Identifying Critical Parameters in SIR Model for Spread of Disease ()
1. Introduction
A system of kinetic equations can be applied to describe the behaviours of biochemical kinetics. The system is obtained from the reaction mechanisms by the law of mass action. The rate constant of any given elementary reaction, known as the proportionality constant, depends on the reaction condition (temperature, solvent, PH, etc.). The reaction conditions are generally held by biochemists to avoid dealing with higher-order complexities [1] [2] [3] .
An arbitrary number 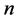 of elementary reactions can be expressed as follows:
of elementary reactions can be expressed as follows:
 (1)
(1)
where  is the
is the 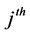 species and
species and 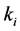 is the rate constant of step
is the rate constant of step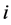 . The non-negative integers
. The non-negative integers 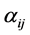 and
and 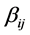 are the stoichiometric coefficients for species j occurring as a reactant and product, respectively, in the
are the stoichiometric coefficients for species j occurring as a reactant and product, respectively, in the  reaction step. The rate of product of species j can be given by:
reaction step. The rate of product of species j can be given by:
 (2)
(2)
where  is the concentration of
is the concentration of 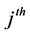 species and
species and  is the rate of change of
is the rate of change of  species in reaction
species in reaction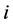 . A reaction rate
. A reaction rate 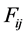 is given by the law of mass action as follows:
is given by the law of mass action as follows:
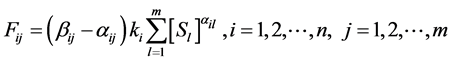 (3)
(3)
The readers can see further details about the chemical kinetics in [4] - [12] .
The idea of sensitivity analysis has been used in dynamical analysis of biochemical kinetics and systems biology models. The concept of sensitivity analysis theory in application to chemical kinetic problems was given by Rabitz in [13] . The local and global methods of sensitivity analysis in chemical kinetics were further studied in [14] . There are also some applications of sensitivity analysis of systems biology models in [15] - [21] . More recently, the methods of local sensitivity analysis have been used to identify the critical model elements for a kinetic model of nuclear receptor signalling, the reader can see further details about the method in [10] [11] . This method is applied to determine which variable or parameter is sensitive to a particular condition which is defined by a variable or parameter. The system of ODEs discussed here is:
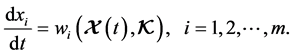 (4)
(4)
The model input ![]() is a vector of parameters, and the model output
is a vector of parameters, and the model output ![]() is a vector of state variables. Local sensitivity is the changes in state variables
is a vector of state variables. Local sensitivity is the changes in state variables ![]() with respect to parameters
with respect to parameters![]() .
.
The general form of the local sensitivity is given as a Jacobian matrix as follows
![]() (5)
(5)
where the matrices ![]() and
and ![]() are defined by
are defined by
![]()
The initial conditions of the Equation (5) are determined by the input parameter ![]() and the initial condition of the output variables
and the initial condition of the output variables![]() .
.
The idea of ELzaki transform was introduced by Tarig Elzaki for solving linear ordinary differential equations with constant coefficients. Then, this became more popular and works as a powerful tool in mathematics for solving some complicated equations, for example, solving high order ordinary differential equations with variable coefficients [22] , applying ELzaki transform for solving the integro-differential equations [23] , showing some relationships between Laplace transform and the ELzaki transform [24] , solving linear systems of integro-differential equations with constant coefficients [25] , a solution for nonlinear systems of differential equations using a mixture of Elzaki transform [26] .
The SIR model, first published by Kermack and McKendrick in 1927 [27] , is surly the most well known mathematical model for the spread of an infectious disease. Then, the model was further developed mathematically in [28] [29] . Here, people are divided into three classes: susceptible S, infective I and removed R. Removed individuals are no longer susceptible nor infective for whatever reason. For instance, they have recovered from the disease or they have been vaccinated. They may have been isolated from the rest of the people [30] [31] [32] . After that further explanation of the model was proposed by the authors of these papers [33] [34] [35] [36] [37] .
The main problem in this study is identifying the critical model parameters. Therefore, the aim in this work is to apply some mathematical tools to simplify and analyse the model and then identifying the model elements (variables and parameters). We proposes a number of steps of model analysis, which plays in reducing the number of elements and in calculating analytical approximate solutions of the SIR model. The proposed steps and their advantages are simply given. The first step is that a proper scaling is used in order to minimize the number of elements. This becomes a good step forward for simplifying the original model. Another step is calculating some analytical approximate solutions using Elzaki transformation. Furthermore, using local sensitivity method is another important step in this study. This helps us to identify critical model parameters of the reduced model. Finally, studying the behaviour at infinity of the reduced model provide us an understanding of global dynamics and drawing the global phase portrait of the system.
2. The SIR Epidemic Disease Model
The SIR model may be diagrammed as in Figure 1. The chemical reactions of the model are given
![]() (6)
(6)
where ![]() is the rate at which susceptible people become infectious and
is the rate at which susceptible people become infectious and ![]() is the rate at which infectious people recover/develop immunity, both parameters are positive constant reactions. By using mass action law, we have the corresponding coupled differential equations
is the rate at which infectious people recover/develop immunity, both parameters are positive constant reactions. By using mass action law, we have the corresponding coupled differential equations
![]() (7)
(7)
![]()
Figure 1. Scheme of the SIR infectious diseases model. Geometric shapes here represent compartments, and arrows indicate flux between the compartments.
with initial conditions
![]() (8)
(8)
Equation (7) has a conservation law
![]() (9)
(9)
where ![]() is a constant population. We assume in this study the population is closed with no death or births, or immigration or emigration.
is a constant population. We assume in this study the population is closed with no death or births, or immigration or emigration.
The dimensionless SIR equations are then given by
![]() (10)
(10)
where ![]() and
and ![]()
Therefore, the conservation law (9) takes the form
![]() (11)
(11)
In addition, the Equation (10) is then reduced to
![]() (12)
(12)
with initial conditions ![]() and
and![]() . It can be concluded that the simplified Equation (12) is minimal in terms of the number of its elements (variables and parameters).
. It can be concluded that the simplified Equation (12) is minimal in terms of the number of its elements (variables and parameters).
Many nonlinear systems of differential equations have not exact solutions. Calculating analytical approximate solutions for such systems is a difficult task. The SIR epidemic disease model is given as the non-linear system of ODE’s. Equation (12) can not be solved analytically. Therefore, calculating some analytical approximate solutions for the system provides more information about the behaviours of the model dynamics. Applying the idea of Elzaki transform, we can obtain some analytical approximate solutions of the Equation (12). Take Elzaki transform of Equation (12) to get:
![]() (13)
(13)
Take the inverse Elzaki transform of Equation (13), the recursive relations are then given by
![]() (14)
(14)
where
![]() (15)
(15)
For![]() , and from Equation (15) and Equation (14), we have
, and from Equation (15) and Equation (14), we have
![]() (16)
(16)
For![]() , and from Equation (15) and Equation (14), we have
, and from Equation (15) and Equation (14), we have
![]() (17)
(17)
where,
![]() ,
, ![]()
![]()
![]()
For![]() , and again from Equations ((15) and (14)), we obtain
, and again from Equations ((15) and (14)), we obtain
![]() (18)
(18)
where
![]()
![]()
![]()
![]()
![]()
In general, a series form of the model solution can be given
![]() (19)
(19)
3. Proposed Steps of Model Analysis and Simplification
In this work, we propose some steps of model analysis and simplification for the SIR epidemic disease model. The suggested steps are used for minimizing the number of elements (variables and parameters), calculating analytical approximate solutions and identifying critical model parameters. The proposed steps in this study are presented below:
1) Define chemical mechanisms of SIR Model.
2) Define a kinetic model of biochemical reactions as a system of ODEs using the mass action law.
3) Eliminate some variables based on the stoichiometric conservation laws, and use proper scaling for the kinetic equations and determine the minimal number of parameters.
4) Apply Elzaki transform method to calculate analytical approximate solutions of the reduced model.
5) Simulate the reduced model dynamics for different parameter values using Pplane8 for Mathlab. Analyse the reduced models to identify the critical model parameters by the local sensitivity analysis in numerical simulations using the SimBiology Toolbox for Mathlab.
6) Study the global behaviours of the reduced model at infinity.
The above steps can be simply presented in the following flowchart:
![]()
The flowchart of proposed steps of model analysis and simplification, the steps are presented in the order of their application.
4. Local Behaviours of the Simplified Model
We use Pplane8 for Mathlab to study the stability analysis of the Equation (12) and to compute numerical simulations in ![]() -plane, we also identify model nullclines; see Figure 2. It can be concluded that the model has the same stability properties at different values for the parameter used.
-plane, we also identify model nullclines; see Figure 2. It can be concluded that the model has the same stability properties at different values for the parameter used.
Furthermore, we identify the model interacting and numerical simulations in two and three dimensional planes, for different values of![]() ; see Figure 3 and Figure 4. We notice that the dynamics of susceptible and infective people go to stable state very quickly as
; see Figure 3 and Figure 4. We notice that the dynamics of susceptible and infective people go to stable state very quickly as ![]() becomes larger. Results are given here by using Pplane8 for Mathlab.
becomes larger. Results are given here by using Pplane8 for Mathlab.
In addition, we calculate the local sensitivity of state variables ![]() and
and ![]() with respect to the given parameter
with respect to the given parameter ![]() to identify critical mode parameters. We identify that infective people are more sensitive to the constant rate
to identify critical mode parameters. We identify that infective people are more sensitive to the constant rate ![]() when
when ![]() while
while
![]()
Figure 2. Dynamics of the Equation (12) and numerical simulations using Pplane8 for Mathlab in ![]() -plane for different values of parameter
-plane for different values of parameter![]() , red lines stand for model nullclines.
, red lines stand for model nullclines.
![]()
Figure 3. Dynamics of the simplified model and numerical simulations using Pplane8 for Mathlab in two dimensional plane, for different values of parameter![]() .
.
they are less sensitive to the given parameter ![]() as
as![]() . More interestingly, both susceptible and infective people have same sensitivity to
. More interestingly, both susceptible and infective people have same sensitivity to ![]() when
when![]() ; see Figure 5. Results here are computed in numerical simulations using the SimBiology Toolbox for Mathlab in the time interval
; see Figure 5. Results here are computed in numerical simulations using the SimBiology Toolbox for Mathlab in the time interval ![]() unites of time. Identifying critical model parameters in this study is a good step forward for describing and understanding the model dynamics in systems biology.
unites of time. Identifying critical model parameters in this study is a good step forward for describing and understanding the model dynamics in systems biology.
5. The Global Behaviours of the Simplified Model
This section is devoted to examine the global phase portrait for Equation (12) by studying the isoclines and the behaviours at infinity. The isoclines are the lines with equal slope. These lines are an important role in sketching the phase portrait. It is easy to know where the trajectories have vertical and horizontal tangent lines by finding the
isoclines for ![]() and
and![]() . There are no motion horizontally and vertically when
. There are no motion horizontally and vertically when ![]() and
and ![]() respectively. The vertical trajectories are given by
respectively. The vertical trajectories are given by ![]() which are
which are ![]() and
and![]() . The horizontal trajectories are given by
. The horizontal trajectories are given by ![]() which are
which are![]() . From above, we note that
. From above, we note that ![]() is an invariant line and
is an invariant line and ![]() is a line of singularities. Since the Jacobian matrix at each points on the line of singularities,
is a line of singularities. Since the Jacobian matrix at each points on the line of singularities, ![]() , are 0 and
, are 0 and![]() , therefore the singular points that are belonging to the left side of the vertical line,
, therefore the singular points that are belonging to the left side of the vertical line, ![]() , are stable and on the other side are unstable.
, are stable and on the other side are unstable.
Here, we study the direction of trajectories in the quadrants. In the first quadrant,
![]()
Figure 4. Dynamics of the simplified model and numerical simulations using Pplane8 for Mathlab in three dimensional plane, for different values of parameter![]() .
.
since ![]() and
and ![]() are positive, then
are positive, then ![]() is always negative,
is always negative, ![]() is negative where
is negative where ![]() and
and ![]() is positive where
is positive where![]() . In the second and third quadrants,
. In the second and third quadrants, ![]() and
and ![]() respectively. In the fourth quadrant,
respectively. In the fourth quadrant, ![]() is always positive, but
is always positive, but ![]() is positive where
is positive where ![]() and
and ![]() is negative where
is negative where![]() .
.
The second required one is the line at infinity. It is a projective line that is added to the affine plane. Finding them including singular points and studying the behaviours at infinity of the reduced Equation (12) is very important to an understanding its global dynamics. For this purpose, the two below nonlinear change of variables are used individually.
![]() (20)
(20)
![]() (21)
(21)
Salih in [38] have used above transformation in three dimensional case. The points ![]() and
and ![]() where
where ![]() and
and ![]() vanish are obtained from the nonlinear change of coordinates (20) and (21) respectively. These are the singular points of the new system that are corresponding to the singular points at infinity for simplified Equation (12).
vanish are obtained from the nonlinear change of coordinates (20) and (21) respectively. These are the singular points of the new system that are corresponding to the singular points at infinity for simplified Equation (12).
Applying the nonlinear change of variables (20) on the reduced Equation (12) and after rescaling of the variables, the new system is obtained
![]()
![]() (22)
(22)
The above system has two singular points ![]() and
and![]() . The first one is the intersection point of the line at infinity
. The first one is the intersection point of the line at infinity ![]() and
and ![]() axis, the system at that point has Jacobian matrix
axis, the system at that point has Jacobian matrix
![]() (23)
(23)
with a positive ![]() and one zero eigenvalues, therefore the singular point is unstable. The Jacobian at the second singular point
and one zero eigenvalues, therefore the singular point is unstable. The Jacobian at the second singular point ![]() is given by
is given by
![]() (24)
(24)
with two negative eigenvalues ![]() and
and![]() , the singular point is stable.
, the singular point is stable.
The system below is obtained by applying the nonlinear change of variables Equation (21) on Equation (12) after rescaling of variables
![]()
![]() (25)
(25)
System (25) has only two singular points ![]() and
and![]() . At the first singular point
. At the first singular point![]() , Equation (12) has Jacobian matrix
, Equation (12) has Jacobian matrix
![]() (26)
(26)
which it has one negative eigenvalue ![]() and a zero eigenvalue. At the second singular point
and a zero eigenvalue. At the second singular point![]() , Equation (12) has Jacobian matrix
, Equation (12) has Jacobian matrix
![]() (27)
(27)
which it has two positive eigenvalues ![]() and
and![]() . Figure 6 shows the dynamics behaviours at an affine plane and also at infinity, which exams a good understanding of the global behaviours of the Equation (12). We note that, the sum of the ratio of eigenvalues of either line is unity according to an index formula Lins Neto [39] .
. Figure 6 shows the dynamics behaviours at an affine plane and also at infinity, which exams a good understanding of the global behaviours of the Equation (12). We note that, the sum of the ratio of eigenvalues of either line is unity according to an index formula Lins Neto [39] .
6. Conclusion
Mathematical presentations and numerical simulations of infectious disease models are crucial topics in systems biology. Describing the dynamics of such systems often requires some techniques of model analysis. We studied an epidemic disease model called SIR model with three species and two parameters. We proposed a number of steps of
![]()
Figure 6. Global phase portraits of the simplified Equation (12); the green solid curves depict the line at infinities, the green dotted lines depict unstable singular points and the red dotted line depict stable singular point.
model analysis. The suggested steps of model analysis here importantly play in reducing the number of elements and in calculating analytical approximate solutions of the SIR model. The simplified model helps to study the full model of SIR in different ways. Firstly, identifying the critical model parameters of the reduced model becomes much easier compared to the full model. Secondly, the mathematical representation of the reduced model can help to integrate experimental knowledge into a coherent picture. Furthermore, studying the behaviour at infinity of the reduced model helped us to understand its global dynamics and to draw the global phase portrait of the system. Finally, the reduced model could be accurate, robust, and applied by biologists for various purposes. The proposed techniques here of model analysis will be applied to a wide range of complex infectious disease models in systems biology.
Acknowledgements
We thank the Editor and the referee for their comments. We also thank both Raparin and Koya universities for providing us with wonderful facilities. This support is greatly appreciated.