Variation in Thermal time model Parameters Between Two Contrasting Chickpea (Cicer arietinum) cultivars ()
Received 3 November 2015; accepted 5 December 2015; published 10 December 2015

1. Introduction
Chickpea (Cicer arietinum L.) is the third most extensively planted grain legume in the world after dry bean and field pea [1] . Besides being an important legume crop for human nutrition, chickpea also plays an important role in increasing and maintaining soil fertility and the recovery of marginal lands, especially in arid and semi-arid regions [1] . Chickpea is currently grown on about 12 million hectares worldwide with an average annual production of 10.9 million tons [2] . About 95% of chickpea cultivation and consumption is in the developing countries [3] . In Sudan, chickpea is traditionally grown as a winter crop in the northern part, however, its production has expanded recently to the central clay plain of central Sudan [4] . The growing season is restricted to a short period of time by the high temperatures prevailing at the beginning and at the end of the season [4] . The chickpea yields in Sudan vary from 0.83 to 2.8 t/ha, depending on weather conditions [5] . Temperature is one of the most chickpea production constraints in the main production areas in Sudan. Worldwide, chickpea is either grown during the post rainy season on stored soil moisture (South Asia and spring sown Mediterranean) or as a Mediterranean winter crop in season rainfall; in both instances the crop is exposed to terminal drought which is accompanied by rising temperatures. The South Asian crop may also experience high temperatures in the seedling phase if planted early [6] . Chickpea productivity is constrained by several abiotic stresses [7] [8] and temperature is one of the most important determinants of seed germination and seedling growth over a range of environments and may limit chickpea yield [9] . Cultivars grown in Sudan are either local (desi) type (called “Jabel Marra”) characterized by smaller, angular, light brown (pigmented) seeds and early mature or introduced Mediterranean (kabuli) type with larger, rounded seeds which lack pigmentation which is late mature cultivar such as NEC 2491 (locally called “Shendi”). The second cultivar has been released and recommended to be grown in Northern Sudan due to their vigorous growth and high yield potentiality [4] . Since kabuli-types are indigenous to Mediterranean areas, we presume that some genotypes may have a degree of adaptation to low temperature. Genotypic variability amongst the desi and kabuli cultivars in Sudan has been reported recently [4] .
The establishment of plants is often limited by temperature when moisture conditions are favourable [10] . Germination characteristics have often been used for evaluating response of plant development to temperature. Many researchers [11] - [13] found a linear relationship between the inverse time of germination (germination rate) and temperature. Temperature is the single most important factor governing the germination [14] . Previous studies have shown that germination rate usually increases linearly with temperature, at least within a well-defined range and declines sharply in higher temperatures [14] [15] . This temperature range has been defined as cardinal temperatures, i.e., a minimum or base temperature (Tb), maximum temperature (Tc) that germination rate at above of that will be zero and optimum temperature (To) at which the germination rate is the highest [15] . Several researchers have shown that the cardinal temperatures for germination depend on species and within species vary significantly [16] . At suboptimal temperatures the thermal time approach can be used to predict germination time. Thermal time (degree-day or hour) principle was first introduced by [13] to describe the linear relationship between germination rate and constant temperature in pearl millet.
Seed size is a genotypic character which varies between and within species. It has been well known that larger or heavier seeds usually germinate faster than smaller or lighter seeds of the same species [17] . Because thermal time model is based on germination rate, we presume that thermal model parameters may differ for two contrasting chickpea cultivars varying in their seed size.
The objectives of this study were to test the assumption of the thermal model using two contrasting chickpea cultivars and to determine the effect of thermal requirement on germination and early growth of this crop.
2. Material and Methods
Two chickpea cultivars were obtained from the Agricultural Research Corporation (ARC) in Sudan, namely; Shendi (kabuli-type) and Jabel Marra (desi-type). Shendi cultivar; large seeded cultivar is described as a late mature and small seeded variety and the other cultivar (Jabel Marra); small seeded cultivar is described as an early mature variety. Germination tests of fresh mature seeds were carried out by putting seeds in Petri dishes with two layers of moistened filter papers (Whatman, No.1). The Petri dishes were randomly put in six incubators (Sanyo Versatile Environmental Chamber MLR-350 H), six temperatures from 10˚C to 35˚C, at 5˚C interval, were randomly assigned to each incubator. The relative humidity inside the incubator was 32% and the photoperiod is 12 hr with 30 µmol photons. m−2・s−1, 400 - 700 nm. A randomized complete design (RCBD) was used with four replicates of 25 seeds each, at 7-day interval between replicates in each incubator. Clear plastic bags were used to seal Petri dishes to reduce water evaporation. Seeds were considered germinated when 2 mm of radicle was visible [18] .
Final (cumulative) germination percentages were transformed using log10 Probit [18] [19] . Probit transformation linearizes the cumulative normal distribution, which facilitates modeling efforts [18] [20] . For modeling purposes a seed population was considered to be composed of subpopulations based on relative germination rate [18] [21] . The Probit procedure in SPSS, version (15) was used to estimate germination time or the 10%, 20%, 30%, 40%, 50%, 60%, 70% and 80% subpopulations. Pearson Chi square test was used as the goodness-of-fit test (p ≥ 0.10). The base temperature (the minimum temperature at which germination could occur) is generally assumed to be constant for a specific species. The base temperature (Tb) was determined by plotting germination percentage (on a probit scale) against log-thermal time (log10(T − Tb)tg), where Tb is unknown and estimated by changing the value of Tb until the minimum residual variation is obtained [22] . On a plot of probit (germination) against log (thermal time), the median thermal time required for seed of the population corresponds to the thermal time when probit (germination) = 5. The inverse of the slope of the fitted line is the standard deviation of the population responses to thermal time (i.e. the flatter the slope of the fitted line, the greater variation in response to thermal time between individual seeds).
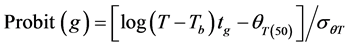 [18]
[18]
where Tb ≡ base temperature, 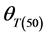 ≡ median thermal time, and
≡ median thermal time, and 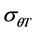 ≡ standard deviation for thermal time.
≡ standard deviation for thermal time.
These parameters were estimated for each seed size class of each cultivar for the construction of the thermal time model. General linear model (GLM) in SPSS was used to test differences in base temperature (Tb) between subpopulations and the effects of genotype and seed classes on thermal model parameters (Tb, 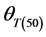 and
and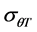 ). Mean were separated with Fisher’s protected least significant difference (FPLSD). Accuracy of thermal time model was measured by coefficient of determination (R2). R2 was calculated as: 1-(SS residual/SS total), where, SS residual represents residual sum of squares and SS total is the total sum of squares, while SS residual = observed value − predicted value and SS total = observed value − mean. Germination rate index (GRI) which is independent on thermal time model, is calculated as:
). Mean were separated with Fisher’s protected least significant difference (FPLSD). Accuracy of thermal time model was measured by coefficient of determination (R2). R2 was calculated as: 1-(SS residual/SS total), where, SS residual represents residual sum of squares and SS total is the total sum of squares, while SS residual = observed value − predicted value and SS total = observed value − mean. Germination rate index (GRI) which is independent on thermal time model, is calculated as:
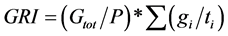 [18]
[18]
where Gtot is the total number of germinated seeds in a dish at the end of the germination test; P is the total number of seeds incubated; gi is the number of germinated seeds accumulated between time (d) 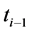 and ti. The effect of cultivars on GRI was also analyzed using general linear model (GLM) in SPSS (version 16).
and ti. The effect of cultivars on GRI was also analyzed using general linear model (GLM) in SPSS (version 16).
Temperature tolerance index (TTI) was calculated as total plant (shoot + root) dry weight at different temperatures compared to the total plant dry weight obtained at optimum temperature as indicate below:
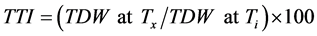 [23]
[23]
where TDW = total dry weight; Ti = control treatment (at optimum temperature); Tx = x treatment (at corresponding temperature).
Statistical Analysis:
Hundred Seeds of each cultivar were put in incubators at six different temperatures and arranged in a complete randomized design (CRD) with four replicates. If there was a significant difference at p < 0.05, the means separation was done for different treatments using least significant (LSD) method. To validate the thermal time model, correlation test was done and accordingly, the coefficient of determination (R2) and level of significance (P) were calculated. All statistical tests were done using a statistical package (SPSS version 16). Figures were constructed by using the computer software (Excel).
3. Results
The final germination percentage significantly increased with increasing temperature up to 25˚C, and thereafter there was a sharp decrease in final germination at 30˚C and 35˚C (Figure 1). The cultivar Jabel Marra (small seeded type) significantly recorded higher germination percentage than Shendi cultivar (large seeded type) at all temperatures (Figure 1).
Germination rate (d−1) of subpopulations, GR(g), calculated from the predicted germination time course correlated strongly (R2 = 0.92, P < 0.01) with temperature at the suboptimal range 10˚ to 25˚C (Figure 2). Base temperature was in the range between 1˚ to 3˚C, and significantly differed between the two cultivars according to their seed size (Figure 2) the large seeded cultivar (Jabel Marra) obtained higher germination rate at all sup- population and different temperatures (Figure 2(b)) compared with other cultivar (small seeded cultivar “Shensi”) (Figure 2(a)). Table 1 showed that the mean Tb for cultivar Shendi (1.2˚C) was about 1.8˚C lower than that of Jabel Marra (3.0˚C). Also, there were significant differences in 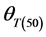 and
and 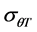 between the two cultivars (Table 1). The small seeded cultivars (Jabel Marra) scored higher thermal requirements compared with the large seeded cultivars.
between the two cultivars (Table 1). The small seeded cultivars (Jabel Marra) scored higher thermal requirements compared with the large seeded cultivars.
Germination rate index was significantly different between the two cultivars due to the difference in their seed sizes at various temperatures (Figure 2)., and GRI was dependent on incubation temperature (Figure 3). The large seeds germinate faster than small seeds for both cultivars (p < 0.05), and cultivar “Jabel Marra” (Arman or kabuli type) germinated faster than the other cultivar “Shendi” (Pirooz or desi type) (Figure 3).
The mean total dry weight was significantly different between the two cultivars at different temperatures (Table 2). The cultivar (Shendi) obtained higher shoot and root dry weight than the other cultivar Jabel Marra. Increasing temperature significantly increased the total dry weight from 10˚ to 25˚C, but at 30˚ and 35˚C, the dry matter was decreased significantly for both cultivars. The TTI was followed the same trend as total dry weight, and revealed that at 10˚C, the effect of low temperature was very pronounced in dry matter, only there were 75% - 70% decrease in dry matter compared with that at optimum temperature (Table 2). However, cultivar Jabel Marra scored lower dry matter and TTI compared to the other cultivar Shendi at all temperature studied (Table 2).
4. Discussion
It has long been known that seeds can germinate over a wide range of temperatures, but maximum germination and rate of germination typically reduce at the extremes of the range [24] - [26] . Recently, [27] have developed
![]()
Figure 1. The effect of constant temperatures on the final seed germination percentage of two chickpea cultivars. Each data point represents a mean of four replicates of 25 seeds each.
![]()
![]()
Figure 2. Germination rate (d−1) within the range of suboptimal temperatures as function of temperature and germination percentage or subpopulation (10%, 20%, 30%,…..etc) in two chickpea cultivars (a) Shendi and (b) Jabel Marra. The base temperature point was manipulated according to each germination subpopulation line.
![]()
Table 1. Parameter estimates in thermal time equation to fit germination progress curves of two chickpea cultivars in water at suboptimum temperatures (10˚, 15˚, 20˚C) and optimum temperature (25˚C).
Tb(0): base temperature, θT(50): median thermal time, σθT: Standard deviation of thermal time. *R2: Coefficient of determination; **p-value: level of significance.
![]()
Table 2. The mean total (shoot and root) dry weight (TDW) and temperature tolerance index (TTI) in two chickpea cultivars grown at different temperatures.
![]()
Figure 3. Germination rate index (GRI) of two chickpea cultivars seeds: Shendi, Jabel Marra, incubated at temperatures from 10˚ to 25˚C. Bars indicate standard deviations.
and modified the thermal time model (which first proposed by [13] ) to corporate suboptimal and super optimal temperatures. This study was conducted to assess the effect of genotypes and seed size on thermal requirement for germination. Our results revealed that although there were considerable difference between the two cultivars in all germination parameters, the thermal time requirement for 50% germination (![]() ) and standard deviation of thermal time (
) and standard deviation of thermal time (![]() ) were also significant (p < 0.05) between the two cultivars. On the other hand, the base temperature was significantly lower in large seeded cultivars (Shendi) than that of small seeded cultivars (Jabel Marra), allowing large seeds to accumulate more thermal time units than small seeds at the same temperature and subsequently to germinate faster. Our results were in accord with [18] but did not agree with the assumption that base temperature had little variation among genotypes of the same species [12] [27] [28] . The reason that Tb was often considered as a constant may be for the ease of modeling [29] . Similar to this experiment, [30] reported that base temperature in the thermal time did not appear stable for wild species due to distinct genetic variability within the population. The variation in base temperature between seed size classes of chickpea may favor large seeds, allowing rapid germination and establishment. Poor emergence at low temperature is sometimes attributed to low seed quality, genotypes, disease resistance, and high thermal requirement [30] . According to the results of this study we recommend to introduce kabuli-types (the cultivar “Shendi”) in Northern Sudan, and sowing should be done in the early winter (September-October) when temperature is around 20˚C - 25˚C to guarantee uniform and rapid germination and a good seedling establishment.
) were also significant (p < 0.05) between the two cultivars. On the other hand, the base temperature was significantly lower in large seeded cultivars (Shendi) than that of small seeded cultivars (Jabel Marra), allowing large seeds to accumulate more thermal time units than small seeds at the same temperature and subsequently to germinate faster. Our results were in accord with [18] but did not agree with the assumption that base temperature had little variation among genotypes of the same species [12] [27] [28] . The reason that Tb was often considered as a constant may be for the ease of modeling [29] . Similar to this experiment, [30] reported that base temperature in the thermal time did not appear stable for wild species due to distinct genetic variability within the population. The variation in base temperature between seed size classes of chickpea may favor large seeds, allowing rapid germination and establishment. Poor emergence at low temperature is sometimes attributed to low seed quality, genotypes, disease resistance, and high thermal requirement [30] . According to the results of this study we recommend to introduce kabuli-types (the cultivar “Shendi”) in Northern Sudan, and sowing should be done in the early winter (September-October) when temperature is around 20˚C - 25˚C to guarantee uniform and rapid germination and a good seedling establishment.
Acknowledgements
The authors are grateful for funding received from Prof. Mustafa Salih, University of King Saud.