) with Cu Ka (λ = 1.5405 Å) to identify the phase structure of samples. Figure 1 shows the XRD patterns of the BaTiO3 film which has a high pure cubic perovskite structure (JCPDS card 31-0174). The surface structure of the film could be obviously observed by SEM (S-4800, Hitachi, Japan) as shown in Figure 2. The SEM image reveal a bimodal particle distribution of the transparent films with small cubic particles with the diameter between 40 and 60 nm and large particles with the diameter 80 - 90 nm. The bimodal distribution is much desired in DSSCs which can increase light scattering and increase theoretical path length in the photoelectrode [13] [23] .
The absorption spectra of the BaTiO3 film deposited on a quartz substrate were recorded on an UV-1240 spectrophotometer (Shimadzu, Japan) as shown in Figure 3. Absorption tail at long wavelength was due to the diffusion and reflection of nanostructured BaTiO3 film. The onset at 385 nm was corresponded to a band gap of 3.23 eV.
![]()
Figure 3. Absorption spectrum of a nanostructured BaTiO3 film.
3.2. Spectroelectrochemistry and Flat Band Determination
All electrochemical and spectroelectrochemical experiments were carried out in a typical three-electrode system, in which a nanostructured BaTiO3 electrode, a platinum wire and a saturated Ag/AgCl electrode acted as working, counter and reference electrodes respectively. Spectroelectrochemistry measurements were undertaken according to the published literature [10] . A quartz cell with three electrodes and electrolyte was incorporated into the sample compartment of a Shimadzu UV-vis spectrophotometer and connected to a CHI 800 potentiostat. All aqueous electrolyte solutions were prepared based on LiClO4 as supporting electrolyte and the pH value of the electrolyte solutions was adjusted by HClO4 (for pH 3.0) or NaOH (for pH 13.0). The electrolyte solutions were thoroughly degassed with N2 prior to experiments. All potentials were given with reference to the saturated Ag/AgCl electrode. The working area of BaTiO3 electrodes were
3 cm
2.
The potential-dependent absorption spectra of nanostructured BaTiO3 electrodes measured in aqueous electrolytes of different pHs were shown in Figure 4. The absorbance increased at longer wavelengths as the application of more negative potential. The absorption onset shifted to more negative potential as pH increased.
Spectroelectrochemical measurement was usually applied for the monitoring of the electron filling in conduction band. Because the filling of electrons in conduction band could be monitored by the absorbance changes at long wavelength, the conduction band edge of a semiconductor could be calculated [11] -[13] . In Figure 4, the absorbance at 800 nm were plotted against applied potentials and shown in insetting figures. The flat band edges were closely dependent on pH of the electrolyte. The flat band edges were −0.70, −0.92 and −1.20 V at pH value of 3.0, 6.8 and 13.0 respectively.
The flat band potential is dependent on pH of electrolyte and is shown by the equations [10] [24]
 (1)
(1)
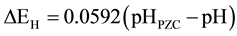 (2)
(2)
where pHPZC is the pH at point of zero charge (PZC), 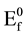 is the Fermi level at pHPZC, ΔEH is the potential drop in Helmholtz layer. When pH is smaller than pHPZC, there are net positive charges on surface, so ΔEH > 0. The smaller the pH is, the larger the ΔEH is and the flat band potential shifts more positively. On the other hand, when pH is larger than pHPZC, there are net negative charges on surface, so ΔEH < 0. The larger the pH is, the smaller the ΔEH is and the flat band potential shifts more negatively.
is the Fermi level at pHPZC, ΔEH is the potential drop in Helmholtz layer. When pH is smaller than pHPZC, there are net positive charges on surface, so ΔEH > 0. The smaller the pH is, the larger the ΔEH is and the flat band potential shifts more positively. On the other hand, when pH is larger than pHPZC, there are net negative charges on surface, so ΔEH < 0. The larger the pH is, the smaller the ΔEH is and the flat band potential shifts more negatively.
The dependence of the flat band edge of a nanostructured BaTiO3 film on pH of electrolyte is shown in Figure 5. A linear relation is obtained from fitting data linearly and expressed as Efb = −0.56 − 0.029 pH.
3.3. Time-Resolved Current and Trap State Distribution
Time-resolved current at pH 3.0: The current-time curves of a nanostructured BaTiO3 electrode were measured in LiClO4 solution with the concentration of 0.2 mol∙L−1 at the pH value of 3.0 under different potentials and were shown in Figure 6(a). The applied potential was significantly influenced by the current. The currents were almost decreased to zero quickly when the potential was from 0 to −0.3 V. After the applied potential was more negative than −0.3 V, the current was decreased slowly. The results showed the band gap region was filled by trap. A nanostructured BaTiO3 electrode had a flat band edge of −0.70 V at pH 3.0 (seen from Figure 5). When the potentials was more positive than −0.3 V, the density of traps was low, and thus the trap-filling time was short. At the same time, the decay of the time resolved current was fast [22] . On the other hand trap density was increased and the time of filling these traps needed long. The longest trap-filling time was −0.36 V which was just below the conduction band edge.
The accumulated charge Q under the current-time curves in Figure 6(a) was calculated and was shown in Figure 6(b). If the accumulated charge Q from trap-filling reflects the density of states, Equation (3) can be obtained [12] [20]
Ntrap(U) = (1/q)(dQ/dU) (3)
where Q is accumulated charge, Ntrap(U) is density of trap states at potential U and q was electron charge. Equation (3) clearly indicates that trap density is directly proportional to dQ/dU, which provides a direct measurement of trap distribution. By differentiating the accumulated charge to the applied potential, a plot of dQ/dU against U is obtained and shown in the insert of Figure 6(b). The totally trap states are 3.73 × 1015 cm−2.
![]() (a)
(a)![]() (b)
(b)![]() (c)
(c)
Figure 4. Differential spectra of nanostructured BaTiO3 electrodes in: (a) 0.2 mol∙L−1 LiClO4 at pH 3.0; (b) 0.2 mol∙L−1 LiClO4 at pH 6.8; (c) 0.2 mol∙L−1 LiClO4 at pH 13.0. The inserts show absorbance changes at 800 nm. Spectra are recorded after polarized for 5 min at indicated potentials. The spectrum measured after stabilization for 15 min at +0.8 V has been subtracted.
![]()
Figure 5. Flat band potential of nanostructured BaTiO3 films as a function of pH.
![]() (a)
(a)![]() (b)
(b)
Figure 6. (a) Current-time curves of a nanostructured BaTiO3 electrode in 0.2 mol∙L−1 LiClO4 of pH 3.0. The electrode was initially polarized at 0.8 V for 5 min and then measured at different applied potential. (b) Cathodic charges at different potentials derived by integrating the current-time curves in Figure 6(a). The insert shows dQ/dU distribution against potential.
Time resolved currents at pH 6.8 and 13.0: The current-time curves in pH 6.8 and 13.0 solutions are similar to that in pH 3.0 solution and the cathodic charges at different potentials by integrating current-time curves measured in both solutions are shown in Figure 7(a), Figure 7(b) and Figure 8(a), Figure 8(b) respectively. The longest trap-filling time was at −0.5 V for pH 6.8 and −0.8 V for pH 13.0. The total amounts of trapped electrons were 4.02 × 1015 cm−2 for pH 6.8 and 6.48 × 1015 cm−2 for pH 13.0. It is obvious that the trap-filling process is faster in the pH 3.0 solution and pH 6.8 than that in pH 13.0 solution.
3.4. Cyclic Voltammetry and Surface Traps Determination
Cyclic voltammetry is a method for detecting and characterizing surface traps in nanocrystalline electrodes. A
![]()
Figure 7. (a) Current-time curves of a nanostructured BaTiO3 electrode in 0.2 mol∙L−1 LiClO4 of pH 6.8; (b) Cathodic charges accumulated at different potentials as derived by integrating the current-time curves at: pH 6.8. The insert shows dQ/dU distribution against potential.
![]()
Figure 8. (a) Current-time curves of a nanostructured BaTiO3 electrode in 0.2 mol∙L−1 LiClO4 of pH 13.0; (b) Cathodic charges accumulated at different potentials as derived by integrating the current-time curves at: pH 6.8. The insert shows dQ/dU distribution against potential.
shoulder was appeared in the current-potential curves which was more positive than the conduction band edge. The shoulder has been generally assigned to the presence of electron traps [12] [20] . The cyclic voltammograms of nanostructured BaTiO3 electrodes were recorded at different pH and were shown in Figure 9.
The conduction-band edge of the nanostructured BaTiO3 electrode at pH 3.0 is approximately at −0.70 V (see Figure 5) and a feature at −0.36 V in the cyclic voltammogram is corresponded to trap state filling below the conduction band edge which confirms the presence of surface traps [12] [25] [26] . The most trap state distribution at each pH are located at −0.36, −0.51, and −0.82 V at pH 3.0, 6.8, and 13.0 respectively, which are in good agreement with the results obtained from Figure 6(b), Figure 7(b) and Figure 8(b). It should be noticed that the size of the peak increases dramatically with pH increasing, so this well indicates that the traps are the most surface- related.
![]()
Figure 9. Cyclic voltammograms of nanostructured BaTiO3 electrodes measured at different pH values. The electrodes were initially polarized at 0.8 V for 15 min before scanning, the scan rate was 5 mV/s.
4. Conclusion
Transparent nanostructured BaTiO3 film electrodes have been synthesized and the band energetics and the flat band edges (Efb) of nanostructured BaTiO3 electrodes have been determined by electrochemical and spectroelectrochemical methods. The flat band edges of the nanostructured BaTiO3 electrodes greatly depended on pH of electrolytes. The potentials turned more negative with the increase of pH value. The trap state distribution was investigated by the measurements of time resolved current. Total trap state densities were 3.73 × 1015, 4.02 × 1015 and 6.48 × 1016 cm−2 at pH 3.0, 6.8 and 13.0 respectively with maximum located at −0.36 V, −0.50 V and −0.80 V. The CVs results were in good agreement with that obtained from the measurements of time-resolved currents. The size of the peak in the cyclic voltammograms increased dramatically with the increase in pH value, indicating that traps were the most surface-related.
Acknowledgements
This work was supported financially by the National Natural Science Foundation of China (Grant No. 20773103), Science & Technology Program of Education Department of Henan (
12A
150021).
NOTES
*Corresponding author.