This paper essentially deals with the formation of spherical TiO2 particles by means of the introduction of Triton X-100. This technique has been well known in order to provide simple and inexpensive alternative routes to synthesize nanoparticulate materials.
2. Experimental
Titanium tetrachloride was used as the starting material to prepare the TiO2 films. All the chemicals were of analytical grade and used without further purification. The influence of Triton X-100 was examined in two steps: (1) the preparation of an aqueous solution of TiCl4 (hydrolysis step), and (2) the precipitation of Ti (IV) polymers from the hydrolyzed TiCl4 solution onto a conducting substrate (precipitation step) [14]. In the hydrolysis step, 22 mL of TiCl4 was added dropwise to 78 mL of distilled water containing 1.3 mM Triton X-100 to produce a 2.0 M Ti (IV) solution. Before the precipitation process, fluorine-doped tin oxide (FTO) conducting glass plates (TCO glass, fluorine-doped SnO2 overlayer, transmission >70% in visible and sheet resistance 20 W/square) were first cleaned with water and ethanol. In the precipitation step, 1.0 ml of the hydrolyzed 2.0 M Ti (IV) solution were pipetted into the conducting glass plates. The plates were dried at 353 K in order to allow the Ti (IV) polymers to deposit onto the conducting glass plates. Subsequently, the glass plates were annealed at 623 K for 1 h. The TiO2 films thus obtained were coated with 0.3 mM of Orange IV in absolute ethanol for 24 h at room temperature. Carbon counter electrode was prepared by sputtering a thin layer of carbon on a transparent conducting glass support (TCO glass, fluorine-doped SnO2 overlayer, transmission >70% in visible and sheet resistance 20 W/square) using a graphite rod. The carbon electrode was placed over the dye-coated electrode, and the edges of the cell were sealed. A redox electrolyte consists of a mixture of iodide/triiodide in acetonitrile was injected by a syringe into the space between the two electrodes. The resulting cell had an active area of 1 × 2 cm2. A schematic diagram showing the construction of the cell is represented in Figure 1.
The photocurrent-voltage (I - V) curves were obtained using a digital source-meter (Keithley 2400). Direct sun light was used to illuminate the working electrode. The surface morphology was examined using SEM (Jeol JXA-840 electron probe microanalyzer). The XRD measurements were carried out with Diano Corporation USA diffractometer using Co radiation radiation. The surface areas were measured using a conventional or classical
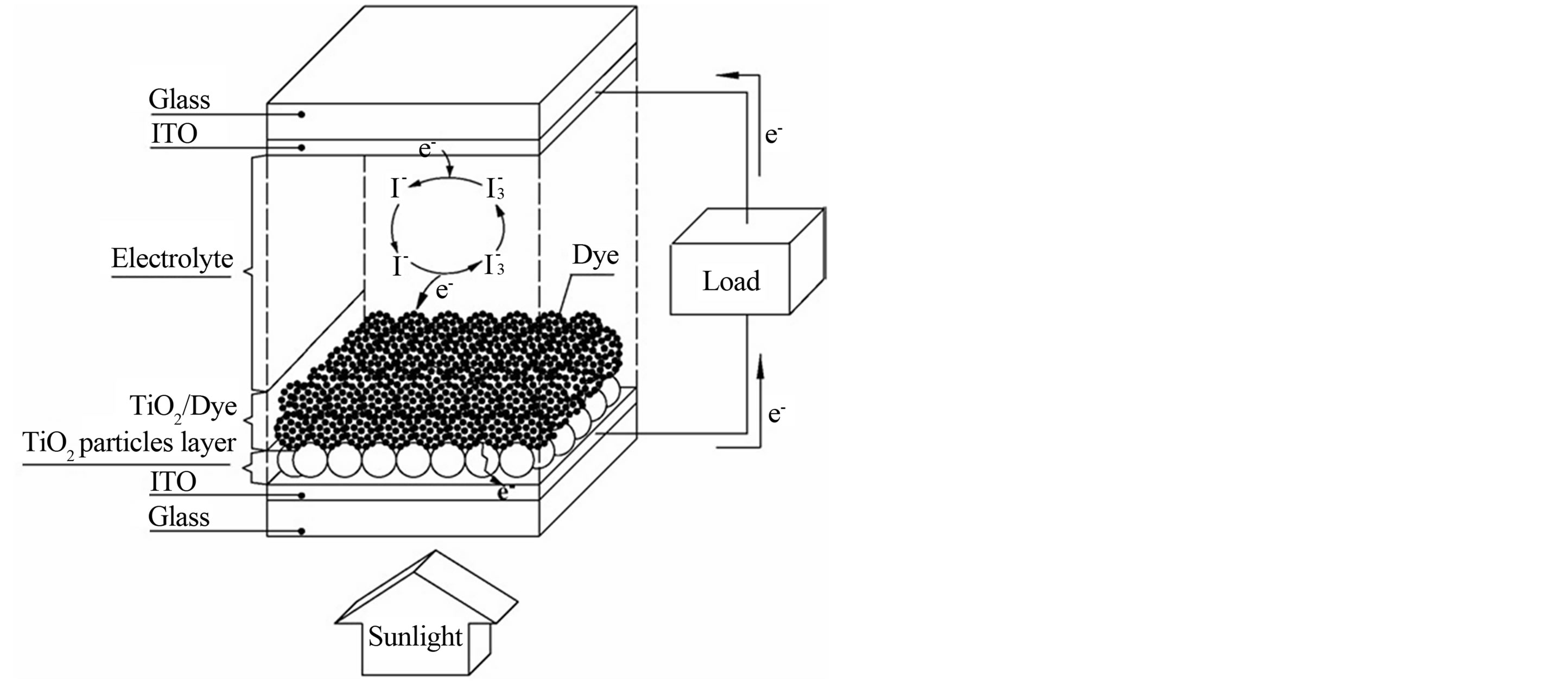
Figure 1. Schematic of the dye-sensitized solar cell with a sandwich TiO2 thin-film.
volumetric apparatus. FTIR spectra were taken using Thermo Avtar 370 spectrometer.
3. Results and Discussion
Figure 2 shows the influence of Triton X-100 on the XRD patterns of the TiO2 films, deposited on FTO. It can be seen that the TiO2 film formed with Triton X-100 completely crystallize as anatase (DB card, No. 01–070- 7348).
The average crystallite size of the anatase TiO2 was calculated using the Scherrer equation [15]. The determined average size of the prepared TiO2 was found to be 60 nm.
FT-IR studies of the TiO2 film show the characteristics of the formation of high-purity material. The FT-IR spectrum of Figure 3 clearly shows the peaks corresponding to TiO2. Peaks located in the area of 400 - 650 cm−1 correspond to the vibration of Ti-O and Ti-O-O bonds [16]. It is also clear from data of Figure 3 the presence of peak at 2900 cm−1 corresponds to –CH2 and at 1500 cm−1 corresponds to C-O-C due to organic moiety of Triton X- 100.
Figure 4 shows the top view SEM image of clusters of the film. The Triton X-100 influenced clusters have a uniform size and consist of well defined spherical TiO2 particles. The clusters composed of these particles tended to fuse together to form large aggregates.
The BET surface areas were 31.1 and 44.6 m2∙g−1 for the TiO2 powders (without and with Triton) and TiO2 formed with Triton, respectively. This increase in surface area was further evidenced by the observation that the amount of dye desorbed from the Triton influenced TiO2 film surface higher than that from the film without the Triton. This observation is consistent with the observation derived from XRD pattern that Triton facilitates
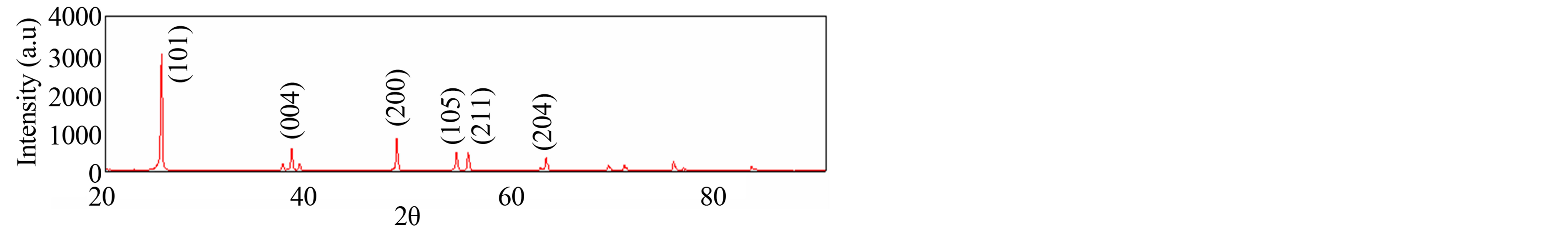
Figure 2. X-ray diffraction patterns of TiO2 electrode.


Figure 4. SEM images of the prepared TiO2.
smaller TiO2 particles.
Form the above results; we expected an enhanced performance of a DSSC fabricated with a Triton-influenced TiO2 film. The rationale behind this expectation lies in the regular structure of the film, composed of spherical particles and clusters. Figure 5 and Table 1 present the photocurrent-voltage (I - V) curves and photovoltaic performance data of DSCs fabricated with the TiO2 film electrodes in the presence and absence of Triton. The DSSC fabricated using the Triton-influenced TiO2 film shows a higher short-circuit photocurrent (Isc), opencircuit voltage (Voc) and solar conversion efficiency than that prepared without Triton. The increase in Isc may be

Figure 5. I - V curves of the DSSCs fabricated with TiO2 films and orange IV dye.

Table 1. Photovoltaic parameters of Orange IV dye-sensitized nanocrystalline TiO2 solar cells.
primarily related to the increased surface area. Furthermore, this increase in Isc can be related to an increase in the number of pathways due to the increased packing of round particles [14]. A more densely packed film improves inter-particle electrical contact. The observed increase in Voc might be due to an increase packing density of the TiO2 particles in the film. Increasing packing of small and round TiO2 particles produce small pores. The effective concentration of  ions in the TiO2 film with smaller pores would be lesser than that in the film with larger pores, because the rate of
ions in the TiO2 film with smaller pores would be lesser than that in the film with larger pores, because the rate of  ion replenishment in the TiO2 film with smaller pores becomes slower. This slow replenishment of
ion replenishment in the TiO2 film with smaller pores becomes slower. This slow replenishment of  ions leads to the accumulation of photo-injected electrons in the TiO2 conduction band in the open-circuit condition, which in turn leads to an increase in the Voc.
ions leads to the accumulation of photo-injected electrons in the TiO2 conduction band in the open-circuit condition, which in turn leads to an increase in the Voc.
4. Conclusion
Spherical TiO2 nanoparticles were obtained using Triton X-100 in a hydrolyzing of TiCl4 solution. XRD results reveal that Triton generally suppresses the formation of anatase phase of TiO2. The DSSC prepared with TiO2 film using Orange IV as sensitizer showed improved Isc, and Voc as well as substantially improved solar energy conversion efficiency, compared with those of a cell fabricated with TiO2 film obtained without Triton.
Acknowledgements
The authors wish to thank Research Units in Science College at University of Dammam for their support and cooperation through this work.
NOTES